Unlock The Magic Of A South African Summer – An English Essay
A South African summer is a magical time. The days are long and lazy, the weather is warm and sunny, and the evenings are cool and perfect for spending time with family and friends.
There is something about the summer that just makes everyone happy. Maybe it’s the fact that the days are so long that you can really make the most of them. Or maybe it’s the warm weather that just makes you feel good. Whatever the reason, a South African summer is a special time.
One of the best things about summer is the food. Fresh fruit and vegetables are in abundance, and there are so many delicious recipes to try. From salads and BBQs to braais and potjies, there is something for everyone. And of course, no South African summer is complete without a few cold beers or glasses of wine.
Another great thing about summer is the opportunity to get out and about. There are so many beautiful places to explore, and the long days mean that you can really make the most of them. Whether you’re hiking in the mountains, swimming in the sea, or just relaxing on the beach, there is something for everyone.
So, if you’re looking for a summer that is

The Magic Of A South African Summer English Essay
The magic of a South African summer is something that cannot be put into words. From the beauty of the bushveld to the rolling hills of the Cape Winelands, the country is a stunning visual feast of natural beauty. The warmth of the sun, the sound of the birds singing, and the sound of the ocean crashing against the shore all combine to make a truly mesmerizing experience. During the summer months, the days are long and the nights are filled with stars, making it the perfect time to relax and watch the sunset over the horizon. The vibrant colors of the flora and fauna, the lush green grass, and the abundance of wildlife are a reminder of the joys of living in one of the most unique places on earth. A South African summer is a special time of the year, and one that will be treasured for a lifetime.
The Culture: Discussion of the culture and traditions that define the South African summer.
The summertime in South Africa is a season of vibrant culture and tradition. It’s a time of joyous celebration, of feasting and family reunions, and of revelling in the beauty of the African continent. From the rolling hills of the Kruger National Park to the bustling streets of Johannesburg, South Africa’s summertime is full of unique cultural experiences and traditions that make it truly magical.
The celebration of the summertime in South Africa starts with the Zulu Reed Dance Festival. This traditional ceremony is held each year in August, when thousands of Zulu women aged 16 and above come together to perform traditional dances and songs in honour of the Zulu King. It’s a time of reverence, celebration and unity, with the festival taking place in the heat of the summer sun.
Another unique feature of the South African summertime is the Umhlanga or Reed Dance Ceremony. This is performed each year in August by the Zulu people, who come together from far and wide to celebrate the new harvest and the coming of the new year. The women of the tribe wear traditional clothing and carry reeds to perform traditional dances and songs in honour of the Zulu King. The festival is a vibrant display of South African culture and tradition, with the whole event coming alive with the sound of singing, music and laughter.
Finally, no South African summertime would be complete without a visit to the beach. South Africa has some of the most beautiful beaches in the world , and they offer a great opportunity to relax and have fun. Whether it’s beach cricket, surfing, or simply soaking up the sun, the South African coastline offers a wealth of activities to enjoy during the summer months.
The culture and traditions that define the South African summertime create a unique and magical atmosphere. From the Zulu Reed Dance Festival to the Umhlanga, from barbecuing to beach fun, South Africa’s summertime is full of culture and entertainment that are sure to delight. So come to South Africa and experience the culture and traditions that make the South African summertime so special.
The Wildlife: Description of the vast and diverse wildlife that is native to South Africa.
Ah, the magical and captivating beauty of a South African summer! With its lush landscapes, diverse wildlife, and endless sunshine, it’s no wonder that so many people flock to the rainbow nation to experience the joys of life in the southern hemisphere. From the stunning beauty of the Cape Floral Kingdom to the vastness of the Kalahari Desert, South Africa is an absolute haven for nature lovers.
The wildlife of South Africa is one of the country’s most treasured natural resources. With its unique biodiversity, South Africa is home to a wide variety of wildlife species – from the big five (lion, leopard, rhino, elephant, and buffalo) to the smallest of birds and insects. The big cats of South Africa , such as lions and cheetahs, are some of the most iconic and beloved of these creatures, as well as the majestic African elephants that roam the grasslands and savannahs.
South Africa is also home to some of the most distinct and diverse bird species in the world. From the brightly coloured Lilac-breasted Roller to the soaring Fish Eagle, there is something to delight bird watchers of all ages. And if you’re looking for something a little different, then why not take a trip to the coast and explore the many species of seabirds, such as gannets, albatross, and cormorants.
The marine life of South Africa is equally impressive. With its rich seas and abundant coral reefs, South Africa is a paradise for scuba divers and snorkelers alike. From the vibrant reefs of the Cape of Good Hope to the shallow waters of the Indian Ocean, the underwater world of South Africa is a stunning display of beauty and grace.
Whether you’re looking to explore the wildlife of South Africa on land or in the sea, the country offers a wealth of experiences for the intrepid explorer. From the majestic wildlife of Kruger National Park to the stunning beauty of the Cape Town coastline , South Africa is an absolute must-visit destination for anyone looking to experience the magic of a South African summer.
The Landscape: Explanation of the stunning landscapes and sceneries South Africa has to offer.
Ah, the magical beauty of a South African summer! From the stunning mountain peaks of the Drakensberg to the rolling hills of the Eastern Cape, South Africa offers a wealth of breathtaking landscapes and sceneries that never cease to amaze. From the majestic Karoo to the lush beauty of the Garden Route, South Africa is a land of incredible diversity.
The Drakensberg Mountains, also known as the ‘Mountains of the Dragon’, are a spectacular sight. With soaring peaks, deep valleys, and stunning waterfalls, the Drakensberg is a hiker’s paradise. On a clear day, the views from the top are simply breathtaking.
The Eastern Cape is a stunning province with rolling hills, wooded valleys, and pristine beaches. Here, one can explore the rugged coastline and sandy beaches, or take a leisurely drive through the rolling hills. The Eastern Cape is a great place to experience the beauty of South Africa’s diverse wildlife .
The Karoo is a vast semi-desert region of South Africa . With its rugged terrain and sparse vegetation, the Karoo is an exotic landscape that is unmatched in its beauty and tranquility. The Karoo is home to a wide variety of plant and animal life, making it a haven for nature lovers.
The Garden Route is a lush coastal strip along the south-western coast of South Africa . Perfect for a road trip , the Garden Route is dotted with stunning vistas and unspoiled beaches. With its picturesque villages and scenery, the Garden Route is a great place to explore and relax.
The Magic Of A South African Summer is a wonderful experience that everyone should experience at least once in their lifetime. The warm, sunny weather, the stunning scenery , and the friendly people make this a great place to visit. The only downside is that it is a bit hot and humid, but that is a small price to pay for the wonderful experience that is a South African summer.
Austin Finnan
Related articles, zambia on africa map, revealed: most popular xitsonga names for new born babies, discover the world’s most populous state or province.
- Activities & Things To Do
- Attractions
- Wildlife Encounters
- Restaurants and Eateries
- Reviews and Visitors’ Perspectives
- Travelling with Kids
- Travelling Tips
- Visitorials: We Love South Africa
- Weddings in South Africa
- Western Cape
- KwaZulu Natal
- Eastern Cape
- The Garden Route
- Limpopo Province
- North West Province
- Northern Cape
- South Africa Accommodation
- South Africa Hotels
- Guest Houses
- Bed & Breakfasts
- Self Catering Accommodation
- Interests & Special Needs
- Western Cape Accommodation
- KwaZulu Natal Accommodation
- Gauteng Accommodation
- Eastern Cape Accommodation
- Free State Accommodation
- Garden Route Accommodation
- Limpopo Accommodation
- Mpumalanga Accommodation
- North West Accommodation
- Northern Cape Accommodation
- Johannesburg
- Port Elizabeth
- Kruger Park
- Cape Winelands
- Drakensberg
- Garden Route
- Getting to South Africa
- Tourist Attractions
- World Heritage Sites
- Botanical Gardens
- Wildlife of South Africa
- Birdlife of South Africa
- Plant Life in South Africa
- Adventures & Outdoor Activities
- Restaurants in South Africa
- Things To Do
- South Africa’s Hiking Trails
- Game Reserves
- Health Resorts & Spas
- Golf Courses
- Wine Estates

10 Reasons Why You Should Visit South Africa this Summer
Visit South Africa this Summer ~ South Africa hots up between October and February.
On the highveld the heat of summer South Africa comes alive with afternoon thunderstorms that leave the earth rank with a smell uniquely African.
By contrast, down in the Western Cape summer is when it’s least likely to rain, when the Winelands are at their best , and when the Atlantic is at its warmest because the heat of the sun and sand have left you gasping.
On the lowveld it’s when most antelope species have their babies, when the bushveld is at its most lush and when afternoon siestas are a necessity to counteract the soaring temperatures.
But these are not the only reasons why you visit South Africa this Summer.
Here are 10 reasons to visit South Africa this summer …
The slopes of table mountain.
Early light in Cape Town means you can find yourself climbing to the top of Table Mountain as early as 5 am, when the day is still, and the clouds less likely to converge on the table in an effortless tablecloth.
The long summer days are practically European in nature in Cape Town, but you’ll want to make the top long before the sun heats up as it can become scorching, and the climb is tough for those who aren’t regular hikers.
There are many routes to the top. The fastest, but most steep, is Plattekloof Gorge. Go with a guide.

Summer Music Festivals
Summer South Africa is the time to be out of doors and its music festivals are a highlight, although some of them fall just outside of official summer. We list the best three:
Rocking the Daisies – usually in early October, Rocking the Daisies is a three-day multi-sensory experience named after the flowers of the Namaqualand.
Held just outside the little town of Darling (about an hour’s drive from Cape Town) the festival offers a diverse line-up of entertainment to kickstart summer.
Up the Creek Music Festival – live music comes with comedy and riverside fun on the banks of the Breede River in February every year – 4 days, 50+ bands, one lilo! Get together and celebrate summer with another 3 000 people just outside Swellendam .
Ultra Music Festival is an annual outdoor electronic music festival event (named after Depeche Mode’s album, Ultra ) that happens in Cape Town either at the Cape Town Stadium or The Ostrich Ranch, and Johannesburg at the Nasrec Expo centre (the original was in Miami, Florida and there are others just like it around the world) in early March.

The Lighthouse route
There’s no official lighthouse route but summer is the best time of year to visit these 10 lighthouses on more treacherous parts of the coastline between Paternoster , on the West Coast, and Durban on South Africa’s east coast.
It includes lighthouses at Port Edward, Port Alfred , Port Elizabeth and Gansbaai.

Rock Climbing in the Craggy Cederberg
Another fabulous reason to Visit South Africa this Summer is the Cederberg . It’s quite usual to see hardened rock climbers, their odd mattresses on their backs, heading from the car park just inside the Cederberg Wilderness, less than three hours from the traffic jams of Cape Town.
The Cederberg, most notable for its rooibos tea production, is now considered of the world’s top 10 destinations for bouldering (no ropes or mattresses and fairly close to the ground).
Rocklands is a 20 square kilometre area full of boulders and rocks perfect for the sport. And climbers stay on farms, campsites and in guest houses close by.

Surfing off the coast of the Eastern Cape
With names similar only for their use of the word ‘bay’ – Stillbay Victoria Bay, Jeffreys Bay and Cape St Francis – the Eastern Cape’s warm waters, balmy summer South Africa days make for superb surfing.
If you include the Wild Coast – Haga Haga , Morgan’s Bay and Mdumbe (which, according to some, rivals JBay) then you can spend weeks driving and surfing in these parts.

The old-growth forests of the Garden Route
The Garden Route with its sumptuous vegetation wins hands down when it comes to natural beauty in summer South Africa.
The combination of the Outeniqua and Tsitsikamma indigenous forests with their unique blend of Cape Fynbos and Temperate Forest are awash with hiking trails and routes.
A perfect reason to Visit South Africa this Summer.

Overnight hikes in the caves of the Drakensberg
The Drakensberg is a huge expanse of mountain roughly three hours northwest of Durban. It’s so vast that it’s divided into two main parts – the Midlands and the high Drakensberg.
It’s not only incredibly beautiful, but it’s also best explored on foot down the craggy ravines and up the soaring peaks.
Multi-day hikes are best and you will sleep either in a cave or in a hiking tent on a mountain. The caves are great for shelter and incredible views.
You can plan your own hike (book accommodation here ), join a hiking club or hire a guide.

Viewing loggerhead & leatherback turtles
Between November and March the loggerhead and leatherback turtles come to South Africa’s northeast shores in the iSimangaliso Wetland Park .
The perfect reason to Visit South Africa this Summer – Hatchlings emerge roughly 70 days later and these you can see too, if you plan your trip around summer.
Tours at iSimangaliso (nov to mar), hatchlings emerge after 70 days (jan to mar).

Driving two of the world’s most dramatic coastal roads
Chapman’s Peak links Cape Town’s Hout Bay to Noordhoek on the Atlantic Ocean. Only 9 km in length its effortlessly undulating curves and bends make it one of the most picturesque drives in the world.
Clarence Drive or Route 44 is yet another spectacular coastal road linking Gordons Bay with Betty’s Bay, an easy day’s drive linking nature reserves, seaside towns, a botanical garden and hiking trails.

Migrant birds
Over one hundred bird species head to South Africa during summer , flying routes across the world that cause one only to marvel at how such tiny creatures can navigate such journeys, sometimes from as far away as the Arctic Circle.
In summer South Africa not only does the list of migrants far exceed expectations, but the breeding plumage of some of the birds just adds to the delight.
There is no one best place for birds. The country is full of nature reserves and areas that best provide viewing, and we can recommend Kruger National Park , iSimangaliso Wetland Park, and Nylsvley Nature Reserve .

Explore South Africa More
- South Africa Attractions
- Things to Do in South Africa
Visit South Africa this Summer …
Further Reading...

Natures Valleys Best Kept Secrets

Get sassed about your fish when travelling – use the sassi cell phone app

Top 5 South African Cities
Best places to stay on the south coast, south africa’s all-year-round beach destination, escape to these quiet places in south africa, sa-venues.com.


More on News

Sri Lanka’s S.A.D.
3 weeks ago

Summer Surf Sale
1 month ago

South Africa Surf Trip with a Purpose
8 months ago

Meet the Team – Sri Lanka Surf House
11 months ago
Why We Love South African Summer: January in Cape Town From England to Cape Town

Monica Lewis 6 years ago in News
I know you don’t need an excuse to plan a surf trip this winter to escape the UK, but if you’re looking for one, here are 10 reasons to take advantage of the South African Summer…
1. The Weather
Let’s start in true British fashion and talk about the weather. The days are getting shorter, it’s time to say goodbye to fresh autumnal days and unwillingly welcome cold, windy winter. But this doesn’t have to be your routine for the next 4 months. Why not swap things up and chase the endless summer (even if it’s just for a bit)? South African summer runs from November-February, so expect beautifully hot, dry, sunny days. With average highs of 29° it’s too tempting to resist.
Let’s be honest, the waves are what we’re all here for. Due to Cape Town’s unique peninsula, throughout the South African Summer months, south easterly winds create amazing offshore conditions in the crystal blue waters of the Atlantic Coast. Then jump over to False Bay and you’ve got a whole new playground in the Indian Ocean. On the Cape Peninsula alone there are over 30 surf spots and waves for all abilities so everyone can get involved! Check out Wavescape for conditions.
3. Food and Drink
Everyone knows that when you’re on holiday you can eat whatever you like and it doesn’t count. South Africa takes eating and drinking to a whole new level, and for very reasonable prices. Famous for their Braai, a bit like a BBQ but better, you’ll have to experience to understand. Pair this with wine tasting in one of Cape Town’s many vineyards or a beer in the hot sun and you’ve got yourself the perfect evening. Make sure you get yourself to Noordhoek winery for a sundowner on a Thursday. You can thank me later.
4. Wildlife
You have traditional African animals on your doorstep and some unconventional ones too. Head Northeast for two hours and you’ll discover Aquila Private Game Reserve . These 10,000 hectares are home to the Big Five – Rhinos, Elephants, Lions, Leopards and Buffalo which were previously on the brink of extinction due to game hunters. Head to Aquila for a day trip or stay overnight for a whole new experience. Don’t miss the chance to see Zebras at the Cape of Good Hope as well. Now onto the unconventional African dweller, the penguin. Join the party at Boulders Beach and prepare to melt from their cuteness but never really shift the confusion of penguins in Africa.
As previously mentioned, the Cape of Good Hope is a great place to see Zebras, but even without this exotic animal it’s definitely worth going for the incredible views. Hiking up table mountain will take you a good 2 hours but you’ll be blown away by the beauty once you reach the top. Besides, you’ll be so surf-fit that that’s basically a walk in the park. Although, if you’re looking for a more cruisey walk and a beer whilst watching the sunset then head to Lion’s head for a spectacle.
It’s time to chill out again whilst watching some local sport and January is cricket season. At this point you’re either really excited or really not, there’s not really a middle ground when it comes to cricket. If it’s the latter then Newlands cricket ground might just change your mind. Imagine lazy days at a unique stadium with a mountain backdrop supporting the South African cricket team, the Proteas, it’s lush.
Photo credit: Sport 360
If sport isn’t your thing then check out the music scene. It’s not the first thing you think of but Cape Town is a buzzing city with a fix for every genre. From pop to rock, jazz to acoustic. There’s even the opportunity for an open mic night if you fancy it. Our favourite thing to do on a Sunday is head to the Kirstenbosch National Botanical gardens for a Sunday evening session. Set out a picnic in the shade of Table Mountain and enjoy. You’ll wish every day is a summer Sunday in South Africa.
For more music information check out Cape Town tourism
Photo credit: www.capetownetc.com
8. Community Projects
You’re imagining endless fun at this point, so why not give back to the community and share the love? Cape Town is home to some great charities and community projects that you won’t have to look hard to find something that interests you. Whether it’s building, education or teaching surfing, it’s covered. Check out Waves for Change for an insight into just one of the charities working to help the community in Cape Town.
“W4C provides a child-friendly mental health service to at-risk youth living in unstable communities. Through access to safe spaces, caring mentors, and a provision of weekly Surf Therapy sessions, W4C gives children skills to cope with stress, regulate behaviour, build healing relationships, and make positive life choices.”
We’ve skipped ahead a bit though. It’s obvious now that Cape Town has so much to offer, but you also need to get there first. This is where it all gets a little bit sweeter with flight prices. Normally a stress and big expense of your trip, but the beauty of Cape Town is that you can fly there direct from Heathrow or Gatwick in under 12 hours. No longer do you have to sit in an airport on a long layover trying not to look at the food you just can’t afford. Let’s double up on the good news too, it won’t cost you an arm and a leg. It’s cheaper than Indo and there are constant deals too. Check out Jack’s Flight Club – they love a good South Africa flight deal almost as much as we do!
10. The Potential for a Road Trip
You’ve made it here, you’ve experienced the epic lifestyle of Cape Town and your wanderlust cannot be contained. This amazing South African city is the perfect gateway to an epic road trip. Why not join us on an immense 10 week road trip travelling 1000 miles along the coast to Durban, exploring places you couldn’t even dream of and surfing world class surf spots such as Jeffreys Bay. You can even pass your surf instructor and lifeguard training along the way, setting you up for the ‘real world’ of work when you return – but it can hardly be classed as work when it’s living the dream.
How could we not love Cape Town?
Comment with your number one reason why you’d go to Cape Town!
If this sounds like your ideal winter destination then check out our Cape Town Surf Camp and please don’t hesitate to get in touch.

Watch 13 Week Surf Adventure in Africa with Ticket to Ride

Watch TTR Meribel Mountain Lodge
Southern Africa’s summer has been wetter than normal: here’s why
Postdoctoral Fellow Climatology, University of the Free State
Disclosure statement
Sarah Roffe works for the Geography Department at the University of the Free State. She receives funding from the National Research Foundation of South Africa.
University of the Free State provides funding as a partner of The Conversation AFRICA.
View all partners

March marks the end of southern Africa’s 2021/2022 summer wet-season . Since its onset in October, most summer rainfall zone regions have experienced wetter than normal conditions.
Wetter conditions are positive for southern Africa’s rainfed agricultural activities and water reservoirs. But excessive rainfall has caused widespread flooding . This is in addition to flooding caused by tropical storm Ana , which made landfall in late January. And there is more to come: flooding will likely occur when tropical cyclone Batsirai – currently affecting the southern Indian Ocean – makes landfall.
These unusually wet conditions are the result of a weak La Niña event since December 2021 . This weather pattern typically brings above-average summer rainfall across southern Africa.
A La Niña event is one of three phases of a climate cycle called the El Niño-Southern Oscillation (ENSO) . The others are the El Niño and Neutral phases. These phases influence global atmospheric circulation and consequently global rainfall and temperature patterns. So the El Niño-Southern Oscillation is one of Earth’s most important climate phenomena.
What causes the La Niña event
The El Niño-Southern Oscillation is a naturally recurring phenomenon in which the ocean and the climate are related to each other. Every two to seven years, there are shifts in the temperature of the surface water of the tropical Pacific Ocean. There are also changes in the region’s east-to-west trade winds.
The Neutral phase, representing “normal” conditions, occurs almost half of the time. Sea surface temperatures are near-average and steady trade winds blow. The trade winds cause warm water to pile up in the western Pacific, while cool waters are drawn up from the bottom of the ocean to the eastern Pacific surface. Warm, moist air rises in the western Pacific, while in the eastern Pacific cool, dry air descends. This cycle of moving air is called the Walker Circulation . It pushes air over the equatorial Indian and Atlantic Oceans – which is one reason why the El Niño-Southern Oscillation makes an impact far away from the equatorial Pacific.
La Niña phases are basically enhanced Neutral phases. Trade winds blow harder, causing an expanded western Pacific warm pool and a more intense eastern and central Pacific cool pool. This causes a stronger sea surface temperature gradient which intensifies Walker Circulation.
El Niño phases are almost the direct opposite of La Niña phases. Trade winds weaken, or even reverse in direction, allowing warm waters to drift to the central and eastern Pacific. This eventually breaks down Walker Circulation.
The degree to which tropical Pacific Ocean waters deviate from average temperature determines the strength of La Niña or El Niño phases. That in turn determines their impact on global climate.
Influence on southern Africa’s weather
El Niño-Southern Oscillation phases have a regionally unique, although generally predictable influence on weather. For instance, while southern African summers typically have more frequent and heavier rainfall during La Niña events, southern USA regions usually experience the opposite conditions.
The current La Niña is the second event in two years. Impacts for this La Niña event are similar to last year, when southern African summer rainfall zone regions experienced a generally wetter and cooler than normal wet-season. This is the typical impact of a La Niña event. El Niño events typically have the opposite effects.
El Niño-Southern Oscillation phases influence the number of typical summer weather systems and where they form. That in turn influences rainfall and temperature patterns. Wetter and cooler conditions occur during La Niña phases because tropical temperate troughs – large rain-producing cloud bands – form more frequently and are located more eastward. Tropical lows – storms similar to very weak tropical cyclones – form more frequently and further south. Conversely, high-pressure systems – which promote hot, dry conditions – have reduced intensity and are located further south. The opposite occurs during El Niño phases.
Future outlook
El Niño-Southern Oscillation forecasts suggest the current weak La Niña should migrate to a Neutral phase during April-June . Southern Africa’s summer rainfall regions will likely continue experiencing wetter than normal conditions and additional flooding might occur.
Farmers, water managers and people generally should closely monitor daily weather forecasts and weather alerts issued by the meteorological bureau’s such as the South African Weather Service .
Looking beyond the current summer wet-season, it’s important to highlight the difference between climate variability and climate change. Southern Africa’s current above-average rainfall is a climate variability signal – a short-term fluctuation in average wet-season conditions. It will not necessarily cause an increase in the long-term wet-season rainfall average. Rather, the outlook under human-induced climate change suggests that southern Africa’s summer wet-seasons will likely become drier and shorter, but individual rainfall events may intensify .
Wet-season drought events may intensify and happen more often . These changes will have implications for long-term crop yield and surface water supply. Adaptation strategies need to account for this. Climate variability signals will, however, induce short-term implications, and to prepare for these seasonal forecasts need to be monitored.
Climate change will also likely have an impact on how El Niño-Southern Oscillation cycles behave and influence global climate. It’s not entirely clear exactly how the cycles will change. Some research suggests that El Niño and La Niña cycles may intensify, causing more intense impacts on global rainfall and temperature. El Niño-Southern Oscillation cycle changes are also expected to intensify current impacts on southern African summer wet-season periods.
- Agriculture
- Climate change
- Weather forecasting
- Extreme weather
- Tropical storms
- Indian Ocean
- Tropical cyclones
- Southern Africa
- El Niño Southern Oscillation
PhD Scholarship

Senior Lecturer, HRM or People Analytics

Senior Research Fellow - Neuromuscular Disorders and Gait Analysis

Centre Director, Transformative Media Technologies

Postdoctoral Research Fellowship

Afristay Blog
Top 4 reasons to visit South Africa during summer
South Africa, ever a tourist destination of note, provides one with a true African experience, with its melting pot of dynamic and diverse cultures and traditions to discover, uniquely individual cities and a landscape, wildlife and biosphere second to none.
And although it makes for pleasant and exciting travelling all year round, there’s no question that a South African summer is a glorious thing… Here are some of the top reasons this beautiful country during summertime:
1. Beach Beauties

South Africa has some of the best beaches in the world and although Cape Town and Durban in particular are famed for their beaches, the Eastern Cape and Garden Route regions have some glorious offerings too.

If you want to soak up the sun, try Durban’s South and North Beaches; Cape Town’s delightful Camps Bay, Clifton, Boulders and Llandudno beaches and Elandsbay in the West Coast; Port Elizabeth’s Hobie Beach; Jeffreys Bay’s Dolphin and Paradise Beaches; East London’s Gonubie Beach or the Garden Route’s Victoria Bay, Buffels Bay and Mossel Bay.
And of course, whether you find yourself encountering the Atlantic or Indian Ocean, sunny SA, with its oceans of contrast, is known for its surf and, although surfers brave the waters – even of the icy Atlantic, year-round – summer is the best time to catch a wave.

Surf at Durban’s Dairy beach, New Pier or Cave Rock at the Bluff; Cape Town’s Muizenberg, Kommetjie (Long Beach or the Kom) or if you’re brave enough, the Dungeons of Hout Bay (these produce some of the biggest waves on the African coastline) – or perhaps Jeffreys Bay (or J-Bay, as it’s fondly called) for some super tubing.
2.Game on… in the South African bushveld

Like many African countries, South Africa offers some of the world’s best game watching, with its wealth of long-standing and wonderfully well-loved game reserves, national parks and protected areas, dedicated to wildlife conservation, protection and appreciation.
Of course, everyone will tell you to head to the Kruger National Park – but please don’t forget about some of SA’s other wonderful game reserves.

KwaZulu Natal and the Eastern and Western Capes also have several top game reserves and national parks to visit.
South African National Parks (or SanParks) especially offers a wonderful selection of protected, yet public natural spaces, containing a vast number of wild animals, birds and flora to both educate and enchant you, as you discover more about South Africa.
3. Food, glorious food (and wine)

If you are looking for a food experience of note, then South Africa is your place. With so many different cultures and heritage roots, this country is blessed with the best in terms of local cuisine.
Try Durban for its Indian curries and famous bunny chows; Cape Town for its plethora of restaurants and eateries, serving international and local cuisines alike and for its lovely Cape Malay cooking – and of course, wherever you are in SA, you must enjoy a braai (like a barbeque, only better) with locals or tuck into some juicy shisa nyama (meat freshly prepared on an open street fire, often in township areas).
Other local delicacies include boerewors, samosas, rotis, Smileys and Walkie Talkies… see which best tantalises your taste buds and sets your stomach rumbling.
Of course, a trip to South Africa would not be complete without a visit to the Cape Winelands.
Well-known for its fine wine and beautiful wine estates, the Cape is blessed with the best. Wine-producing regions near or in Cape Town famed for their wine estates include: the Constantia Wine Valley, Stellenbosch, Franschhoek, Paarl and even Somerset West.
Further abroad, some two to three hours from Cape Town , small towns, found along South Africa’s iconic Route 62, like Ashton and Robertson are well-known for their wine, while if you head to Montagu, you can enjoy some special Muscadel (or Muscat), which is a red and white grape variety and is usually consumed as a sweet dessert wine.
While you’re visiting these wonderful estates, why not buy some take-home wine, explore a manor house and simply spend a day soaking up the stunning surroundings?
4.Natural beauty like no other
South Africa is a country blessed with some wonderful natural beauty and amazing attractions.
In the Cape, make a point of visiting the famous Table Mountain (a ride up in the cable car is a must but you can also hike up!), Oudtshoorn’s Cango Caves (a labyrinth of beautiful underground caves) and Cape Point, which is one of the most fiercely beautiful places you will encounter anywhere in the world.

In KZN, there’s no doubt that the uKhahlamba-Drakensberg Park and the Midlands (reminiscent of England’s green countryside) are everyone’s top picks. But Oribi Gorge (a spectacular gorge home to more than 300 bird species and one of the world’s biggest bungee swings), Zululand and the Valley of a Thousand Hills are all worth exploring too.
For excellent travel deals, see Drakensberg accommodation on Afristay.
Then, finally, Mpumalanga province is perhaps one of the most beautiful local regions, and is especially blessed with some awe-inspiring natural attractions. Find accommodation in Mpumalanga .
These include: Blyde River Canyon (the world’s largest green canyon), the Lake District (a grouping of majestic lakes, home to some 20,000 flamingos) and last but not least there’s God’s Window, where one is afforded a truly amazing, panoramic view out across the Lowveld, nearby mountains, rivers and the area’s many waterfalls too.
These are just some of the worthy reasons to visit South Africa during summertime although the favourable exchange rate and glorious, hot weather are always major draw cards too. May your travels to South Africa be safe, wonderful and memorable!
Content Created by Speckled Egg Digital Team.
Leave a Reply Cancel reply
Your email address will not be published. Required fields are marked *
You are here
Seasons in south africa.

South Africa ( officially the Republic of South Africa or abbreviated to RSA ) has always been one of the most beautiful places on the planet. This is a country where almost no end to the attractions you can see each person, as the variety of landscapes and activities here are limitless.
There are several climatic zones in the Republic of South Africa: deserts, locations with Mediterranean and even subtropical climate. Difference of temperature ranges and humidity levels between those zones is significant. In general seasons in the country are reverse comparing to North Hemisphere.

Summer weather lasts from October to March. At this time temperature is in quite wide between 15 and 35 °C (59-95 °F). Range of temperature at winter (June-August) is equally wide. At night temperature may drop below 0 °C (32 °F) and at noon rise to about 20 °C (68 °F) again depending on location of observation. Spring and autumn last for two months each season.

In South Africa there is no animal that with the onset of cold weather would fall into hibernation. Representatives of the local fauna generally behave the same at any time of the year. The fauna here is quite varied. Can be seen as predatory animals such as jackals, hyenas, leopards and lions and herbivorous elephants, antelopes, zebras and all kinds of monkeys.
Spring in South Africa

Spring weather comes to RSA with the very first days of September. Precipitation decreases and temperature rises. In coldest Cape Town air warms up to 18 °C (64 °F), in Durban to 20 °C (68 °F) and 26 °C (79 °F) in Pretoria. Both Atlantic and Indian oceans are too cold for swimming at this time. October is even warmer. Average temperatures gain several positive degrees and famous jacarandas of Pretoria start to bloom. At November average temperature gain couple more degrees while amount of precipitation decreases. It is wonderful time to travel around the Republic of South Africa.
Amount of precipitation distributed unevenly across territory of the country. In north-western regions there are about 200 mm (7.8”) of annual precipitation. Western regions receive 900 mm (35.4”) and central 400 mm (15.7”).
Climate of the Spring Months

| Max average t°: +26 °C (79 °F) Min average t°: +9 °C (48 °F) Sundial in the day: 11.5 hours Rainy days: 0~1 day Precipitation: 8 mm (0,3") |
| Max average t°: +28 °C (83 °F) Min average t°: +13 °C (55 °F) Sundial in the day: 10 hours Rainy days: 1 day Precipitation: 15 mm (0,6") |
| |
( Pretoria )
Spring Holidays

Here is a list of festivals that are actively celebrated in South Africa:
September, 24 — heritage day.
October, 10 — day Kruger;
Summer in South Africa

At December hot tropical summer comes to the Republic of South Africa. Average daytime temperature is in the range between 26 °C (79 °F) in Cape Town and 28 °C (82 °F) in Pretoria and Durban. The west of the country becomes rainy but showers occur after noon with sunny half of a day before.
Rainy Season (December to February)
Main beach life is concentrated on coasts of Indian Ocean in Province of Natal. Swimming here is available year-round because temperature of water is 24 °C (75 °F) at summer and 20 °C (68 °F) at winter. Those who don’t like winds weather shouldn’t go to Cape Town at summer. “Cape-doctor” wind is usefull but slightly bothering.
Climate of the Summer Months

| Max average t°: +29 °C (84 °F) Min average t°: +15 °C (60 °F) Sundial in the day: 7 hours Rainy days: 2-3 days Precipitation: 37 mm (1,5") |
| Max average t°: +29 °C (84 °F) Min average t°: +16 °C (61 °F) Sundial in the day: 7 hours Rainy days: 2-3 days Precipitation: 37 mm (1,5") |
| Max average t°: +28 °C (83 °F) Min average t°: +16 °C (60 °F) Sundial in the day: 8 hours Rainy days: 2 days Precipitation: 33 mm (1,3") |
Summer Holidays

At this time of year accounts for most of the holidays of the countries of South Africa, including:
December, 16 — day of reconciliation; December, 25 — Christmas day; December, 26 — day of goodwill;
January, 1 — New year.
Autumn in South Africa

March is the last hot month of a year like some threshold of autumn. Average daytime temperature is about 25 to 28 °C (77-82 °F). It is still warm enough for swimming at March. At April water cools down but instead hunting season begins especially on baboons (to control population and protect antelopes). Also at April temperature drops by couple of degrees and occasional fogs are occurring.
At May climatic autumn comes to RSA. This month bring showers, heavy clouds and yellow on branches of trees. It is time to harvest corn, cotton and sugarcane. Temperature in Cape Town barely reaches 19 °C (66 °F) while Johannesburg and Pretoria are warmer with 22 °C (72 °F).
Climate of the Autumn Months

| Max average t°: +27 °C (81 °F) Min average t°: +14 °C (57 °F) Sundial in the day: 9 hours Rainy days: 1-2 days Precipitation: 21 mm (0,8") |
| Max average t°: +25 °C (77 °F) Min average t°: 10,5 °C (51 °F) Sundial in the day: 11 hours Rainy days: 1-2 days Precipitation: 10 mm (0,4") |
| Max average t°: +22,5 °C (73 °F) Min average t°: +6 °C (42 °F) Sundial in the day: 12.5 hours Rainy days: 1-2 days Precipitation: 20 mm (0,8") |
Autumn Holidays

Before you top South African festivals, which are celebrated in the autumn:
March, 21 — the day of protection of consumer rights; March, 29 — Holy Friday; March, 31 — Easter.
April, 1 — family day; April, 6 — founder's day; April, 27 — independence day;
May, 1 — labour day; May, 24 — Empire day; May, 31 — Republic day.
Winter in South Africa

June, July, August in RSA is the time of winter in the Southern Hemisphere. Weather is quite unpredictable and strongly depends on location. For instance in Cape Town average temperature is about 17 °C (63 °F) while in Durban is around 21 °C (72 °F) while they are almost on the same latitude. East coasts of RSA are warmer than west by average of 6 °C in Celsius (≈11 °F on the Fahrenheit scale ) due to influence of warm Agulhas current and cold Benguela current respectively. Very rarely in various parts of South Africa you can see a small short-term snowfall. This is usually a slightly cool and dry season..
Dry Season (June to August)
Visit to the country at the middle of summer is unique opportunity to see whales. At this time they are approaching shore close enough for spectating with bare eyes. Unique experience attracts lots of tourists. August is the last month of winter and it is good to visit the blooming Kalahari Desert.
Climate of the Winter Months

| Max average t°: +20 °C (68 °F) Min average t°: +2 °C (36 °F) Sundial in the day: 14 hours Rainy days: 0~1 day Precipitation: 7 mm (0,3") |
| Max average t°: +20 °C (68 °F) Min average t°: +2 °C (36 °F) Sundial in the day: 14 hours Rainy days: 0 day Precipitation: 3 mm (0,1") |
| Max average t°: +23 °C (73 °F) Min average t°: +5 °C (40 °F) Sundial in the day: 13 hours Rainy days: 0 day Precipitation: 5 mm (0,2") |
Winter Holidays

Before you top South African holidays celebrated in winter:
June, 16 — youth day; June, 26 — day of struggle for freedom in South Africa;
July, 10 — family day. August, 9 — national women's day.
Our project

Stay connected

Seasons of the Year
- About the Project
- Terms of Use
- Copyright & Permissions
- Live on Sky
- Get Sky Sports
- Sky Mobile Apps
- Kick It Out
- Black Lives Matter
- British South Asians in Football
Men's T20 World Cup 2024: Full results after India beat South Africa in Barbados final to win second title
India beat South Africa by seven runs in Barbados to win Men's T20 World Cup for a second time; Proteas denied a first World Cup title after heart-breaking defeat at Kensington Oval - watch England men and women matches and more live on Sky this summer, stream with NOW
Saturday 29 June 2024 20:33, UK

Full results from the 2024 Men's T20 World Cup after India beat South Africa in Barbados to win their second title in the tournament.
T20 World Cup - Knockout stage results
Saturday june 29.
Final - India beat South Africa by seven runs - Bridgetown, Barbados
Thursday June 27
Semi-final #1 - South Africa beat Afghanistan by nine wickets - Tarouba, Trinidad and Tobago
Semi-final #2 - India beat England by 68 runs , Providence, Guyana
T20 World Cup - Super 8s results
- Group 1 - India, Australia, Afghanistan, Bangladesh
- Group 2 - USA, West Indies, South Africa, England
Wednesday June 19
South Africa beat USA by 18 runs - North Sound, Antigua
- England through to QFs - reaction and your thoughts LIVE!
- They've done it again! Georgia shock Spain with opener LIVE!
- World Cup of Darts LIVE! England take on Austria in final
- Transfer Centre LIVE! Premier League clubs face PSR deadline today!
- England player ratings: Bellingham the poster boy of Euro 2024 redemption
- England's route: Three Lions face Switzerland in QF on Saturday at 5pm
- Norris: I might lose respect for 'stupid and reckless' Verstappen
- Verstappen-Norris collision hands Russell dramatic Austrian GP win
- Liverpool's move for Newcastle winger Gordon breaks down
- Neville: Woeful England must make dramatic changes now
- Latest News
Thursday June 20
England beat West Indies by eight wickets - Gros Islet, Saint Lucia
India beat Afghanistan by 47 runs - Bridgetown, Barbados
Get Sky Sports on WhatsApp
Stream Sky Sports with NOW
Cricket news, analysis and features
Latest cricket videos and highlights
Friday June 21
Australia beat Bangladesh by 28 runs (DLS) - North Sound, Antigua
South Africa beat England by seven runs - Gros Islet, Saint Lucia
Saturday June 22
West Indies beat USA by nine wickets - Bridgetown, Barbados
India beat Bangladesh by 50 runs - North Sound, Antigua
Sunday June 23
Afghanistan beat Australia by 21 runs - Kingstown, Saint Vincent and the Grenadines
England beat USA by 10 wickets - Bridgetown, Barbados
Monday June 24
South Africa beat West Indies by three wickets (DLS) - North Sound, Antigua
India beat Australia by 24 runs - Gros Islet, Saint Lucia
Tuesday June 25
Afghanistan beat Bangladesh by eight runs (DLS) - Kingstown, Saint Vincent and the Grenadines
T20 World Cup - first-round results
Sunday june 2.
USA beat Canada by seven wickets - Grand Prairie, Texas
West Indies beat Papua New Guinea by five wickets - Providence, Guyana
Monday June 3
Namibia beat Oman in Super Over - Bridgetown, Barbados
South Africa beat Sri Lanka by six wickets - Long Island, New York
Tuesday June 4
Afghanistan beat Uganda by 125 runs - Providence, Guyana
England vs Scotland: Match abandoned - Bridgetown, Barbados
Netherlands beat Nepal by six wickets - Grand Prairie, Texas
Wednesday June 5
India beat Ireland by eight wickets - Long Island, New York
Thursday June 6
Uganda beat Papua New Guinea by three wickets - Providence, Guyana
Australia beat Oman by 39 runs - Bridgetown, Barbados
USA beat Pakistan in Super Over - Grand Prairie, Texas
Scotland beat Namibia by five wickets - Bridgetown, Barbados
Friday June 7
Canada beat Ireland by 12 runs - Long Island, New York
Saturday June 8
Afghanistan beat New Zealand by 84 runs - Providence, Guyana
Bangladesh beat Sri Lanka by two wickets - Grand Prairie, Texas
South Africa beat Netherlands by four wickets - Long Island, New York
Australia beat England by 36 runs - Bridgetown, Barbados
Sunday June 9
West Indies beat Uganda by 134 runs - Providence, Guyana
India beat Pakistan by six runs - Long Island, New York
Scotland beat Oman by seven wickets - North Sound, Antigua
Monday June 10
South Africa beat Bangladesh by four runs - Long Island, New York
Tuesday June 11
Pakistan beat Canada by seven wickets - Long Island, New York
Wednesday June 12
Sri Lanka vs Nepal: Match abandoned - Lauderhill, Florida
Australia beat Namibia by nine wickets - North Sound, Antigua
India beat USA by seven wickets - Long Island, New York
Thursday June 13
West Indies beat New Zealand by 13 runs - Tarouba, Trinidad and Tobago
Bangladesh beat Netherlands by 25 runs - Kingstown, Saint Vincent and the Grenadines
England beat Oman by eight wickets - North Sound, Antigua
Friday June 14
Afghanistan beat Papua New Guinea by seven wickets - Tarouba, Trinidad and Tobago
USA vs Ireland: Match abandoned - Lauderhill, Florida
Saturday June 15
South Africa beat Nepal by one run - Kingstown, Saint Vincent and the Grenadines
New Zealand beat Uganda by nine wickets - Tarouba, Trinidad and Tobago
India vs Canada: Match abandoned - Lauderhill, Florida
England beat Namibia by 41 runs (DLS) - North Sound, Antigua
Sunday June 16
Australia beat Scotland by five wickets - Gros Islet, Saint Lucia
Pakistan beat Ireland by three wickets - Lauderhill, Florida
Monday June 17
Bangladesh beat Nepal by 21 runs - Kingstown, Saint Vincent and the Grenadines
Sri Lanka beat Netherlands by 83 runs - Gros Islet, Saint Lucia
New Zealand beat Papua New Guinea by seven wickets - Tarouba, Trinidad and Tobago
Tuesday June 18
West Indies beat Afghanistan by 104 runs - Gros Islet, Saint Lucia
Please use Chrome browser for a more accessible video player

How did teams qualify?
West Indies and USA earned automatic entry by being hosts, with the latter to now play in the tournament for the first time, while England, Pakistan, New Zealand, India, Australia, South Africa, Netherlands and Sri Lanka secured their places by finishing in the top eight of the previous T20 World Cup in 2022.
Afghanistan and Bangladesh were then included after being the next two highest teams in the T20I rankings, with every other spot determined by regional qualifiers.
Ireland and Scotland came through the European section and Papua New Guinea emerged from the East Asia-Pacific Qualifier.
Canada - who will also make their tournament debut - progressed through the Americas Qualifier, while Nepal and Oman are the Asian qualifiers, with Namibia and Uganda coming through the African Qualifier to complete the line-up.
Watch the 2024 ICC Men's T20 World Cup this June live on Sky Sports or stream the best sport with NOW .

Stream the T20 World Cup, the World Cup of Darts and more

- Upgrade Now

- Compounds and hostels: history and transformation
- The significance of vernacular architecture
- Into the future: the Kruger National park as a region
- Mastery of societal fissures as a way to approach economic opportunity
- Sisters of wilderness
- The Okavango Delta:paradise in peril
- A Guided tour of D-School: Africa's state-of-the-art building
- Body Donation why and how?
- Campus Walking Tours
- Indigenous tree identification in Newlands Forest
- Map my way curated heritage route
- The early UCT Grootte Schuur Rondebosch campus: architects and architecture
- AI unveiled: Understanding artificial intelligence and its impact on our world
- Will robots take my job
- Cyper space and human rights
- Know your rights under the Consumer Protection Act and the National Credit
- Pension funds in South Africa: learnings from tales of greed, folly and virtue
- A Wander through the wonder of some fascinating English words
- Art in motion
- Books that changed my life
- Five favourites: Poetry in Person
- From the Cape to Cairo
- I know what I Iike
- Iintrospection and documentation: an overview of dominant strands in contemporary South African photography
- Indian classical music: reverie and understanding
- Landscapes in art: creating heaven on earth
- Mathematics in the plays of Tom Stoppard
- Moulded by the hands of god
- Mozart: myths, mysteries and music
- Notes and curiosities from Florence
- Painting and the eye of the beholder
- Rising stars in concert: Opera UCT
- Robert and Clara Schumann: very private lives
- Schizophrenia: Journey and care
- Sir Abe Baily: a biography
- The first true romantic: exploring the troubled musical genius of Robert Schumann
- The medical road less travelled
- The people's bible: fresco cycles and the golden legend
- The unprecedented media cult of Daisy de Melker
- A tale of two disorders: ADHD and ASD
- Anaesthesia: more than just a deep sleep(part 1)
- Anaesthesia: more than just a deep sleep(part 2)
- Body donation: why and how
- Breaking the mould: the real story of penicilin
- Child Abuse: a socio-historical perspective
- Molecular oncology and personalised cancer care
- Painful Osteoarthritis: treating pain that is not all about the joint
- 100 years of Radio: from a woman's perspective
- Africa's Nobel Laureates: have we received enough credit?
- Antisemitism in South Africa: a history
- Are you living up to your blue china?
- Boneshakers and b: the colourful history of the bicycle
- Bringing the looters to justice: the case for an international anti-corruption court
- Cartels and Collusion: exposing South Africa's organised crime reality
- Cuba and Africa 1959 -1994: writing an alternative Atlantic history
- Eskom: power, politics, and the (post)apartheid state
- Fantastic fishes
- From Metternich to Merkel: the search for German identity
- Generals Botha and Smuts and the First World War
- How South Africa got out of 80s growth slump: can it o so again?
- Indefinite life extension: is it a good idea?
- Is it still safe to drink tapwater and swim in our waterways
- Lightbulb moments
- Mining the complex legacy of Harry Oppenheimer
- NHI: PANEL DISCUSSION ON THE NATIONAL HEALTH INSURANCE ACT
- Operation Vula: the secret communications network that helped end apartheid.
- Robert Grendon's dream: Remarkable black intellectual, poet, cricketer
- Russian invasion of Ukraine: Ukrainian diplomacy, new era and opportunities for Ukraine and the global south
- South Africa beyond 2024: coalition politics and democratic stability amidst political fragmentation
- The impact of Charles Darwin's visit to the Cape of Good Hope in 1836 on his theory of evolution and natural selection
- The intelligence war in South Africa: 1939 -1948
- The world and South Africa beyond 2024: the lates flags, scenarios, and probabilities
- War on Cuba: Belly of the beast
- What does compassion, or Ubuntu, mean in Contemporary South Africa?
- Who was Napoleon 11?
- Women and Culture in the Renaissance
- German for beginners
- Intermediate Italian
- Irma Stern Museum : creative art workshop
- Isixhosa Communication Skills For Beginners
- Italian For Beginners
- Nature writing
- Spanish for beginners
- Story development
- Through music into image
- To control or lose control: drawing with ink
- Turkish for beginners
- Write Your Short Story, from concept to completion
- Asteroid collisions: planetary curse or blessing
- Cederberg rock paintings as a social archive
- Fire and ice: the earth's special place
- Interesting physical phenomena
- Sharks: the perfect predators
- Sustainable seafood production through integrated aquaculture
- The cell: a biologist's peep into an awesome micro-universe of riveting beauty and of life's origins
- Short Courses
- Friends of Summer School
- Nicole Loser
- Nkateko Masinga
- Patricia Mcleod
- Carin van den Heever
- Denise Zinn
- Fiona Hulme Brophy
- Lebogang Montewa
- Simthandile Mhlambiso
- Suzanne Gardener
- Aadhiya Tulsi
- Kat Volkwyn
- Madeline Gray Glass
- Heidi Maritz
- Olivia Wright
- Trish van der Nest
- News archive
- EMS events calendar
- EMS Outreach
- Guiding Lights: Great Writers From Ancient Greece To Seventeenth Century Japan
- In Conversation
- Even more excellent, little-known books
- Books As Magic Carpets: Why, How And What We Read
- Madness in Greek tragedy: a study of three plays
- Art as a mirror
- Bird and fish: their enduring symbolism
- The Gothic Cathedral
- The great divorce: abstraction and expressionism in the 20th century
- Christo vs Warhol: brilliant art and eccentric personalities
- Dialogues Across Time And Space: A Noisy History Of African Art
- Enchanting the eye and the ear
- Vive la France! A festival of French music through the ages
- Tipping points: the evolution of music
- Film music: the new classical?
- IN CONVERSATION: CHILDREN OF SUGARCANE
- In conversation: the boy who never gave up
- A MORNING WITH IRMA STERN
- Music across time
- Jazz on the lawn
- I Came From The Wilderness
- The Long View: Getting Behind The Horror Of South Africa’s Headlines
- The Enlightenment
- Exploring Eastern Thought: Religions, Philosophies And Cultures Of The East
- A ‘history of the Future’ In the 21st Century: Yuval Noah Harari
- Escape From Pretoria
- The Russian War In Ukraine: The Origin, Narratives And Potential Impacts On The African Continent
- 882 Ad: The Origins Of Putin’s Ukrainian War
- The World And South Africa Beyond 2023: The Latest Flags, Scenarios And Probabilities
- The ANC Below 50%: What Is The Road Ahead?
- Anatomy Of State Capture
- South Africa’s Black Middle Class: Past, Present And Future
- Philosophical Lives
- A Voyage Of Discovery: The Trans-siberian Railway
- Napoleon, The Rosetta Stone, And The Deciphering Of Ancient Egypt
- OCEANIC HISTORIES
- FAMILY SILVER: BURIED SEPTEMBER 1939; RETRIEVED OCTOBER 2019
- Human Sexuality: Understanding The New Paradigm
- Neuroscience Then And Now
- Science Marches On: Fascinating Developments
- Understanding And Managing Dementia
- Functional Medicine And Nutrition Therapy
- No Pain, No Brain
- Encounters With Table Mountain And The History Of Geology
- Water For Cape Town: 370 Years Of ‘not Quite Enough’
- In The Aftermath Of Extinctions
- Astronomy Highlights With The Meerkat Radio Telescope
- Seeing Black Holes With An Earth-sized Telescope
- Inside Schrodinger’s Cat
- Let’s go tracking in the Jurassic: fossil footprints of Southern Africa
- Where death delights helping the living
- Cape Town’s Rivers And Wetlands: Field Trip
- Laetoli footprints
- EXTRAORDINARY GARDENS FROM AROUND THE WORLD
- Extraordinary Gardens From Around The World
- The Evolution Of A Great Garden: Kirstenbosch 1913 To 2023
- Kirstenbosch National Botanical Garden: A Journey Through 110 Years
- Cloud Technologies that are changing our world today
- ‘life Is Not Still’: Let’s Bring Still-life Back To Life
- The Joy Of Drawing: A Course For Beginners
- Introduction To Botanical Painting
- Turn Your Writing Dreams Into Reality
- How To Write A Novel
LET’S WRITE A MEMOIR ESSAY

Sally Cranswick is a writer, editor, ghost writer, story coach and workshop facilitator with a special interest in life writing and memoir.
Sally Cranswick
Saturdays from 10:00–12:00
Dates: 13 April– 10 May
Online platform: The course will be offered on Zoom. Upon registration the link will be provided.
Course fee: R2 000
Booking is through Webtickets: https://www.webtickets.co.za/event.aspx?itemid=1542956820
In a genre of its own, the memoir essay is a wonderful form which allows the writer to explore the beauty of an encapsulated memory or feeling without committing to a whole novel.
During this workshop series we will look at examples of wonderful memoir essays from different genres and we’ll discuss ways to shape our own narratives around the essay form, using structure, voice, style and theme, how to write about people, memories and truth, in a way that heals not harms, and how to extend one essay into a collection of work.
Each workshop will include formal presentations, group discussions, writing exercises and plenty of opportunity to ask questions. There will be weekly homework (for those who want it) and writers may submit a final piece of work to Sally for feedback.
The aim of this course is to write a memoir essay and to have fun as a creative community whilst writing towards our highest goals.
Inspiration, ideas and thinking about how a memoir essay can work for the story you want to tell.
Discovering your unique voice and style and working with timelines, setting and era.
Structuring your story around the essay form and how to create a collection of work that forms one narrative.
How to honour other people in your story whilst telling the truth.
What to do with your finished essay. Editing, markets, publishing ideas, and the chance to share your story ‘out loud’.
| Places are limited to |
|
|
| For further information, please email , or phone 021 650 2634 (office) or Medeé at 083 707 6420. |

8 Steps to Reduce Anxiety Over Summer Break
Evidence-based strategies to reduce anxiety for college students..
Posted June 27, 2024 | Reviewed by Tyler Woods
- What Is Anxiety?
- Find counselling to overcome anxiety
- Summer break is an opportunity to reduce anxiety.
- Work on getting physically and emotionally fit.
- Plan for the future while also attending to interests, work, and a social life.
Summer break never comes soon enough and never lasts long enough. It’s a much-anticipated time for college students, offering a chance to relax and step away from academic pressures. However, for many, the transition from a busy semester to a relatively unstructured break can bring its own set of anxieties. Whether it’s concerns about internships, future plans, or simply managing downtime effectively, summer break can sometimes be more stressful than expected. This blog post explores evidence-based strategies for college students to reduce anxiety and make the most of their summer break.
1. Nail Down a Routine: While it’s tempting to sleep in and take each day as it comes, maintaining a loose routine can provide a sense of stability. It also helps your brain have some amount of predictability (which reduces anxiety). Plan your day with a balance of activities, including exercise, hobbies, and social time. Also, set small, achievable goals for the summer. These can range from reading a certain number of books to learning a new skill. Goals provide a sense of purpose and accomplishment. I think physical fitness, reading, or even cooking goals lend themselves to measurable outcomes that can be easily tracked and very satisfying.
2. Get Fit: Physical activity is the number one evidence-based way to reduce anxiety. Aim for at least 30 minutes of exercise a day, whether it’s a morning run, yoga session, or playing a sport with friends. This also helps with sleep. Pay attention to your diet . Eating a diet full of fruits, vegetables, and whole grains can improve mood and energy levels. Avoid excessive caffeine (don’t consume after 11:00 am) and sugar, which can exacerbate anxiety (and actually make you feel more tired).
3. Learn How to Chill Out: Mindfulness and meditation practices can significantly reduce anxiety with few side effects. Several apps offer guided meditations that are perfect for beginners. Simple deep breathing exercises can help manage stress in the moment. Practice techniques like the 4-7-8 breathing method or box breathing to calm your mind and body.
4. Stay Connected: Maintain social connections with friends and family. Regular social interaction can provide emotional support and reduce feelings of isolation. No, it doesn’t count if you’re on Snapchat or Instagram —they are actually designed to increase your anxiety. It’s also not the same as being physically around people and interacting face-to-face. Maybe it’s a bit corny, but consider joining a summer group or club that aligns with your interests. This can be a great way to meet new people and stay engaged. I love the idea of college students joining a running club or performance group. They're challenging, social, and get you out of the house.
5. Practice Playing Again: Use the summer break to lean into hobbies you’ve pushed off during the semester. Whether it’s painting, playing an instrument, or gardening, engaging in activities you love can be therapeutic while also helping you to practice using your time without a device. Try challenging yourself to learn something new. This could be a new language, cooking, or a craft. Learning new skills can boost confidence and provide a sense of accomplishment.
6. Prep for the Future: If you’re anxious about your career , use the summer to gain experience. Internships, part-time jobs, or volunteer work can be valuable for your resume and give you a sense of direction. The days are long, so there’s plenty of time to really load up your day with a job, volunteering, fitness, and time with friends. It’s also a great time to reflect on your career goals and plan steps to achieve them. Research potential career paths, network with professionals, and consider your next steps. I especially love the idea of using summer break for informational interviews with family friends who can provide insight into job hunting and preparing for the career landscape.
7. Get Help: When anxiety becomes overwhelming, don’t hesitate to seek professional help. Summer is the time to get comfortable with a therapist. Many colleges offer counseling services during the summer, and there are also numerous online therapy options available. A great way to learn more about anxiety and how to mitigate it is by joining a support group for anxiety. Sharing experiences with other college students facing similar challenges can provide comfort and practical coping strategies.
8. Get Outside: Nature has a calming effect on the mind. Just seeing the color green reduces peoples anxiety and depression . Spend time hiking, biking, or simply walking in a park. If possible, plan a weekend getaway to a park, beach, or lake. Camping, beach trips, or mountain retreats can offer a refreshing break from daily routines and activate other senses and parts of the brain you may not use as often.

Summer break is a valuable opportunity for college students to relax, recharge, and reduce anxiety. By establishing a routine, focusing on physical and mental health, staying connected, engaging in hobbies, planning for the future, seeking professional help if needed, and enjoying nature, students can make the most of their summer break. It’s not about balance but about attending to each of these areas to reduce anxiety and increase satisfaction. Remember, the goal is to find an authentic structure that works for you and allows you to return to the next semester refreshed and ready to tackle new challenges.
To find a therapist, visit the Psychology Today Therapy Directory .
Liu Kexiu, Mohamed Elsadek, Binyi Liu, et al (2021). Foliage colors improve relaxation and emotional status of university students from different countries. Heliyon. Cell Press.
Elizabeth A. Hoge, Eric Bui, Luana Marques, et al. (2013). Randomized Controlled Trial of Mindfulness Meditation for Generalized Anxiety Disorder: Effects on Anxiety and Stress Reactivity. Journal of Clinical Psychiatry.
Elizabeth Aylett, Nicola Small, and Peter Bower. (2018). Exercise in the treatment of clinical anxiety in general practice – a systematic review and meta-analysis. BMC Health Services Research.
Fengxia Lai, Lihong Wang, and Jiyin Zhang, et al. (2021). Relationship between Social Media Use and Social Anxiety in College Students: Mediation Effect of Communication Capacity. International Journal of Environmental Research and Public Health.

Rob Danzman is a licensed professional counselor and mental health counselor and founder of Motivate Counseling in Bloomington, Indiana.
- Find Counselling
- Find Online Therapy
- South Africa
- Johannesburg
- Port Elizabeth
- Bloemfontein
- Vereeniging
- East London
- Pietermaritzburg
- Asperger's
- Bipolar Disorder
- Chronic Pain
- Eating Disorders
- Passive Aggression
- Personality
- Goal Setting
- Positive Psychology
- Stopping Smoking
- Low Sexual Desire
- Relationships
- Child Development
- Self Tests NEW
- Therapy Center
- Diagnosis Dictionary
- Types of Therapy

At any moment, someone’s aggravating behavior or our own bad luck can set us off on an emotional spiral that could derail our entire day. Here’s how we can face triggers with less reactivity and get on with our lives.
- Emotional Intelligence
- Gaslighting
- Affective Forecasting
- Neuroscience
- Open access
- Published: 24 June 2024
A metagenomic investigation of the faecal RNA virome structure of asymptomatic chickens obtained from a commercial farm in Durban, KwaZulu-Natal province, South Africa
- Vivian C. Nwokorogu 1 ,
- Santhosh Pillai 1 ,
- James E. San 3 ,
- Charlene Pillay 1 ,
- Martin M. Nyaga 2 &
- Saheed Sabiu 1
BMC Genomics volume 25 , Article number: 629 ( 2024 ) Cite this article
251 Accesses
Metrics details
Virome studies on birds, including chickens are relatively scarce, particularly from the African continent. Despite the continuous evolution of RNA viruses and severe losses recorded in poultry from seasonal viral outbreaks, the information on RNA virome composition is even scantier as a result of their highly unstable nature, genetic diversity, and difficulties associated with characterization. Also, information on factors that may modulate the occurrence of some viruses in birds is limited, particularly for domesticated birds. Viral metagenomics through advancements in sequencing technologies, has enabled the characterization of the entire virome of diverse host species using various samples.
The complex RNA viral constituents present in 27 faecal samples of asymptomatic chickens from a South African farm collected at 3-time points from two independent seasons were determined, and the impact of the chicken’s age and collection season on viral abundance and diversity was further investigated. The study utilized the non-invasive faecal sampling method, mRNA viral targeted enrichment steps, a whole transcriptome amplification strategy, Illumina sequencing, and bioinformatics tools.
The results obtained revealed a total of 48 viral species spanning across 11 orders, 15 families and 21 genera. Viral RNA families such as Coronaviridae, Picornaviridae, Reoviridae, Astroviridae, Caliciviridae, Picorbirnaviridae and Retroviridae were abundant, among which picornaviruses, demonstrated a 100% prevalence across the three age groups (2, 4 and 7 weeks) and two seasons (summer and winter) of the 27 faecal samples investigated. A further probe into the extent of variation between the different chicken groups investigated indicated that viral diversity and abundance were significantly influenced by age ( P = 0.01099) and season ( P = 0.00099) between chicken groups, while there was no effect on viral shedding within samples in a group (alpha diversity) for age ( P = 0.146) and season ( P = 0.242).
The presence of an exceedingly varied chicken RNA virome, encompassing avian, mammalian, fungal, and dietary-associated viruses, underscores the complexities inherent in comprehending the causation, dynamics, and interspecies transmission of RNA viruses within the investigated chicken population. Hence, chickens, even in the absence of discernible symptoms, can harbour viruses that may exhibit opportunistic, commensal, or pathogenic characteristics.
Peer Review reports
Introduction
Globally, chicken’s productive performance and feed conversion rate are greatly influenced and dependent on the state of health and proper functioning of its gastrointestinal tract (GIT), which is the site of nutrition, metabolism, and build-up of diverse microorganism [ 1 ]. The chicken GIT is often colonized by microorganisms like bacteria, fungi, and viruses which could be harmless, symbiotic, or pathogenic. Therefore, poor GIT health, even in the absence of a recognized disease state, can affect poultry performance, resulting in low productivity [ 2 ]. Decades of avian research, particularly microbiome studies have focused on characterizing bacteria, while utilizing the 16S rRNA gene sequencing as a powerful tool to investigate the dynamics, biological and ecological roles of the GIT microbiota in chicken. Unlike bacteria, viruses are difficult to sequence and characterize because of their lack of markers or conserved regions that can be employed for taxonomic identification, high genetic variability, and short genome lengths [ 3 ]. Although immense studies have availed valuable information about the gut microbiome, comprehensive analysis on the gut virome of chicken is still limited. Importantly, in the light of the frequently dense nature of chicken flocks and their possession of homogeneous gene traits, often result in their increased vulnerability to breakouts of viral infections. Hence, even on high precaution poultry farms, a wide range of viruses can accumulate, especially if the poultry birds are of different age groups. This is because the infections are often asymptomatic and therefore undetected allowing them to spread quickly, hence causing substantial economic losses [ 4 ]. In South Africa, nearly all bird flu viral outbreaks in poultry have been recorded in chickens and have cost the nation millions of rands, despite chicken being the most consumed source of animal protein. Notably, about 145 avian-flu outbreaks were recorded from different South African poultry farms between April 2021 and September 2023 with more than 4 million birds culled [ 5 , 6 ]. Hence, it has remained crucial to critically examine the dynamics of viral pathogens that maybe implicated in underperformance, low productivity, and mortality in poultry production, particularly chickens.
Worldwide, next-generation sequencing (NGS), a high throughput sequencing method has allowed unprecedented advances in the characterization of complex microbial communities including viruses [ 1 , 7 , 8 ]. The NGS approach offers the combined advantages of speed, sensitivity, automation, and high-throughput deep sequencing and has successfully been used to characterize faecal microbiota of avian species including both wild [ 9 , 10 ] and domestic birds [ 11 , 12 ]. In addition, the revolutionization of viral metagenomics concerning epidemiological studies has allowed credible, faster, better detection and surveillance of multifunctional viruses in poultry. Majority of recent avian viral research have focused on zoonotic viral pathogens, or viruses causing significant economic losses in poultry while overlooking other viruses which also constitute the avian virome [ 10 , 13 ]. Continuous viral surveillance aimed at characterizing viruses in chicken is necessary to enhance knowledge of key viral agents associated with poultry related infection. While DNA viruses such as adenoviruses [ 14 , 15 ] and parvoviruses [ 16 , 17 ] identified in chickens have been associated with enteritis, studies have shown that RNA viruses constitute a greater proportion of all infections caused by viruses [ 18 , 19 ]. This has been attributed to the highly unstable nature of RNA viruses, and rapid mutation rates due to their error-prone replication mechanisms often leading to diverse variants and multi-species [ 20 ]. As a result of these attributed factors and their remarkable capacity to transcend species boundaries, many viruses possessing RNA genomes have emerged as noteworthy pathogens with the potential to cause widespread epidemics or even pandemics. Some notable examples of RNA viral outbreaks associated with animal origin, include severe acute respiratory syndrome coronavirus 2 (SARS-CoV 2) [ 21 ], Ebola [ 22 ], Swine flu [ 23 ], and Middle East respiratory syndrome [ 24 ]. Unfortunately, virome studies on birds including chickens are relatively scarce, particularly from the African continent, and the information on their RNA virome composition is even scantier despite the continuous evolution of RNA viruses and their associated disease outbreaks. Considering the high instability of RNA viruses, their high adaptive mechanism and quasi species emergence for instance SARS-CoV 2, raise concerns as undetected and unidentified RNA viruses may become the basis for a potential outbreak in the nearest future [ 3 ]. This study was underscored as a crucial step toward future epidemiological investigations of chicken’s faecal RNA viruses in South Africa. Importantly, the focus of the study was not on any specific pathogen (s) or disease condition(s) but rather on the entire RNA virome, hence no diseased group/chickens with defined disease condition was included. Therefore, this pilot study looked to obtain baseline data and unveil the dynamics of the faecal RNA virome of asymptomatic South African chickens.
In this study, for the first time, we conducted a comprehensive analysis to elucidate the RNA virome in the GIT of asymptomatic chickens from Durban, KwaZulu-Natal province in South Africa. Through optimized enrichment strategies, non-invasive faecal sampling methods, and bioinformatics tools, we explored the RNA virome structure of apparently healthy chickens, tracking viruses across different stages and seasons. This study provided insights into the genomes of faecal RNA viruses, their diversity, structure, and colonization in healthy chickens’ gut, while also contributing to the understanding of potential disease agents and those with potential to cross species barriers.
Materials and methods
Sampling design and collection.
The faecal samples were obtained from a commercial poultry farm in Durban, KwaZulu-Natal, South Africa. A total of 10 chickens were used in all, five for summer and five for winter periods. Samples were collected at three different time points, namely the early (2 weeks), intermediate (4 weeks), and mature (7 weeks) developmental stages of chickens (Fig. S1 ). Five asymptomatic chickens were randomly pre-selected from flocks at 2 weeks of development during the summer period, July/August 2021. At the first collection time point, the selected chickens ( n = 5) at 2 weeks were marked and kept separately from the flock in medium-sized metal cages, layered with sterile plastic wrap and sawdust. The faecal contents from each cage were obtained from individual chicken immediately as they dropped using sterile plastic bags and the chicken returned to its flock. At 4 and 7 weeks of age, the same sample collection procedure was repeated for the marked chickens (same summer chickens). During winter (December 2021 to January 2022), another five asymptomatic chickens were selected at 2 weeks, the same separation, faecal sample collection time points and collection procedure used for summer sampled chickens was followed. The demographic data of the individual samples and the 10 chickens are shown in Table S1 . Notably, at age 7 weeks, only two samples were obtained from summer samples, while the remaining 3 chickens under study whose faecal samples were not obtained had been sold at the time of sample collection. This reduced the expected sample number from 30 to 27. Hence, a total of 27 faecal samples ( n = 12 for summer) and ( n = 15 for winter) were achieved and stored at -80 °C.
Antibiotic pretreatment
The treatment of faecal sample with antibiotics in this study was employed as an enrichment process of selectively depleting/minimizing the interferences of non-viral nucleic acids. Pre-treatment of the faecal samples with antibiotics was done as described by Theuns et al. [ 25 ]. A 20% chicken faecal suspension was formulated by adding 200 mg of chicken feces into 1 ml of freshly prepared phosphate-buffered saline (PBS) (Sigma Aldrich, USA) at pH 7.5 containing 1000 U/ml penicillin, 1 mg/ml streptomycin, 1 mg/ml gentamicin and 500 U/ml amphotericin B (all from Sigma-Aldrich, USA) for antibiotic treatment. The faecal suspensions were homogenized at 3000 rpm for 1 min.
Viral RNA enrichment
Enrichment of viral particles was carried out using the standard NetoVIR protocol [ 26 ]. Following homogenization, a centrifugation step was done at 17,000 g for 3 min, prior to filtration. The only modification to this method was in the filtration stage, simultaneously carried out in two steps, using a 0.8 μm syringe filter (Sigma-Aldrich, USA) and thereafter with a 0.45 μm pore size (Merck, Millipore) to filter off largely sized nuclei, mostly those pertaining to bacterial cells. The resulting filtrate obtained from each sample was subjected to nuclease treatment using a cocktail of degrading enzymes; 2 µl of 25 U/µl benzonase nuclease (Sigma-Aldrich, USA) and 100 U/µl micrococcal nucleases (Thermo Scientific, Massachusetts, USA) combined with 7 µl of freshly prepared buffer containing 1 M Tris buffer (Merck, Germany), and 30 mM MgCl 2 (Sigma Aldrich, USA) and 100 mM CaCl 2 (Sigma Aldrich, Missouri, USA), pH 8.0, incubated for 2 h at 37 °C to destroy the naked free-floating nucleic acids. The nuclease enzyme was inactivated with 0.5 M EDTA. Total viral RNA was extracted using QIAamp viral RNA mini kit (Qiagen, Hilden, Germany) following the manufacturer’s instructions, without the use of carrier RNA. To deplete viruses with DNA genomes, DNase treatment was performed in 50 µl reaction mix using the DNase I M0303S kit (New England Biolabs), while column purification and concentration was carried out with the RNeasy Plus Micro Kit (Qiagen Hilden, Germany) according to their manufacturer’s guidelines. To further enrich for viral RNA, the host ribosomal RNA (rRNA) depletion was achieved using the NEBNext ribosomal RNA depletion (Human/Rat/Mouse) kit (New England Biolabs, Massachusetts, USA) following the manufacturer guidelines. All viral RNA quantifications were done using a highly sensitive RNA-specific fluorometric method on Qubit 3.0, with Qubit HS RNA reagent (Life Technologies, USA). Four negative controls were incorporated at different stages to monitor kitome contamination, and their details are described in Table S2 .
Library preparation and sequencing
Prior to library preparation, cDNA synthesis and whole transcriptome amplification (WTA) of the purified ribosomal depleted RNA samples was done as described by [ 27 ]. This WTA approach involves a first step of reverse transcription of RNA into complementary DNA, flanking the primer ligation sites, and a final amplification step of the cDNA library independent of poly A-tailing and oligo d(t) priming. The libraries of all samples were individually prepared using the QIASeq FX Single Cell RNA Library Preparation Kit (Qiagen, Hilden, Germany) following the manufacturer’s guidelines. The concentrations of the resulting cDNA libraries were determined fluorometrically, and their fragment sizes assessed using the Agilent 2100 Bioanalyzer (Agilent Technologies, USA). Additional information regarding the chicken faecal samples collected are found in the supplementary data, including the quantitative data of individual chicken faecal samples at key enrichment steps (Table S3 and S4 ). The constructed cDNA libraries were sequenced on a MiSeq platform (Illumina, San Diego) (151 × 2 cycles using a V3 600 cycle kit) alongside four control samples at the next-generation sequencing unit of the University of the Free State, Bloemfontein, South Africa.
Bioinformatics analyses
All processing of the resulting raw paired-end reads retrieved as FASTQ files was carried out using Genome Detective (GD) virus tool- panviral version 2.52 [ 28 ]. Briefly, this automated high performance online pipeline for viral detection incorporates Trimmomatic [ 29 ] and FastQC integrated FASTQ [ 30 ] for the removal of low quality, uninformative reads, as well as in-depth quality control. Candidate viral reads are then identified using the protein-based alignment method DIAMOND [ 31 ] against a subset of the Swissprot UniRef90 protein database to improve sensitivity and speed. The database contains more than 490,000 representative clusters of proteins linked to taxonomy IDs and is constantly updated. By performing the primary search at amino acid (aa) level, GD can accurately classify reads that have diverged from the references on nucleotide (nt) level. Short reads representing the same viral species are separated into separate groups, or buckets. Each bucket contains all reads from a single taxonomy ID based on the Least Common Ancestor (LCA) of hits identified by DIAMOND score. Each bucket is then de novo assembled separately using metaSPAdes [ 32 ] for paired-end reads and scaffolds classified. BLASTx and BLASTn was used to search for reference sequences against the NCBI RefSeq virus databases to confirm that the viral taxonomic ID of the resulting de novo contigs produced agrees with the bucket and identify candidate reference sequences of the resulting de novo contigs produced against NCBI RefSeq virus database. The GD pipeline then combines the total blastn score and blastx score results for every detected contig to simultaneously take into account amino acid and nucleotide similarity and selects five best scoring references for each contig to be used during alignment. Finally, the contigs for each individual species are stitched together using Advanced Genome Aligner (AGA) [ 33 ] to produce the consensus sequence. In this study, viruses were assumed to be contaminants due to index-hopping from another library if the total read count was less than 0.1% of the most abundant read count of the same virus(es) and cross-checked with a pre-existing compilation of viral contaminants [ 10 , 34 ]. Also, viruses detected in the negative control libraries of reagent mix incorporated at different stages, and/or belonging to the same clades as those detected in blank libraries was presumed to have originated from contamination that is most likely linked to laboratory reagents [ 35 , 36 ]. Hence, those viruses were eliminated from the chicken libraries and excluded from all further downstream investigations.
Viral diversity and abundance across age groups and seasons
To adjust for variations in read depth across libraries, the measure of abundance was expressed as the read count of reads per million, achieved by dividing the read count by the total number of reads in the library and then multiplying the quotient by one million. The effects of age and seasons on chicken gut virome diversities was investigated using ecological diversity metrics (alpha and beta diversity). While the alpha diversity measures explored were Sobs (number of observed genera), Shannon-Weiner and Simpson index, the beta diversity was determined using the abundances of the principal coordinate analysis (PCoA) based on the Bray-Curtis dissimilarity index [ 37 ] and Jaccard presence of absence theorem [ 38 , 39 ]. The statistical evaluation of the alpha and beta diversities was achieved using Kruskal-Wallis’s rank sum and Adonis test using the Bray-Curtis beta diversity results computed with 1000 replicates, respectively on sample sequences from each sample as well as each group.

Data visualization
The data visualizations and statistical analyses employed in the present study were mainly performed using the R Software (version 4.3.0)). The abundance data generated as reads and taxonomy tables were used as input files for further visualization on R, while utilizing a suite of packages including Vegan [ 40 ], ggplot2 [ 41 ], cowplot [ 42 ], pheatmap [ 43 ], and tidyverse [ 44 ]. Determination of differences between the sample groups observed in relation to chicken age and seasons was explored. Linear models and permutation analysis of variance were employed to test for these differences in richness and diversity indices across groups (age and season).
Phylogenetic analysis
The analysis of genetic phylogenies was conducted with only viruses having full-length genomes with ≥ 90% of its total genome coverage or partial genomes that have complete RNA-dependent RNA polymerase (RdRp) genes. Multiple sequence alignment was performed in the MUltiple Sequence Comparison by Log-Expectation (MUSCLE) software [ 45 ] comparing them with global sequences retrieved from GenBank and best hit results of BLAST searches with an expected e-value threshold 1 × 10 − 3 . The maximum likelihood method was applied to infer phylogenies using the software Molecular Evolutionary Genetics Analysis (MEGA v 11.0) [ 46 ]. The resulting phylogenies were visualized and annotated using FigTree v1. 3.1 [ 47 ].
Ethics approval
The ethical approval of this study was obtained from the Animal Research Ethics Committee (AREC) of the University of KwaZulu-Natal with the protocol Ethics Number, AREC 012/020. In addition, in compliance with Sect. 20 of the Animal Disease Act (Act No 35 of 1984), a research permit was obtained from the South African Department of Agriculture, Land Reform and Rural Development, with the reference no 12/1/5/4 (1511AC). Furthermore, for faecal samples, a provincial no restriction permit was obtained from the KwaZulu-Natal Department of Agricultural and Rural Development.
Results and discussion
Overview of viral metagenomic analysis.
In this study, a total of 9,429276 sequence reads, were generated from 27 cDNA libraries of chicken faecal samples and a notable Phred score of 38 achieved across all samples using the Illumina Miseq. The unassigned reads accounted for 5,559638 reads; a larger proportion of the reads generated in this study. It was extrapolated that slightly above half of the generated reads were non-viral reads, while viral reads with hits to eukaryotic viruses far outweighed those aligning to phages (0.06%), insects (0.04%) and plant (0.03%) viruses with a 99.88% prevalence (Fig. 1 A). Despite the lower viral reads, from a total of 8,722,416 reads after quality check, 3,869638 were assembled into 4328 viral contigs. Higher dominance of non-viral read to viral reads has been reported in metagenomics studies, including domestic birds [ 48 , 49 ]. Among 48 viral species identified, 15 viral species, majorly comprising avian viruses had complete and near complete genomes (> 95%) (Table 1 ). In addition, other viral species with less than 95% genome, such as Quail picornavirus QPV1/HUN/2010 , Gallivirus A and Megrivirus C , had full length RdRp or polyprotein gene except Infectious bursal disease virus from 7W5 with only 23% genome coverage of its segment B.
Chicken faecal RNA virome
The taxonomic analysis of the 3,869638 viral reads obtained revealed a total of 48 different viral species spanning across 15 viral families, 21 genera and some unclassified viruses. Based on genome type, a total of 43 viral species pertaining to RNA viruses, 2 DNA tail phages (from Siphoviridae family) and 3 unclassified viruses (Fig. 1 B). It was observed that within the RNA viral species obtained, 25 were dsRNA viruses (75.88%) while 18 were ssRNA viruses (24.01%), with total viral read percentages 75.88% and 24.01% excluding phage and unclassified viruses (Fig. 1 B). The two dsDNA tail phages ( Escherichia virus DE3 and Lambdavirus lvO276 ) recovered occurred only in one winter sample each, 7W3 and 2W4 respectively. Notably, previous studies on chicken gut virome reported the presence of tail phages from families Myoviridae Podoviridae and Siphoviridae in American [ 2 ] and UK [ 48 ] chickens, despite the former employing DNA removal procedures. Also, the absence of avian DNA viruses associated with the gut of chicken could mean that the enrichment and DNase treatment procedures undertaken were effective. The number of RNA viruses identified showed high RNA viral diversity.
The classification of identified viruses based on their established host specificity, exhibited a remarkably broad host range with the avian viruses having the highest prevalence of 48% [ 23 ] while mammalian and plant viruses accounted for 12.5% each (Fig. 1 C). Phages were the least abundant, constituting only 4% of the host-virus population while Insect viruses accounted for 8% of the population (Fig. 1 C), in addition to 3 Unclassified ShiM-2016 viruses (Table S5 ). The considerably high prevalence of avian viruses can be attributed to the targeted enrichment strategy utilized which significantly reduced the presence of non-viral genomes.
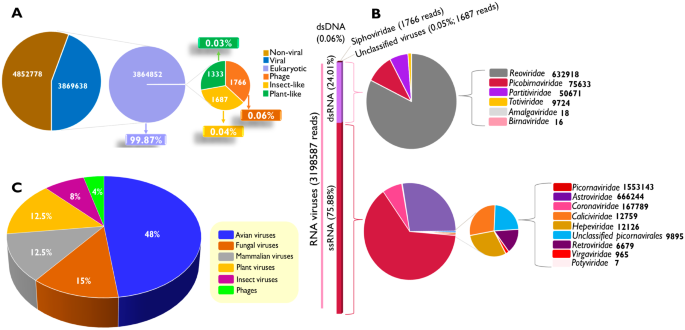
The distribution of sequence reads obtained from metagenomic analysis chicken faecal samples. (A) . The distribution of viral and non-viral sequence reads obtained from studied chickens (first pie), the middle pie depicts the prevalence of eukaryotic viral reads, from which smaller proportion of viral reads (%) were illustrated in the last pie. (B) The composition and diversity of viruses identified in chickens based on reads. The left bar denotes the numbers of assigned viral reads by genome type in brackets, RNA viruses (ssRNA and dsRNA). The pie charts show the composition of viral families identified by total number contig of its respective members in bold faces. (C) A Pie chart of the percentage host distribution of 48 different viral species identified from the study as correlated by the corresponding color in legend on the right
Members of seven viral families, Picornaviridae, Reoviridae, Astroviridae, Coronaviridae, Caliciviridae, Picobirnaviridae and Retroviridae were majorly to found across samples. However, it is noteworthy that the most abundant viral families are Picornaviridae, Astroviridae , Reoviridae and Coronaviridae with 1,550559, 666,244, 632,918 and 167,789 viral reads, respectively (Fig. 2 ; Table S5 ). The recovery of these viral families across samples may imply that they are more often detected from chickens. Importantly, the occurrence of these viral families in this study is consistent with previous reports on metagenomic investigation of chicken gut virome [ 1 , 2 , 11 , 48 ]. These studies have commonly identified all the viral families with exception to only few studies reporting member of the Retroviridae mainly in diseased chickens presented with Rou sarcoma or avian leukosis lesions [ 50 , 51 ]. Notably, the observed most abundant viral families ( Picornaviridae , Astroviridae , Reoviridae and Coronaviridae ) have been reported from similar studies on chicken virome from Netherlands [ 49 ], Brazil [ 11 ], UK [ 48 ], and Switzerland [ 52 ]. In addition, tracheal virome and respiratory studies on broiler chicken reported only two of these families, Picornaviridae and Coronaviridae in high abundance [ 12 , 53 ].
At species level, Sicinivirus A and Avian coronavirus were found be abundant across all sample group (Fig. 2 ). In addition, viral specie read abundance for each individual sample showed that many viral species from Picornavirida e family were found in nearly all samples (Fig S2 ). Based on the occurrence of viral species across the 27 samples, Picornaviruses , Sicinivirus A and Gallivirus A were highest, occurring in 27 and 26 samples respectively while nearly fungal and plant viruses had low occurrence across samples except the Infectious bursal disease virus (Fig S3 A). At family level, Picornavirida e was 100% prevalent across the 27 samples followed by Reoviridae , occurring in 24 (88.9%) samples (Fig S3 B). Recent studies have identified a variety of picornavirus genera in seemingly healthy bird species from different avian orders particularly, the Galliformes [ 12 ], Anseriformes [ 10 ], and Charadriiformes [ 9 ]. Hence, it could be deduced that picornaviruses are normal flora of the chicken’s gut since studies have reported them to be prevalent in healthy and/or diseased states [ 1 , 11 , 48 , 52 ] and across different developmental stages [ 1 , 12 , 49 ].

The overall read abundance of viral species from 27 chicken study samples across 2-, 4- and 7-weeks age groups categorized by summer and winter sample collection time points
Overall, the data obtained from this metagenomic study of asymptomatic South African chickens reveals high faecal RNA viral diversity that may be circulating in poultry farms. In addition, it could be highlighted that even in the absence of an observable overt disease condition, the gut RNA virome of chicken are highly diversified.
Identification and characterization of avian RNA viruses from asymptomatic chicken faeces
A total of 3,129,023 viral reads were assigned to avian RNA viruses encompassing 11 viral families (Table S6 ). This includes three dsRNA avian viral families ( Reoviridae, Birnaviridae and Picobirnaviridae ) comprising five viral species, 6 distinct ssRNA viral families with 14 viral species and unclassified ssRNA viruses (Table S6 ). The ssRNA viral families ( Coronaviridae, Picornaviridae , Caliciviridae , Astroviridae , Retroviridae , and Hepeviridae ) yielded about 77% (2,418,740) of the entire reads across all families including non-avian viral reads (Table S6 ).
Three reoviral species were obtained in this study from two genera, Orthoreoviru s and Rotavirus . The two rotaviral species obtained were Rotavirus F and Rotavirus G with 205,450 and 413,022 reads, respectively (Table S6 ). The nucleotide and protein-coding sequences of all eleven segments of Rotavirus F and Rotavirus G were obtained from 2W1, 2W2, and 2W2 for the former, and 2W3 and 7W5 for the latter. Seasonally, Rotavirus G prevailed in both seasons, while Rotavirus F only occurred in winter samples only which may imply a seasonal emergence of Rotavirus F in for the studied chickens. The phylogenetic result of Rotavirus G , VP4 segment with 100% genome coverage from sample 2W3 and VP7 segment from 7W5 showed that the virus is most similar to the VP4 of strain (YP_008136232.1) (Fig. 3 A) and VP7 of strain (YP_008136233.1) (Fig. 3 B) of German chicken/03V0567/DEU/2003, respectively. It was also observed that it clustered distinctively as a sister clade with the RVG/chicken/ZAF/MRC-DPRU1679/2011/Gxp strain previously isolated from South Africa chicken from the Limpopo province (Fig. 3 C) while the VP4 segment of Rotavirus F from 2W1, was most related to Rotavirus sp. from China (UHS71878.1) (Fig S4 ). The Orthoreovirus had partial and nearly complete coding gene sequences of only 7 segments (L1, L2, L3, M1, M2, S3, and S4), excluding the S1 segment coding for the sigma C protein. The segments include L1 (95%), L2 (98.7%), L3 (99.0%) and M2 (87.5%) from 2W1, 2W2 had only segment L2 (99.7%) and M2 (99.4%), while 2W4 had L1 (93.3%), L2 (99.7%), M1 (94.4), M2 (99.4), M3 (90.4%), S3 (82.6%) and S4 (91.9%). Phylogenetic result of nearly complete Lamda B of Avian Orthoreovirus showed highest relatedness to isolate Reo/PB47I16/Switzerlamd/2019 (Fig. 4 ). Overall, the higher viral abundance of Avian Orthoreovirus and Rotavirus G at both 2 summer and winter samples 2 weeks samples and Rotavirus F at week 2 winter samples may suggest high susceptibility of juvenile chickens to reoviral infections particularly rotaviruses, which are listed among the major causes of death in young chickens. Recent studies have demonstrated that younger chicks are more highly susceptible to reoviral infections, particularly Rotavirus G and F [ 9 , 54 ]. In addition, studies have attributed Avian Orthoreovirus to arthritis and tenosynovitis [ 55 , 56 ] while Rotavirus G and F have been associated with acute gastroenteritis in poultry [ 11 , 52 , 54 ]. Although, a minimal rate of enteritis is expected for large commercial chicken flocks, the chickens in this study were asymptomatic, showing no signs of any disease conditions as confirmed by the poultry farmers through their veterinary experts. Currently, there is limited information on Avian orthoreoviruses, Rotavirus G , and F from South Africa for more analysis and comparison, and this may imply that the true diversity of reoviruses including the subtypes circulating in South African chickens may not have been fully unravelled.

Phylogenetic analysis of Rotavirus G virus segments, using the maximum likelihood, Tamura 3 parameter model. The trees were midpoint rooted for clarity and the branch length support was estimated using 1000 bootstrap replicates and virus labels in red fonts denotes the viruses identified from chicken faecal sample in this study. A . Phylogeny of theVP4 segment of Rotavirus G . B . Phylogeny of the VP7 segment of Rotavirus G. C . The phylogeny of the NSP3 segment of Rotavirus G

The maximum likelihood phylogenetic analysis of Avian orthoreovirus using the Lambda-B (RdRp). The tree was midpoint rooted for clarity and the branch length support was estimated using 1000 bootstrap replicates with the Tamura 3 model, while Avian orthoreovirus identified in this study is depicted in red fonts. The viruses in blue font had the highest identity with the identified virus
Birnaviridae
In this study, Infectious bursal disease virus or Gumboro disease virus segment B was identified with 4 viral contigs and a total of 626 nucleotides from only one sample, a 7-week winter (7W5) sample. This virus VP1 gene shared it highest nt identity of 95% with a novel variant of Infectious bursal disease virus genotype A2dB1b (OR791872.1) isolated from Egyptian chickens. It also a had 90% and 94% nt and aa identity with the RdRp (VP1) of the European “very virulent strain” isolate “UK661” of Infectious bursal disease virus (NC_004179.1) isolated from chicken. The occurrence of this Avibirnavirus is surprising and of great concern especially since the studied chickens’ received vaccine against Gumboro disease virus before 2 weeks of age. Nevertheless, since this virus was detected from only one sample (7W5), it could be speculated that it may be an instance of missed vaccination for that chicken. Further phylogenetic analysis of this virus could not be achieved as result of very low genome coverage (23%) of the recovered segment B. Also, the administered vaccine Nobilis® Gumboro D78 was not available across all GenBank databases for comparison with the 7W5 identified from this study, hence we could not confirm if it was a vaccine strain. However, recent studies have reported Infectious bursal disease virus to be higher in birds of 3–7 weeks of age, often causing bursal lesions and immune suppression that could result to susceptibility to other secondary infections, which may lead to the death in older birds while infections in younger birds may be mild [ 57 , 58 ].
Picobirnaviridae
Chicken picobirnaviruses in addition to other mammalian picobirnavirus types identified from this study are still under unclassified isolates by the ICTV nomenclature. Chicken picorbirnavirus had a total of 2061 reads from nearly all sample age groups and seasons except 5 samples (7S2, 2W4, 2W5, 7W4, and 7W5). However, only the segment RNA 2 which is the polymerase gene were obtained across samples with 99.9%, 99.6%, and 97.8% gene segment coverage from samples 2S2, 4S5 and 4S1, respectively (Table S1 ). Further analysis of the RdRp gene (99.9%) of Chicken picobirnavirus from 2S2 showed 81.3% highest Identity with putative RdRp of Picorbirnavirus sp isolate 2282-K141-180086 (USE07851.1) isolated from Chinese chicken faeces (Fig S5 ).
Coronaviridae
Avian coronavirus or Infectious bronchitis virus was obtained from this study accruing about 167,789 reads, across the 3 ages groups and two seasons. This virus demonstrated a 74% prevalence occurring in the about 20 samples out of 27 samples (Fig S3 b). Nearly complete genomes of Avian coronavirus were recovered from samples 2S4 and 2S5 with 99.3% and 99.0% nucleotide sequence coverage, respectively. Notably, complete coding sequence (CDS) and 100% sequence coverage of the OR1ab polyprotein, NSP1 to NSP16 proteins, 3a, 3b, 5a, and 5b proteins including the spike, envelope, membrane, and hypothetical proteins were recovered from sample 2S4. Analysis of the ORF1ab polyprotein showed best hit with 95% identity to QX-like Infectious bronchitis virus isolate ck/ZA/3665/11 from South Africa (AKC34132.1) (Fig. 5 A). Similarly, phylogeny result using the RdRp gene (NSP12) demonstrated a distinct cluster with the same ck/ZA/3665/11 South African strain as a sister clade to strain H120 and isolate ZJ971 of Infectious bronchitis virus from UK (QKV27937.1) and Chinese (ACH72792.1) chickens (Fig. 5 B). The studied chickens were vaccinated against Infectious bronchitis virus 4–91 serotype using Nobilis® IB 4–91 and importantly presented no observable disease symptoms. Hence, the high occurrence of the pathogen, across samples is of great concern since analysis of its gene segments showed that it not a vaccine strain. However, an American metagenomic study on the respiratory virome of broilers reported the predominance of Infectious bronchitis virus in the normal healthy virome of studied chickens [ 12 ]. Also, their occurrence though apathogenic, be attributed to high-rate viral transmission of Infectious bronchitis virus / Avian coronavirus among birds, particularly in multi-age commercial farm structures. Nevertheless, recent studies have established Avian coronavirus as a nephropathogene, causing frequent nasal discharge and conjunctivitis in poultry which could lead to death in chickens and turkeys [ 59 , 60 , 61 ].

Phylogenetic analysis of Avian coronavirus , ORF1ab and RdRp NSP12 using the maximum likelihood, Jone-Taylor-Thornton model. The trees were midpoint rooted for clarity and the branch length support was estimated using 1000 bootstrap replicates and virus label in red fonts denotes the viruses identified from chicken faecal sample in this study. (A) Phylogeny of the ORF1ab segment from sample 2W4. (B) Phylogeny of the 1ab gene RdRP (NSP12) from sample 2S4
Caliciviridae
The Calicivirus identified from this avian study belongs to the only species in the Bavovirus genera. This Bavarian virus identified had a total of 12,759 reads (Table S6 ), with its highest nt sequence coverages of 99.3 and 98.5% emerging from 2S4 and 2S5 samples. The complete CDS, polyprotein and VP2 gene segments of this Bavaria virus were retrieved. Further blast analysis using the polyprotein revealed this virus was most similar with 85% nt and 96.1% aa identity to a calicivirus identified from American chickens (KY120883.1). It was thus seen from this study that the Bavaria virus often occurred within samples from younger chickens after which they gradually disappeared through 4 weeks, becoming non-existent at 7 weeks. The phylogenetic result of Bavaria virus with 99.3% genome coverage from chicken sample 2S4 depicted in Fig. 6 showed that this virus is most similar with the provisional reference sequence (RefSeq) of novel Calicivirus chicken/V0021/Bayern/2004 (NC_075411.1) virus from Germany (Fig. 6 ). In addition, it was observed that this virus clustered with the only member of this genus Bavovirus . Further alignment of the virus against the non-redundant (nr) NCBI database also showed it shared only 85% nt and 96.1% aa with Chicken calicivirus strain RS/BR/2015 (KY120883.1) in a different clade in Nacovirus genus.

Phylogenetic analysis of the polyprotein gene of Bavaria virus , using the maximum likelihood (Jone-Taylor-Thornton model). The trees were midpoint rooted for clarity and the branch length support was estimated using 1000 bootstrap replicates. The virus in red font with black node was identified from chicken faecal sample in this study
Picornaviridae
Picornaviridae was observed to be the most abundant viral family with a total of 1,550559 viral reads from eight species belonging to five genera (Table S5 ). The species identified in this study includes, Megrivirus A and C, Gallivirus A, Sicinivirus A, Avisivirus B, Orivirus A, Chicken picornavirus 1 and Quail picornavirus QPV1/HUN/2010. The prevalence of individual species of the picornaviruses identified in this study is shown in Table 2 .
The species Sicinivirus A had a 100% prevalence across the 27 samples with a total of 195,040 reads (Table S5 ). Partial nucleotide sequence (96.0%) of Sicinivirus A was obtained from sample 7W2 with 144420.35 reads per million (rpm). In addition, nearly complete DF53_gp1 CDS (99.5%), complete RdRp gene crucial for its replication known as 3D pol, alongside the complete sequences of the VP0 to VP3, 2 A, 2B, 2 C, 3 A, 3B and 3 C proteins were recovered. Phylogeny showed its closest relative to be strain UCC001, obtained from chicken cecal samples from Ireland (YP_009021777.1) (Fig. 7 ). Megrivirus A and Megrivirus C were obtained with 2303 and 1,121,717 reads, respectively. The results of BLAST alignment at e-value 10 − 3 of Megrivirus A showed its best hits with a Melegrivirus A virus conserved in turkey from Hungary (KF961188.1) and a reference sequence of Turkey hepatitis virus 2993D at 77.1 and 78.3% identity respectively. In addition, Gallivirus A accumulated about 100,247 reads and demonstrated a 96.3% prevalence for picornaviruses (Table 2 ; Table S6 ) with highest nt genome coverage of 94.4% from sample 2S3. The complete (100%) protein sequences of 3 A, 3B,3c and 3D (RdRp) genes of Gallivirus A was recovered and phylogenetic analysis of its 3D pol showed it identical to 3D of Gallivirus A1 isolate 518 C from Hong Kong (Fig. 7 ). Furthermore, Avisivirus B viral species occurred in 7 samples with highest genome coverage of 92.7% from 2S1. Alignment results of its complete 3D pol RdRp gene showed its best hit of 85.8% identity with Chicken picornavirus 2 isolate 44 C from Hong Kong (YP_009055013.1) (Fig. 9 ). Similarly, Orivirus A showed a 51.9% incidence across samples (Table 2 ) with the highest nt sequence coverage of 96.1% from 2W2 sample in which the complete PI35_gp1 CDS and polyprotein genes was recovered. Phylogenetic results showed that it is most related to the polyprotein gene of strain Pf-CHK1/OrV-A2 from Hungarian chicken (Fig. 7 ). Additionally, two unclassified avian picornaviruses isolated from this study are Chicken picornavirus 1 and Quail picornavirus QPV1/HUN/2010 with 88.9 and 70.4% prevalence across samples, respectively (Table 2 ). Chicken picornavirus 1 had very low coverage (59.8%), while Quail picornavirus QPV1/HUN/2010 was observed to have its highest nt sequence coverage of 94.2% from 4W5 sample whose phylogeny showed close relatedness with Picornavirales sp. ULF99733.

Phylogenetic analysis of members of the Picornaviridae , using the maximum likelihood using the polyprotein gene (Jone-Taylor-Thornton model). The trees were midpoint rooted for clarity and the branch length support was estimated using 1000 bootstrap replicates. Viruses in red fonts were identified from chicken faecal samples in this study
Retroviridae
The two retroviruses obtained from this study were Avian leukosis virus and Rous sarcoma virus , accruing a total of 4478 and 2201 reads, respectively (Table S6 ). Although these viruses were identified in very low genome coverage between 36.3 and 63.1%, the two viruses occurred in both summer and winter samples and appeared to have high hit up to 99.6% identity with the same clones Avian leukosis virus subgroup E from the UK (MT263508.1) and USA (MF817820.1). Retroviruses causing infections in birds are conserved within the Alpharetroviru s. Major cancerous infections occurring in avian species including chickens, have been majorly attributed to these two major retroviruses, Rous Sarcoma virus [ 54 ] and Avian leukosis virus [ 55 , 56 ]. The presence of these viruses from both summer and winter samples in this study, though at lower coverage show that they may be circulating in commercial poultry farms in South Africa.
Hepeviridae
Avian hepatis E virus or its new name Avihepevirus magniiecur , was identified in 10 samples mainly from the winter season yielding a total of 12,126 reads (Table S6 ). Its near complete genome (98.8%) with complete CDS (BA77_gp1, gp2 and gp3) and non-structural polyprotein was obtained from 7W4. Additional homology comparison with its recovered complete genome revealed that it shared 83.7% homology with a novel divergent isolate 19/03914 Avihepevirus magniiecur virus from HSS chickens and pheasants in France (ON922634.1) proposed to belong to genotype 7 [ 62 ]. In addition, it also shared 83.66% and 83.68% with isolate W838-14 and W865-14 from liver samples of Australian Gallus gallus believed to be a recombinant progeny of US (genotype 2) and Hungarian strain (genotype 4) associated with poultry importations. The identification of this virus in ten chicken samples in both summer and winter periods at high genome coverage (98.8%) suggests a natural subclinical infection of the studied chicken flock. Avihepevirus magniiecur , has been numbered as an emerging zoonotic pathogen capable of infecting humans with significant public health concern [ 63 , 64 ]. Recent studies have identified these aHEV genotypes from different countries to be a major cause of hepatitis-splenomegaly syndrome (HSS) or big liver and spleen disease (BLSD) in chickens [ 62 , 65 , 66 ]. Chickens infected with aHEV may advance from subclinical to rapidly increased mortality and a severe decrease in egg production [ 67 ]. Nevertheless, some studies have recovered RNA genomes of aHEV and its antibodies from apparently healthy chickens [ 65 , 68 ]. Hence, while the detection of aHEV in apparently healthy layer chickens in this study may not have immediate public health implications as a zoonotic pathogen. However, it does raise concerns as to whether HSS occurs more frequently in South Africa, often undiagnosed or unreported. Hence, further studies including different geographic locations in South Africa is needed to determine the genotypes, prevalence, and spread of aHEV in South Africa.
Identification of non-avian viruses from chicken faeces
The non-avian viruses identified in the current study were mainly food-related viruses, tailed DNA phages, fungal and mammalian viruses with majority of them occurring in very low read abundances (Table S7 ). The two dsDNA tailed phages identified, Escherichia virus DE3 and Lambdavirus lvO276 were both from Lambdavirus genera of Siphoviridae obtained from 7W3 and 2W4 samples respectively (Table S7 ). In addition, distinct fungal viruses belonging to two viral families, Partitiviridae and Totiviridae were found, mainly occurring in low read abundances and in less than three samples, except Aspergillus fumigatus partitivirus 2 found in 9 samples that encompasses both seasons. Furthermore, diet-associated, and insect-like viruses, Hubei orthoptera virus 1 , Hubei picornavirus 24 and Wuhan insect virus (Table S7 ). The occurrence of tail phages Escherichia virus DE3 and Lambdavirus lvO276 was not expected due to the DNA depletion and rRNA enrichment steps employed. Nevertheless, previous studies have reported tail phages from families Myoviridae Podoviridae and Siphoviridae in American [ 2 ] and UK [ 48 ] chickens, despite the former employing DNA removal procedures. Aspergillus fumigatus partititivirus has gained significant attention as a fungal viral pathogen in poultry as an opportunistic pathogen causing immune suppression and respiratory related diseases in chickens [ 69 ]. The identification of the fungal virus, Aspergillus fumigatus partitivirus 2 , is of concern as it is known to cause opportunistic immune related infections in chickens. However, recent studies have also shown that this virus lacks host specificity [ 69 , 70 ] which may be explained by its presence in the studied chickens. While the identification of food related viruses can be associated with different plant sources of the diet regimens given to chickens, the insect-like viruses may be attributed to use of insects as feed ingredients for poultry. The use of insects seems to have gained attention due to their potential as a sustainable and alternative protein source in animal diets [ 71 ]. Previous study by Wille, Harvey [ 72 ] identified the Taggert virus , a nairovirus from chinstrap penguins, as well as a new Bruthen virus from the family Phenuviridae , a sister family of Phlebovirus that consists of tick association viruses. With the identification of viruses thought to be plant, arthropod and insect- associated from bird’s further research is needed to determine the true host of these viruses and the effect of diet sources on the diversity and abundance of RNA viruses in birds.
The mammalian viruses obtained from the investigated chicken samples includes an unclassified Picornavirales Tottori-HG1 and non-avian unclassified Picobirnaviridae members (Table S7 ). While Picornavirales Tottori-HG1 virus , had a low genome coverage ranging within 38–45%, the non-avian members of Picobirnaviridae were moderately abundant across samples with good coverage ranging from 60 to 90%. The identification of diverse mammalian PBVs from more than 10 samples and at high genome coverage in this study is not utterly surprising. The viral family Picobirnaviridae lacks host specificity with only one genus Picobirnavirus and two genogroups, GI and GII. Currently, there is contradictory information regarding the real host of PBVs, owing to the short sequences used to identify PBVs which are not typical of the whole RdRp gene phylogeny, hence making their defined identification and classification difficult [ 73 ]. Not only have PBVs been found in humans, invertebrates, and birds, but they have also been hypothesized to be phages [ 74 , 75 ] and eukaryotic fungal viruses [ 76 , 77 ]. Although, the direct and indirect interactions between some of these mammal and poultry cannot be totally ignored. Particularly, humans are responsible for feeding these commercial chickens, dogs are common pets, monkeys are highly abundant in South Africa, and some commercial poultry farms rear other animals such as pig. Recent metagenomic investigation on the Oral RNA virome of backyard swine farms from the KwaZulu-Natal Province, South Africa, identified Picornavirales Tottori-HG1 , with results showing that it originated from Japan [ 78 ] and other studies reported Picobirnavirus dog/KNA/2015 in non-dog hosts [ 78 , 79 ] in South Africa. Nevertheless, it remains unclear and difficult to explain how the Otarine picobirnavirus occurred in the studied chicken. Notwithstanding, previous studies have identified viruses Otarine picobirnavirus , and Picobirnavirus dog/KNA/2015 in human respiratory samples [ 79 ] and swine faecal samples [ 78 ] in South Africa. Overall, it is imperative to elucidate underlying the transmission mechanisms and spillover events pertaining to various viruses affecting common farm animals, particularly with a focus on swine viruses, and their spillover and impact on poultry populations.
Variations in avian RNA viral abundance and diversity as a function of age and sampling season
Analysis of gut RNA virome structure of the faecal samples of chickens in this study revealed differences in diversity and abundances of viral species across the 27 chicken libraries investigated. There was no distinct trend in the dynamics of total viral read across various collated sample time points in the six categories analyzed, as shown in Table S5 . A look at the week two summer (2 S) and winter (2 W) groups showed that they had total viral reads of 312,202 and 1,581,024, respectively (Table S5 ). Hence, there seems to be an observed increase in viral reads between these seasons at week 2. The observed pattern was way different for the 4 weeks chickens which had comparable ranges of total viral reads, which were 256,094 and 224,309, respectively. Another contrasting phenomenon exhibited was in the 7-week samples, showing an opposite effect. It is noteworthy that the 7 W group exhibited a significantly higher number of total viral reads (799,296) in contrast to the 7 S group (29,114), with a nearly 4-fold increase (Table S5 ). For the two independent sample collection seasons, the findings indicate that during the summer season, there was a negligible reduction of viral reads in the 2 S and 4 S categories. However, a slight increase of viral increase was observed in the 4 S and 7 S chicken groups. Overall, the winter samples exhibited higher number of viral reads.
Based on relative standardised abundance, a high proportion of the relative standardised abundance estimates of sequence reads across all the libraries were classed as RNA viruses and the results of their relative abundance from individual sample and group are presented in Fig. 8 . The results showed that the most relatively abundant viral species reads across age groups are Avian coronavirus, Rotavirus G. Chicken astrovirus, Megrivirus C and Sicinivirus A (Fig. 8 ). In addition, Bavaria virus, Chicken picobirnavirus Chicken picornavirus 1 and Aspergillius fumigatus partitivirus 2 were moderately represented. However, the remaining viral species had very low abundance in individual samples, including those assigned in the other group as shown in Fig. 8 . It was interesting to see the changing dynamics of members of the Reoviridae , particularly, Rotavirus G and Avian orthoreovirus , which decreased with increasing age (Fig. 8 ). Notably, members of the Picornaviridae family were found throughout all age groups and seasons particularly, Sicinivirus A (Fig S3 B; Fig. 8 ). The observed prevalence of Reoviridae members, namely Rotavirus G , and Avian orthoreovirus occurring in both winter and summer samples, with high abundance can be ascribed to the heightened vulnerability of juvenile chickens to reoviruses. This susceptibility arises from the ongoing maturation of their immune system, which is characterized by low immunity to infections. Recent studies have reported a high susceptibility rate of rotaviral infections in younger birds [ 52 , 54 ], , which is a major cause of runting-stunting/malabsorption syndrome resulting in delayed development and lower productivity in chickens. In addition, the observed significant decrease in reoviruses at 7 weeks may be further explained by the stable adaptation features exhibited by these mature chickens with full grown immune systems. The observed prevalence of picornaviruses may imply that major members of this viral family are commensal and hence may form part of the normal flora of the chicken gut virome. Moreover, different studies have reported picornaviruses to be prevalent in both healthy [ 10 , 12 , 80 ] and diseased [ 11 , 48 , 52 , 54 ] bird species including chickens.

The relative abundance profile of major RNA viral species across the chicken faecal samples at different ages (2, 4 and 7 weeks) and seasons (Summer and winter)
In addition, it is notable that certain viral families and/or species within the examined chicken samples exhibited unique trends with respect to age and seasonal variations whereby they are detected in summer samples but are entirely absent in winter samples, or conversely. Notably, it was also revealed that the winter sample were mainly characterized by Chicken astrovirus and Rotavirus G . Interestingly, this observed seasonal distribution exhibited by some viral species, such as Rotavirus F, genus Lamdavirus, Infectious bursal disease virus, Festuca pratensis almagavirus 1, Fusarium poae virus 1 , and unclassified RNA virus ShiM-2016 may be attributed to their emergence during the winter period. Similarly, the exclusive occurrence of Tomato mosaic virus, Pepper mild mottle virus , and some partitiviruses ( Botryotinia fuckeliana partitivirus 1, Cryptosporidium parvum virus 1, Pythium nunn virus 1, Ustilaginoidea virens partitivirus 2 , and Verticillium dahliae partitivirus 1 ) during the summer periods, may either mean that the viruses are more predominant during the summer or may be associated with the diet (milled grains) taken by these chickens which may carry these fungal viruses. Overall, at viral family levels, all viral families occurred throughout the two seasons explored with exception to some non-avian viral families, Siphoviridae, Amalgaviridae, Partitiviridae, Potyviridae and Totiviridae comprising of fungal and plant viruses. However, at species level viral diversity varied with some viruses occurring in winter period while being entirely absent in summer or vice versa for instance; Rotavirus F . In addition, viral abundance varied more with age rather season, where the effect of age chicken may have been higher than the seasonal effect in the abundance of some of these viruses. Nevertheless, their occurrence by mere chance events cannot be entirely ruled out, particularly with viruses that occurred in lower abundance and in < 3 samples. To the best of our knowledge, the present study is the first to explore the effect of season on viral abundance in chickens, particularly from the African continent. Based on available studies as at the time of this study, there was limited information on the effect of season on viral abundance and diversity in domesticated birds. However, a recent Australian study on wild birds (Pacific black ducks, Chestnut teals, Grey teals and Wood ducks) explored the effect of season on the abundance of the viral families Picornaviridae and Parvoviridae [ 80 ]. Findings from their study showed that picornaviruses were mainly found during late autumn to late winter months while parvoviruses were found throughout the year. In addition, Vibin et al. [ 80 ], demonstrated that members of these two viral families varied not only in virus composition across species and time but also in their abundances, despite the sharing the same habitat at the times of sampling. Overall, avian viruses appear to be well represented in all chicken faecal samples. In addition, it could be concluded that about 20 RNA virus species were found in more than ten different chicken samples.
Impact of age and season on the alpha diversity and beta diversity of chicken RNA virome
Alpha diversity.
The results of the observed alpha diversity principally based on expected count for age and season presented in Fig. 9 showed similar total counts (observed) within ages with interquartile range positively skewed and with most values within the lower quartile (Fig. 9 A). For the two seasons, summer and winter, a slight difference was observed at 2 weeks in the within sample counts abundance with the summer sample a bit higher than the winter sample (Fig. 9 B). This is not the case with summer and winter samples at week 4, as they demonstrated to have similar viral counts while at week 7, the viral count for the winter sample was higher than that of the summer samples (Fig. 9 B). Similarly, the result of alpha diversity (within samples) using metrics that examines both species richness as well as evenness are illustrated in the box plots denoted as Fig. 9 C and D. The Shannon’s index considers richness more, while the Simpson’s index places emphasis on the evenness of the viral species. For Shannon diversity, the 4 weeks’ samples had the highest species richness among the three age groups studied (2, 4, 7 weeks). The Shannon’s richness based on season showed that the species dynamics of summer were evidently much higher than winter samples at 2 weeks (Fig. 9 C). In contrast to the trend observed at week 2 summer and winter samples, here, the summer and winter samples observed at week 4 appeared to be similar. However, at week 7, a slightly higher species abundance was observed for summer samples than its winter samples. The Simpson’s diversity findings were remarkably comparable to the Shannon’s diversity results, with the most species even samples detected at week 4 for age groups. (Fig. 9 D). In addition, the results of diversity measures based on seasons revealed that same species evenness was seen at both summer and winter sample at weeks 4 and 7. However, at week 2, the samples had more species unevenness for their summer and winter collection seasons (Fig. 9 D).

The alpha diversity metric determination of chicken RNA virome as a function of age and season. ( A ) Sobs by age ( B ) Sobs by season ( C ) Shannon-Wiener index, and ( D ) Simpson’s index
With the observed results within sample groups exhibiting similar diversity levels for Shannon and Simpson’s indices, the lack of distinct trend across age groups and seasons can be explained further. First, the viral abundance pattern within each age group can be homogeneous since these chickens are grouped, housed, fed, and vaccinated together, however most commercially bred chickens are multi-age in the same farm. In addition, the inconsistence and higher abundance shown by members of the older chicken at week 7 may be attributed to the lowered immunity of some their members in this group arising from different systemic and recurrent viral infections they may have suffered, leading to different survival mechanism by individual chicken in this age group. This can be explained further by the issue of chance events as has been observed for some transient viral infections, thus leading to chicken individual biased survival features or resistance to some viruses even within the same group. Furthermore, other environmental factors, such as sanitation, lack of biosecurity measures and the introduction of birds from other commercial farms may influence viral abundance.
Overall, the alpha diversity for age ( p = 0.116; df = 2) and seasons ( p = 0.172; df = 1) were not significant. Further analysis of variance using the non-parametric Kruskal-Wallis’s H rank sum test on alpha diversity within groups showed that the values for age ( p = 0.146; R 2 = 3.849; df = 2) and observed season ( p = 0.241; R 2 = 1.371; df = 1) were not significant. A post-hoc analysis, specifically the Bonferroni method and Turkey’s multiple comparisons conducted as a follow up to determine which groups means were not significantly different from each other, showed that the alpha diversity of all individually paired group was not significant ( p > 0.05). Hence, the observed ( P > 0.05) no significant effect observed for alpha diversity indicates that regardless of age or seasons, the overall viral abundance of samples within the same group are similar/homogenous. In addition, viral abundance was higher in juvenile chickens (2 weeks) between the three age groups which can be attributed to their immature immune systems resulting in increased susceptibility to infections. Similar no significant alpha diversity result was observed from a recent study that investigated the impact of age in an Anseriformes species, Ruddy turnstones with P-value > 0.05 [ 10 ].
Beta diversity
The results of the beta diversity metrics for age and season determined using the abundances of the principal coordinate analysis (PCoA) based on the Bray-Curtis and Jaccard showed that with exception of few outliers, the individual samples from weeks 4 and 7 samples clustered distinctively by their ages (Fig. 10 A and B). However, for both abundance (Bray-Curtis) and presence/absence (Jaccard) theorems, it was revealed that the 2 weeks samples were more radically spread, clustering with both the weeks 4 and 7 samples (Fig. 10 A and B).The observed distinct clustering for both indices (Jaccard and Bray-Curtis) between viruses at week 4 and week 7 may indicate a marked difference in viral diversity between the acclimatization stage (week 4) and the mature stage (week 7) of the chicken. Hence it appears that there is no relationship/ limited connectivity/viral sharing between weeks 4 and 7. However, for week 2, the observed clustering results indicate significant viral connectivity/sharing between week 2 and week 4 as well as week 2 and week 7. Hence it can be deduced that, some of the viruses identified from this chicken at week 2 followed through to week 4 and week 7.

Principal co-ordinate analysis on chicken viral abundance and diversity as functions of age and season, presented as ( A ) Bray-Curtis’s similarity index and ( B ) Jaccard index. The age means the individual age of birds whose samples were obtained while CSTs means the chicken seasonal collection timepoints
Statistical evaluation of the Bray-Curtis beta diversity metrics showed that the beta diversity between age groups were significant ( P = 0.01099; R 2 = 0.17085; df = 2), and consistent with that of the two seasons revealing high significant difference between the seasons ( P = 0.000999; R 2 = 0.24107; df = 1). Overall, based on the data sets in this study, the beta diversity (between groups) was revealed to statistically significant for both age and season ( p < 0.05) and indicates that viral abundance and diversities are greatly influenced by age and season between sample groups for chicken used in this study. Hence it could be deduced that the diversity and abundance between studied chicken samples groups may differ and may be dependent on distinct features characterising each group. Therefore, between group of one or more distinct feature, the diversity and abundance may be heterogenous. Different avian studies have demonstrated marked differences in viral abundance and diversities between sample sets characterized by different features such as age structure, seasonality, host species and latitude [ 80 , 81 , 82 ]. Similarly, microbiome studies have attributed bird’s age, feeding pattern/regimens, raising environment and body site to have great impact on the diversity of bacterial population in commercial layers hen [ 83 , 84 ]. Overall, the differences in diversities and abundance between groups whether age or season, may be attributed to factors such as varying temperature changes for season, different feed formulations in form of diet eaten by these chickens at different ages as well as different vaccines and/or treatment administered to chickens in a specific group which may influence viral abundance and diversities.
In this study, the faecal RNA virome of asymptomatic South African chickens were analysed at three developmental ages and over two seasons using mNGS. Viral enrichment and purification strategies adopted allowed significant recovery of viruses, including novel chicken astroviruses (CAstV), many previously known avian, mammalian, fungal and plant viruses that may be circulating in poultry farms. While some of these viruses identified were presumed to have commensal roles such as picornaviruses or may be opportunistic pathogens, viruses like rotaviruses, coronaviruses and Avihepevirus magniiceur have established severe pathogenic effects. The diversity and abundance of RNA gut viruses were significantly influenced between chicken groups characterized by different ages. In addition, it could also be concluded from this study findings that season play a role in the occurrence of gut virus(es) in chickens. While it is acknowledged that the study is limited by sample size, the findings have provided baseline information that future studies could build on for more encompassing generalization on the viral diversity of chickens in SA. The result from this study reiterates that despite the lack of discernible clinical manifestations, RNA viruses remain prevalent within the GIT of chickens. Thus, chickens may experience recurrent viral infections that could significantly impact the development of their normal gut virome and overall well-being. Hence, with the intensification of poultry systems, routine surveillance of chicken viruses is imperative to assess the risk of disease outbreaks in poultry farming. In addition, monitoring cross species transmission of RNA viruses will serve as an epidemiological step towards curbing the wild spread of viruses from poultry to mammals or conversely. Overall, the present study provided insights on the diversity of chicken faecal virome and would serve as a reference for future investigations, looking to compare virome of healthy and diseased chickens in South Africa.
Data availability
All data generated during this study are provided within the manuscript and its supplementary information file. All viral sequences analyzed have been submitted and released by the National Center for Biotechnology GenBank under the accession numbers PP228887-PP228924. Raw sequence data obtained from this study has been deposited in the NCBI Sequence Read Archive (SRA) under the BioProject ID: PRJNA1068849 (SRA accessions; SRR27722602-SRR27722628 and BioSample accessions; SAMN39605134-SAMN39605160).
Abbreviations
Gastrointestinal tract
Ribosomal ribonucleic acid
- Next-generation sequencing
Novel enrichment techniques of viromes
Genome detective
Viral protein
Non-structural protein
Chicken astrovirus
Avian hepatitis E virus
Picobirnaviruses
Principal coordinate analysis
Shah JD, Desai PT, Zhang Y, Scharber SK, Baller J, Xing ZS, et al. Development of the intestinal RNA virus community of healthy broiler chickens. PLoS ONE. 2016;11(2):e0150094. https://doi.org/10.1371/journal.pone.0150094 .
Article CAS PubMed PubMed Central Google Scholar
Day JM, Oakley BB, Seal BS, Zsak L. Comparative analysis of the intestinal bacterial and RNA viral communities from sentinel birds placed on selected broiler chicken farms. PLoS ONE. 2015;10(1):e0117210. https://doi.org/10.1371/journal.pone.0117210 .
Fitzpatrick AH, Rupnik A, O’Shea H, Crispie F, Keaveney S, Cotter P. High throughput sequencing for the detection and characterization of RNA viruses. Front Microbiol. 2021;12:190. https://doi.org/10.17268/sci.agropecu.2021.051 .
Article Google Scholar
Akinyemi FT, Ding J, Zhou H, Xu K, He C, Han C, et al. Dynamic distribution of gut microbiota during embryonic development in chicken. Poult Sci. 2020;99(10):5079–90. https://doi.org/10.1016/j.psj.2020.06.016 .
Africa UNMCS. Poultry industry faces mounting losses amid bird flu outbreaks. University of Nebraska Medical Center: Global Center for Health Security. 2023. Available: https://www.unmc.edu/healthsecurity/transmission/2023/09/05/south-africa-poultry-industry-faces-mounting-losses-amid-bird-flu-outbreaks/ (Accessed on 15 September 2023).
Uwishema O, Adriano LF, Chalhoub E, Onyeaka H, Mhanna M, David SC, et al. Bird flu outbreak amidst COVID-19 pandemic in South Africa: efforts and challenges at hand. J Med Virol. 2021. https://doi.org/10.1002/jmv.27124 .
Article PubMed PubMed Central Google Scholar
Kang K, Hu Y, Wu S, Shi S. Comparative Metagenomic Analysis of Chicken Gut Microbial Community, function, and Resistome to Evaluate Noninvasive and Cecal Sampling resources. Animals. 2021;11(6):1718. https://doi.org/10.3390/ani11061718 .
Chang, Wei-Shan, Eden J-S, Hall J, Shi M, Rose K, et al. Metatranscriptomic analysis of virus diversity in urban wild birds with paretic disease. J Virol. 2020;94(18):e00606–20. https://doi.org/10.1128/jvi.00606-20 .
Article CAS PubMed Google Scholar
Vibin J, Chamings A, Klaassen M, Alexandersen S. Metagenomic characterisation of additional and novel avian viruses from Australian wild ducks. Sci Rep. 2020;10(1):1–14. https://doi.org/10.1038/s41598-020-79413-9 .
Article CAS Google Scholar
Wille M, Shi M, Hurt AC, Klaassen M, Holmes EC. RNA virome abundance and diversity is associated with host age in a bird species. Virology. 2021;561:98–106. https://doi.org/10.1016/j.virol.2021.06.007 .
Lima DA, Cibulski SP, Tochetto C, Varela APM, Finkler F, Teixeira TF, et al. The intestinal virome of malabsorption syndrome-affected and unaffected broilers through shotgun metagenomics. Virus Res. 2019;261:9–20. https://doi.org/10.1016/j.virusres.2018.12.005 .
Mulholland KA, Robinson MG, Keeler SJ, Johnson TJ, Weber BW, Keeler CL Jr. Metagenomic analysis of the respiratory microbiome of a broiler flock from hatching to processing. Microorganisms. 2021;9(4):721. https://doi.org/10.3390/microorganisms9040721 .
François S, Pybus OG. Towards an understanding of the avian virome. J Gen Virol. 2020;101(8):785. https://doi.org/10.1099/jgv.0.001447 .
Ishag HZA, Terab AMA, El Tigani-Asil ETA, Bensalah OK, Khalil NAH, Khalafalla AI, et al. Pathology and Molecular Epidemiology of Fowl Adenovirus Serotype 4 outbreaks in Broiler Chicken in Abu Dhabi Emirate, UAE. Veterinary Sci. 2022;9(4):154. https://doi.org/10.3390/vetsci9040154 .
Lebdah M, Alshaya DS, Jalal AS, Mousa MR, Radwan MM, Samir M, et al. Molecular characterization of aviadenovirus serotypes and pathogenicity of the identified adenovirus in broiler chickens. Poult Sci. 2022;101918. https://doi.org/10.1016/j.psj.2022.101918 .
Pradeep M, Reddy M, Kannaki T. Molecular identification and characterization of chicken parvovirus from Indian Chicken and Association with Runting and Stunting Syndrome. Indian J Anim Res. 2020;54(12):1517–24. https://doi.org/10.18805/ijar.B-3890 .
Cui H, Pan S, Xu X, Ji J, Ma K, Yao L, et al. Molecular characteristics of novel chaphamaparvovirus identified in chickens. Poult Sci. 2022;102449. https://doi.org/10.1016/j.psj.2022.102449 .
King AM, Lefkowitz EJ, Mushegian AR, Adams MJ, Dutilh BE, Gorbalenya AE, the International Committee on Taxonomy of Viruses. Changes to taxonomy and the International Code of Virus Classification and Nomenclature ratified by (2018). Archives of virology. 2018;163(9):2601-31. https://doi.org/10.1007/s00705-018-3847-1 .
Zhang Y-Z, Chen Y-M, Wang W, Qin X-C, Holmes EC. Expanding the RNA virosphere by unbiased metagenomics. Annual Rev Virol. 2019;6:119–39. https://doi.org/10.1146/annurev-virology-092818-015851 .
Naguib MM, Verhagen JH, Mostafa A, Wille M, Li R, Graaf A, et al. Global patterns of avian influenza A (H7): virus evolution and zoonotic threats. FEMS Microbiol Rev. 2019;43(6):608–21. https://doi.org/10.1128/jvi.01300-17 .
Peacock TP, Penrice-Randal R, Hiscox JA, Barclay WS. SARS-CoV-2 one year on: evidence for ongoing viral adaptation. J Gen Virol. 2021;102(4):001584. https://doi.org/10.1099/jgv.0.001584 .
Rajak H, Jain DK, Singh A, Sharma AK, Dixit A. Ebola virus disease: past, present and future. Asian Pac J Trop Biomed. 2015;5(5):337–43. https://doi.org/10.1016/S2221-1691(15)30365-8 .
Ghosh D. Historical Perspectives: Asian Flu vs. Coronavirus-Different Times and Similar Problems. Available at SSRN 3699881. 2020. https://www.easternherald.com/2020/05/14/history-asian-flu-vs-coronavirus/ .
Cunha CB, Opal SM. Middle East respiratory syndrome (MERS) a new zoonotic viral pneumonia. Virulence. 2014;5(6):650–4. https://doi.org/10.4161/viru.32077 .
Theuns S, Desmarets LM, Heylen E, Zeller M, Dedeurwaerder A, Roukaerts ID, et al. Porcine group a rotaviruses with heterogeneous VP7 and VP4 genotype combinations can be found together with enteric bacteria on Belgian swine farms. Vet Microbiol. 2014;172(1–2):23–34. https://doi.org/10.1016/j.vetmic.2014.04.002 .
Conceição-Neto N, Zeller M, Lefrère H, De Bruyn P, Beller L, Deboutte W, et al. Modular approach to customise sample preparation procedures for viral metagenomics: a reproducible protocol for virome analysis. Sci Rep. 2015;5(1):1–14. https://doi.org/10.1038/srep16532 .
Andreou I, Storbeck M, Hahn P, Rulli S, Lader E. Optimized Workflow for Whole Genome and Transcriptome Next-Generation sequencing of single cells or limited nucleic acid samples. Curr Protocols. 2023;3(5):e753. https://doi.org/10.1002/cpz1.753 .
Vilsker M, Moosa Y, Nooij S, Fonseca V, Ghysens Y, Dumon K, et al. Genome detective: an automated system for virus identification from high-throughput sequencing data. Bioinformatics. 2019;35(5):871–3. https://doi.org/10.1093/bioinformatics/bty695 .
Bolger AM, Lohse M, Usadel B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics. 2014;30(15):2114–20. https://doi.org/10.1093/bioinformatics/btu170 .
Brown J, Pirrung M, McCue LA. FQC Dashboard: integrates FastQC results into a web-based, interactive, and extensible FASTQ quality control tool. Bioinformatics. 2017;33(19):3137–9. https://doi.org/10.1093/bioinformatics/btx373 .
Buchfink B, Xie C, Huson DH. Fast and sensitive protein alignment using DIAMOND. Nat Methods. 2015;12(1):59–60. https://doi.org/10.1038/nmeth.3176 .
Bankevich A, Nurk S, Antipov D, Gurevich AA, Dvorkin M, Kulikov AS, et al. SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J Comput Biol. 2012;19(5):455–77. https://doi.org/10.1089/cmb.2012.0021 .
Deforche K. An alignment method for nucleic acid sequences against annotated genomes. BioRxiv. 2017;200394. https://doi.org/10.1101/200394 .
Asplund M, Kjartansdóttir KR, Mollerup S, Vinner L, Fridholm H, Herrera JA, et al. Contaminating viral sequences in high-throughput sequencing viromics: a linkage study of 700 sequencing libraries. Clin Microbiol Infect. 2019;25(10):1277–85. https://doi.org/10.1016/j.cmi.2019.04.028 .
Porter AF, Cobbin J, Li C-X, Eden J-S, Holmes EC. Metagenomic identification of viral sequences in laboratory reagents. Viruses. 2021;13(11):2122. https://doi.org/10.3390/v13112122 .
Lou YC, Hoff J, Olm MR, West-Roberts J, Diamond S, Firek BA, et al. Using strain-resolved analysis to identify contamination in metagenomics data. Microbiome. 2023;11(1):36. https://doi.org/10.1186/s40168-023-01477-2 .
Bray JR, Curtis JT. An ordination of the upland forest communities of southern Wisconsin. Ecol Monogr. 1957;27(4):326–49. https://doi.org/10.2307/1942268 .
Real R, Vargas JM. The probabilistic basis of Jaccard’s index of similarity. Syst Biol. 1996;45(3):380–5. https://www.researchgate.net/profile/RaimundoReal/publication/239604848_The_Probabilistic_Basis_of_Jaccard’s_Index_of_Similarity/links/0c9605268d8ff04ab1000000/The-Probabilistic-Basis-of-Jaccards-Index-of-Similarity.pdf .
Verma V, Aggarwal RK. A comparative analysis of similarity measures akin to the Jaccard index in collaborative recommendations: empirical and theoretical perspective. Social Netw Anal Min. 2020;10:1–16. https://doi.org/10.1007/s13278-020-00660-9 .
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’hara R et al. Package ‘vegan’. Community ecology package, version. 2013;2(9):1-295. https://www.researchgate.net/publication/282247686_Vegan_Community_Ecology_Package_R_package_version_20-2 .
Wickham H, Chang W, Wickham MH. Package ‘ggplot2’. Create Elegant data Visualisations Using Gramm Graphics Version. 2016;2(1):1–189. http://ggplot2.org/ .
Google Scholar
Wilke CO, Wickham H, Wilke MCO. Package ‘cowplot’. Streamlined plot theme and plot annotations for ‘ggplot2. 2019. https://wilkelab.org/cowplot/ .
Kolde R, Kolde MR. Package ‘pheatmap’. R Package. 2018;1(10). https://rdrr.io/cran/pheatmap/ .
Wickham H, Wickham MH. Package ‘tidyverse’. See http://tidyverse tidyverse org. 2019:1–5. https://doi.org/10.21105/joss.01686 .
Edgar RC. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004;32(5):1792–7. https://doi.org/10.1093/nar/gkh340 .
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. Mol Biol Evol. 2013;30(12):2725–9. https://doi.org/10.1093/molbev/msab120 . MEGA6: molecular evolutionary genetics analysis version 6.0.
Rambaut A. FigTree v1. 3.1. 2009. http://tree.bio.ed.ac.uk/software/figtree/ .
Devaney R, Trudgett J, Trudgett A, Meharg C, Smyth V. A metagenomic comparison of endemic viruses from broiler chickens with runting-stunting syndrome and from normal birds. Avian Pathol. 2016;45(6):616–29. https://doi.org/10.1080/03079457.2016.1193123 .
Kwok KT, de Rooij MM, Messink AB, Wouters IM, Smit LA, Cotten M, et al. Establishing farm dust as a useful viral metagenomic surveillance matrix. Sci Rep. 2022;12(1):16308. https://doi.org/10.1038/s41598-022-20701-x .
Mason AS, Lund AR, Hocking PM, Fulton JE, Burt DW. Identification and characterisation of endogenous avian leukosis virus subgroup E (ALVE) insertions in chicken whole genome sequencing data. Mob DNA. 2020;11(1):1–13. https://doi.org/10.1186/s13100-020-00216-w .
Chen, Li H-W, Cong F, Lian Y-X. Avian leukosis virus subgroup J infection alters viral composition in the chicken gut. FEMS Microbiol Lett. 2021;368(10):fnab058. https://doi.org/10.1111/lam.13617 .
Kubacki J, Qi W, Fraefel C. Differential viral genome diversity of healthy and RSS-Affected broiler flocks. Microorganisms. 2022;10(6):1092. https://doi.org/10.3390/microorganisms10061092 .
Rajeoni AH, Ghalyanchilangeroudi A, Khalesi B, Madadi MS, Hosseini H. The tracheal virome of broiler chickens with respiratory disease complex in Iran: the metagenomics study. Iran J Microbiol. 2021;13(3):337. https://doi.org/10.18502%2Fijm.v13i3.6396.
PubMed PubMed Central Google Scholar
de Oliveira LB, Stanton JB, Zhang J, Brown C, Butt SL, Dimitrov K, et al. Runting and stunting syndrome in broiler chickens: histopathology and association with a novel picornavirus. Vet Pathol. 2021;58(1):123–35. https://doi.org/10.1177/0300985820969971 .
Yan T, Guo L, Jiang X, Wang H, Yao Z, Zhu S, et al. Discovery of a novel recombinant avian orthoreovirus in China. Vet Microbiol. 2021;260:109094. https://doi.org/10.1016/j.vetmic.2021.109094 .
Jiang X, Yao Z, He D, Wu B, Wei F, Li G, et al. Genetic and pathogenic characteristics of two novel/recombinant avian orthoreovirus. Vet Microbiol. 2022;275:109601. https://doi.org/10.1016/j.vetmic.2022.109601 .
Fan L, Wu T, Hussain A, Gao Y, Zeng X, Wang Y, et al. Novel variant strains of infectious bursal disease virus isolated in China. Vet Microbiol. 2019;230:212–20. https://doi.org/10.1016/j.vetmic.2019.01.023 .
Article PubMed Google Scholar
Lian J, Wang Z, Xu Z, Pang Y, Leng M, Tang S, et al. Pathogenicity and molecular characterization of infectious bursal disease virus in China. Poult Sci. 2022;101(1):101502. https://doi.org/10.1016/j.psj.2021.101502 .
Van Borm S, Steensels M, Mathijs E, Vandenbussche F, van den Berg T, Lambrecht B. Metagenomic sequencing determines complete infectious bronchitis virus (avian Gammacoronavirus) vaccine strain genomes and associated viromes in chicken clinical samples. Virus Genes. 2021;57(6):529–40. https://doi.org/10.1007/s11262-021-01872-7 .
Yin L, Zhou Q, Huang J, Mai K, Yan Z, Wei X, et al. Characterization and pathogenicity of a novel avian nephritis virus isolated in China. Avian Pathol. 2022;51(1):87–96. https://doi.org/10.1186/s40168-022-01348-2 .
Hassan MS, Najimudeen SM, Ali A, Altakrouni D, Goldsmith D, Coffin CS, et al. Immunopathogenesis of the Canadian Delmarva (DMV/1639) infectious bronchitis virus (IBV): impact on the reproductive tract in layers. Microb Pathog. 2022;166:105513. https://doi.org/10.1016/j.micpath.2022.105513 .
Matos M, Bilic I, Tvarogová J, Palmieri N, Furmanek D, Gotowiecka M, et al. A novel genotype of avian hepatitis E virus identified in chickens and common pheasants (Phasianus colchicus), extending its host range. Sci Rep. 2022;12(1):21743. https://doi.org/10.1038/s41598-022-26103-3 .
Park WJ, Park BJ, Ahn HS, Lee JB, Park SY, Song CS, et al. Hepatitis E virus as an emerging zoonotic pathogen. J Vet Sci. 2016;17(1):1–11. https://doi.org/10.4142/jvs.2016.17.1.1 .
Nan Y, Wu C, Zhao Q, Zhou E-M. Zoonotic hepatitis E virus: an ignored risk for public health. Front Microbiol. 2017;8:2396. https://doi.org/10.3389/fmicb.2017.02396 .
Osamudiamen FT, Akanbi OA, Zander S, Oluwayelu DO, Bock C-T, Klink P. Identification of a putative Novel genotype of Avian Hepatitis E Virus from apparently healthy chickens in Southwestern Nigeria. Viruses. 2021;13(6):954. https://doi.org/10.3390/v13060954 .
Zhang Y, Zhao H, Chi Z, Cui Z, Chang S, Wang Y, et al. Isolation, identification and genome analysis of an avian hepatitis E virus from white-feathered broilers in China. Poult Sci. 2022;101(3):101633. https://doi.org/10.1016/j.psj.2021.101633 .
Matczuk AK, Ćwiek K, Wieliczko A. Avian hepatitis E virus is widespread among chickens in Poland and belongs to genotype 2. Arch Virol. 2019;164(2):595–9. https://doi.org/10.1007/s00705-018-4089-y .
Sun Z, Larsen C, Dunlop A, Huang F, Pierson F, Toth T, et al. Genetic identification of avian hepatitis E virus (HEV) from healthy chicken flocks and characterization of the capsid gene of 14 avian HEV isolates from chickens with hepatitis–splenomegaly syndrome in different geographical regions of the United States. J Gen Virol. 2004;85(3):693–700. https://doi.org/10.1099/vir.0.19582-0 .
Cheng Z, Li M, Wang Y, Chai T, Cai Y, Li N. Pathogenicity and immune responses of aspergillus fumigatus infection in chickens. Front Veterinary Sci. 2020;7:143. https://doi.org/10.3389/fvets.2020.00143 .
Lofgren LA, Lorch JM, Cramer RA, Blehert DS, Berlowski-Zier BM, Winzeler ME et al. Avian-associated aspergillus fumigatus displays broad phylogenetic distribution, no evidence for host specificity, and multiple genotypes within epizootic events. G3 (Bethesda). 2022;12(5). https://doi.org/10.1093/g3journal/jkac075 .
Elahi U, Xu C-c, Wang J, Lin J, Wu S-g, Zhang H-j, et al. Insect meal as a feed ingredient for poultry. Anim Bioscience. 2022;35(2):332. https://doi.org/10.5713/ab.21.0435 .
Wille M, Harvey E, Shi M, Gonzalez-Acuña D, Holmes EC, Hurt AC. Sustained RNA virome diversity in Antarctic penguins and their ticks. ISME J. 2020;14(7):1768–82. https://doi.org/10.1038/s41396-020-0643-1 .
Knox MA, Gedye KR, Hayman DT. The challenges of analysing highly diverse picobirnavirus sequence data. Viruses. 2018;10(12):685. https://doi.org/10.3390/v10120685 .
Boros Á, Polgár B, Pankovics P, Fenyvesi H, Engelmann P, Phan TG, et al. Multiple divergent picobirnaviruses with functional prokaryotic Shine-Dalgarno ribosome binding sites present in cloacal sample of a diarrheic chicken. Virology. 2018;525:62–72. https://doi.org/10.1016/j.virol.2018.09.008 .
Krishnamurthy SR, Wang D. Extensive conservation of prokaryotic ribosomal binding sites in known and novel picobirnaviruses. Virology. 2018;516:108–14. https://doi.org/10.1016/j.virol.2018.01.006 .
Yinda CK, Ghogomu SM, Conceição-Neto N, Beller L, Deboutte W, Vanhulle E, et al. Cameroonian fruit bats harbor divergent viruses, including rotavirus H, bastroviruses, and picobirnaviruses using an alternative genetic code. Virus Evol. 2018;4(1):vey008. https://doi.org/10.1093/ve/vey008 .
Kleymann A, Becker AA, Malik YS, Kobayashi N, Ghosh S. Detection and molecular characterization of picobirnaviruses (PBVs) in the mongoose: identification of a novel PBV using an alternative genetic code. Viruses. 2020;12(1):99. https://doi.org/10.3390/v12010099 .
Chauhan RP, San JE, Gordon ML. Metagenomic analysis of RNA fraction reveals the diversity of swine oral virome on South African Backyard Swine Farms in the uMgungundlovu District of KwaZulu-Natal Province. Pathogens. 2022;11(8). https://doi.org/10.3390/pathogens11080927 .
Ogunbayo A, Mogotsi MT, Sondlane H, Nkwadipo KR, Sabiu S, et al. Metagenomic analysis of respiratory RNA virome of children with and without severe Acute Respiratory infection from the Free State, South Africa during COVID-19 pandemic reveals higher diversity and abundance in summer compared with Winter Period. Viruses. 2022;14(11):2516. https://doi.org/10.3390/v14112516 .
Vibin J, Chamings A, Klaassen M, Bhatta TR, Alexandersen S. Metagenomic characterisation of avian parvoviruses and picornaviruses from Australian wild ducks. Sci Rep. 2020;10(1):12800. https://doi.org/10.1038/s41598-020-69557-z .
Lambrecht B, Marché S, Houdart P, Van Den Berg T, Vangeluwe D. Impact of age, season, and flowing vs. stagnant water habitat on avian influenza prevalence in mute swan (Cygnus olor) in Belgium. Avian Dis. 2016;60(1s):322–8. https://doi.org/10.1637/11132-050815-Reg .
Hill SC, François S, Thézé J, Smith AL, Simmonds P, Perrins CM, et al. Impact of host age on viral and bacterial communities in a waterbird population. ISME J. 2023;17(2):215–26. https://doi.org/10.1038/s41396-022-01334-4 .
Cui Y, Wang Q, Liu S, Sun R, Zhou Y, Li Y. Age-related variations in intestinal microflora of free-range and caged hens. Front Microbiol. 2017;8. https://doi.org/10.3389/fmicb.2017.01310 . :1310.DOI.
Ngunjiri J, Taylor K, Abundo ME, Jang H, Elaish M, Kc M, et al. Farm Stage, Bird Age, and body site dominantly affect the quantity, taxonomic composition, and Dynamics of respiratory and gut microbiota of commercial layer chickens. Appl Environ Microbiol. 2019;85. https://doi.org/10.1128/AEM.03137-18 .
Download references
Acknowledgements
The financial assistance of the Directorate of Research and Postgraduate Support, Durban University of Technology, for the Master’s Scholarship Scheme, awarded to Ms. Vivian C Nwokorogu is duly acknowledged. We would like to thank Dr. AE Ogunbayo and Mr. MT Mogotsi of the University of the Free State Next-Generation Sequencing Unit (UFS-NGS-Unit), Bloemfontein, South Africa for their technical assistance.
The Authors specially acknowledge the financial support of the Directorate of Research and Postgraduate Support, Durban University of Technology, and the National Research Foundation (NRF) Research Development Grant for Rated Researchers (Grant number 120433) awarded to S. Sabiu, as well as the Bill and Melinda Gates Foundation (Grant number INV-046917) awarded to M. Nyaga. All views expressed in this article are solely those of the authors and not those of their affiliated organizations, or their funders.
Author information
Authors and affiliations.
Department of Biotechnology and Food Science, Durban University of Technology, P.O. Box 1334, Durban, 4000, South Africa
Vivian C. Nwokorogu, Santhosh Pillai, Charlene Pillay & Saheed Sabiu
Next Generation Sequencing Unit, Division of Virology, Faculty of Health Sciences, University of the Free State, P.O. Box 339, Bloemfontein, 9300, South Africa
Martin M. Nyaga
Nelson Mandela School of Medicine, KwaZulu-Natal Research Innovation and Sequencing platform unit, University of KwaZulu- Natal, 719 Umbilo Road, Durban, 4001, South Africa
James E. San
You can also search for this author in PubMed Google Scholar
Contributions
Project conceptualization, administration and funds acquisition were carried out by S.S. Sample collection was performed by C.P and V.C.N. Laboratory investigation was carried out by V.C.N. Data analysis, interpretation and presentation was performed by V.C.N, J.E.S and S.S. Supervision was done by S.S, S.P and M.M.N. Original draft was written by V.C.N. All the authors read, reviewed, and approved the final draft for submission.
Corresponding author
Correspondence to Saheed Sabiu .
Ethics declarations
Ethical approval and consent to participate.
The ethical approval of this study was obtained from the Animal Research Ethics Committee (AREC) of the University of KwaZulu-Natal with the protocol Ethics Number, AREC 012/020. In addition, in compliance with Sect. 20 of the Animal Disease Act (Act No 35 of 1984), a research permit was obtained from the South African Department of Agriculture, Land Reform and Rural Development, with the reference no 12/1/5/4 (1511AC). Furthermore, for faecal samples, a provincial no restriction permit was obtained from the KwaZulu-Natal Department of Agricultural and Rural Development. No additional permit was required by commercial farm owner where sample was obtained.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Material 1
Rights and permissions.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Nwokorogu, V.C., Pillai, S., San, J.E. et al. A metagenomic investigation of the faecal RNA virome structure of asymptomatic chickens obtained from a commercial farm in Durban, KwaZulu-Natal province, South Africa. BMC Genomics 25 , 629 (2024). https://doi.org/10.1186/s12864-024-10517-6
Download citation
Received : 13 January 2024
Accepted : 12 June 2024
Published : 24 June 2024
DOI : https://doi.org/10.1186/s12864-024-10517-6
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Viral metagenomics
- Faecal virome
- RNA viruses
- Zoonosis and
- Viral diversity
BMC Genomics
ISSN: 1471-2164
- Submission enquiries: [email protected]
- General enquiries: [email protected]
The magic of a South African summer!!!
People who enjoyed this, also enjoyed:
- New Zealand
- Middle East
- North America
- South America
- Solo Female Travel
- Polar travel – join me!
- Start Blogging
- Confessions
- Get Inspired
- This will probably get me hate mail
I loved Cape Town so much I couldn’t leave
Home » Blog » Accommodation » I loved Cape Town so much I couldn’t leave
Sharing is caring!

Somehow I missed the memo that Cape Town is the coolest city ever!
So when I stumbled off the plane from Durban, hungover as hell, I was pleasantly surprised and immediately blown away by this sprawling metropolis at the edge of the world. Holy shit Cape Town, South Africa is all kinds of pretty!
A country girl at heart, I am not usually a fan of big cities but when there are plenty of green places and designed in a way that makes me feel like I’m not in a city, I’m a fan. Cape Town luckily ticks all those boxes, and even reminded me a bit of Wellington, New Zealand where I used to live. Just with more crazy wildlife.

It spreads out all along a wild and beautiful coastline with sandy white beaches, cute little surf suburbs with the iconic Table Mountain looming above you in the background.
Downtown Cape Town is beautiful, and the harbor area is one of the nicest I’ve seen with amazing shopping OOPS. It doesn’t take a lot to understand why the most expensive real estate in Africa is here.
On my first trip over to South Africa , Cape Town was the first stop after a long conference in Durban. And trust me, I was basically sobbing when we had to leave after only 3 days. Couldn’t I just stay here the whole time?

As my time in Africa was drawing to a close, I made a hasty decision outside a market in Joburg. I wasn’t ready to leave. Have you guys ever felt that way about a place?
So I did the only sensible thing, rang the agent that booked my tickets and changed my flights giving myself an extra week and a half in Africa. First I headed out to Phinda , an amazing safari reserve, and then booked myself a ticket back to Cape Town.
Sometimes you have to listen to that inner voice when you love a place so much and can’t bear to leave. And ok, I’ll admit I’m lucky in that I had a pretty flexible schedule at the time and was able to do this. Wahoo internet jobs!

Luckily I was able to time my return to Cape Town when my friends Hecktic Travels were in town on a stopover on their way to Namibia.
Cue long dinners, cocktails in Camps Bay, road trips and photo sessions, plus lots and lots of coffee and catch ups. While Cape Town certainly has heaps of things to do, I found myself just enjoying being there, going for walks and exploring. It’s a great city just to be in, not necessarily be busy in.
Here is my little introduction to this glorious city with 10 things that helped make me fall in love with Cape Town.

1. Table Mountain
It’s no secret, I love mountains so to be a really cool and fun hip city with an amazing mountain backdrop, is definitely the best of both worlds. With straight cliffs and a long flat summit, it didn’t take long to understand its name.
We headed up first thing in the morning back through the winding streets leading to the base of the cableway, the easiest way to get to the top. There was beautiful mist layering over the town giving a great ephemeral feel to the start of the day.
As we walked out on top, I promptly lost my shit when I spotted a dassie for the first time. A fat little groundhog looking animal, it is adorable and I spent the next 20 minutes taking photos of them instead of admiring the view. Typical.

2. Dinner with a local family
For me, this was probably one of my favorite parts of our time in Cape Town.
Hosted dinners with local families is now a popular way to get to know the city. When you are visiting a place for only a few days, it’s often quite hard to find opportunities to get to know the locals and get a glimpse into what life is really like there.
We were kindly invited to the home of a local musician who not only treated us to an amazing traditional dinner with his wife, but also rocked out with us all evening to the most beautiful music.

3. Staying at the One & Only Cape Town
It’s not every day that I get to stay in a 6 star hotel! Holy moley! If they only knew what I was really like, would they have let me in?
The One & Only Cape Town is part of the One & Only resort chain, and their South Africa property doesn’t fail to impress.
The biggest coziest bed ever complete with a pillow menu, great internet, a huge bathtub and an espresso machine – what more could I want?

I pretty much live in hotels with my job, and getting to stay in places like this is definitely a perk that comes with being a professional travel blogger.
At the risk of sounding snobby, while it was an amazing property, I didn’t feel like 6 stars to me until the first breakfast when I discovered not only champagne and oysters, order off the menu, any coffee you want, and oh, the crispy bacon or grilled bacon station.
Yes, 6 stars or sure!

4. Penguins on the beach!
My oh my! You guys know how much of a #BirdNerd I am, so when I first started researching Cape Town and realized there was a beach nearby home to a colony of penguins, I knew I had to make it happen!
Boulders Beach is near Simon’s Town is home to a colony of Africa Penguins, and they are adorable!

And luck was on my side because not only was the beautiful beach covered in penguins, there were fluffy molting babies everywhere! There is a god and he heard my prayers!
Show me the penguin babies!
I’m not going to lie, I loved it so much I went twice!

All the amazing seafood, all the time, nuff said.

6. Saturday Market
Another reason I was so happy to come back to Cape Town was that it gave me the opportunity to meet up with a few of you guys. I got an email from a kiwi expat living over in Cape Town who had spent a lot of time in Wanaka so it was a great opportunity to see the side of the city I probably wouldn’t have known about and hang out with a local.
The Oranjezicht City Farmer’s Market is super cute and in a beautiful part of town. Even though it drizzled the whole time, there were tents everywhere and so much delicious food and coffee, you hardly noticed.
This market has an amazing story – it was meant to be shut down before it was saved at the last minute and now takes place on the premier’s own gardens in Cape Town, which means its setting couldn’t be more picturesque.

7. Cape of Good Hope
I met up with the Hecks who rented a car and we hit the road for the day. First stop – back to the penguins obviously.
From there we headed to the Cape of Good Hope to live out our elementary school geography dreams. Nothing like finally laying eyes on a place you’ve heard about your whole lives, right guys?
Did I mention there are wild ostriches there? And baboons.
Run for your lives!

8. Chapman’s Peak Drive
On the way back to the city from the Cape Peninsula in the moody late afternoon light, we decided to take in the beautiful Chapman’s Peak Drive on the west coast.
I love a good road trip with friends!
And holy crap did this road not disappoint!

The 9km of road connecting Noordhoek and Hout Bay is pretty stunning.
Picture huge cliffs dropping down to the turquoise sea and crazy bends and turns with more lookouts and views than you could ever hope for. Don’t forget your camera.
As we turned the corner to head back to Cape Town, the sun began to shine through the peaks lighting up the houses below and creating those beams of light that I am obsessed with. What a way to close the trip!

9. Sunsquare Cape Town
I also wanted to mention on my second trip back to Cape Town I had to forgo the 6 star hotel for something more in my range and budget, the Sunsquare Cape Town .
Near the Gardens area of Cape Town, it’s in a really cool part of town with an amazing breakfast.

10. Sunset cruise
The perfect introduction to Cape Town is a sunset cruise in the harbor accompanied by a few breaching whales and champagne.
Sigh, Cape Town, you rock!
Have you ever felt this way about a place? Ever been to Cape Town? Ever changed your flights so that you didn’t have to leave a place?

Many thanks to V isit South Africa and Cape Town Tourism for hosting me in Cape Town. Like always, I’m keeping it real – all opinions are my own, like you could expect anything less from me.
About the author.
85 Comments on “ I loved Cape Town so much I couldn’t leave ”
Leave a reply cancel reply.
Your email address will not be published. Required fields are marked *
So nice, your Pictures made me want to go there RIGHT now ! 😉
Yay love that!
I am single
I heard so many great things about South Africa and Cape Town and dinner with a local family is definitely something I want to try one day! The penguins are so adorable 😀
Such a good way to experience the place!
As always, fab post! I love the idea of joining a dinner with locals – looked fun and a great way to mix with the locals! 🙂
Also…how on earth did you manage to get the cutest penguin pics?!
haha I made everyone wait while I took about 1000 shots of them haha
Love the photos! Cape Town is a magical city, and I’m in the same boat… not a city person either. I spent two months in Africa last year, visiting several places and I had a really hard time leaving at the end.
It’s magical, isn’t it?
Related Adventures

Emails Suck. Mine Don't.

We think you are located in Russian Federation . Is this correct?
- Yes, I reside in Russian Federation
- Change country/curriculum
We use this information to present the correct curriculum and to personalise content to better meet the needs of our users.
8.3 Biomes (ESG9X)
In this section learners will focus on summarising the importance of terrestrial and aquatic biomes of Southern Africa. They will study how climate, soil and vegetation influence the organisms found in each. The location of the different biomes in South Africa will also be introduced.
The biosphere is divided up into a number of biomes . Biomes are regions with similar climate and geography. The key factors determining climate are average annual precipitation (rainfall) and temperature. These factors, in turn, depend on the geography of the region, such as the latitude and altitude of the region, and mountainous barriers. The specific conditions of biomes determine the plant and animal life found within them. The communities of plants, animals and soil organisms in a particular biome are collectively referred to as an ecosystem . Biomes can be aquatic or terrestrial .
Aquatic biomes (ESG9Y)
Water covers a major portion of the Earth's surface, so aquatic biomes contain a rich diversity of plants and animals. Aquatic biomes are divided into two main groups depending on the amount of salt present in the water: freshwater and marine biomes.
1. Freshwater
Freshwater biomes are defined by their low salt concentration, which is usually less than \(\text{1}\%\). Examples include: ponds, lakes, streams, rivers and wetlands.
2. Marine biomes
Marine bodies are salty, having approximately 35 grams of dissolved salt per litre of water (\(\text{3,5}\%\)). Marine biomes are divided into oceans, coral reefs and estuaries. The vegetation of the marine biomes consists of the different types of algae, which is one of the major sources of oxygen in the world. Green algae also play a role in the removal of carbon dioxide from the atmosphere.
Oceans : are very large marine bodies that dominate the Earth's surface and hold the largest ecosystems. The open ocean or sea covers nearly three-quarters of the earth's surface and contains a rich diversity of living organisms. Examples of animals in the ocean biome include whales, sharks, octopuses, perlemoen, crabs and crayfish. Figure 8.2 shows a typical ocean ecosystem.

Figure 8.2: Ocean ecosystem.
Coral reefs : are found in the warm, clear, shallow waters of tropical oceans around islands or along continental coastlines. Coral reefs are mostly formed underwater from calcium carbonate produced by living coral. Reefs provide food and shelter for other organisms and protect shorelines from erosion. South Africa has only one coral reef in the subtropical ocean waters north of Lake St. Lucia in northern KwaZulu Natal. Figure 8.3 shows a typical coral reef system.

Figure 8.3: Coral reef.
Estuaries : are partially enclosed areas of fresh water and silt from streams or rivers, which mix with salty ocean water. Estuaries represent a transition from land to sea and from freshwater to saltwater. Estuaries are biologically very productive areas and provide homes for a wide variety of plants, birds and animals. Figure 8.4 shows an example of an estuary system.

Figure 8.4: Knysna Estuary.
Marine biomes of South Africa
South Africa's long coastline stretches for over 3000 kilometres, from Namibia in the West to Mozambique in the East. There are a few key features to note about South Africa's coastline and marine biomes. South Africa's coastline is rugged, as rocky shores are exposed to high wave energy and the coastline generally experiences high wind for most of the year. There are up to 343 estuaries found along the coast, two thirds of which are found on East Coast between Cape Padrone in the Eastern Cape Province and Mtunzini in KwaZulu-Natal. The Eastern coastline receives the highest rainfall, mostly during summer.
South Africa's East Coast has relatively warm waters (20-25 degrees C), the West Coast receives colder Atlantic waters (9-14 degrees C), and the South Coast experiences intermediate water temperatures (16-21 degrees C). The cold Benguela Upwelling System on the South-West coast supports large numbers of marine animals. The warm Agulhas current off the East Coast has a smaller quantity of fish but a greater diversity of species. Abundant opportunities exist for tourism, recreation, food, export and associated economic development.
Terrestrial biomes (ESG9Z)
Terrestrial biomes occur on land and can be of many types. Examples include: thicket, tundra, forest, grassland and desert. Terrestrial biomes are usually classified based on the dominant vegetation, climate or geographic location. The location and characteristics of the various biomes is mostly influenced by climatic conditions such as rainfall and temperature.
South African Biomes (ESGB2)
The most recent classification of the terrestrial biomes in South Africa divides the region into the following eight biomes:
- Succulent Karoo
The biome regions in South Africa are mapped in Figure 8.5 .

Figure 8.5: Biomes of South Africa.
1. Grassland Biome
Location : grasslands are found on the Highveld.
Climate : they typically have summer rainfall of 400 mm to 2000 mm. Winters are cold, and frost can occur.
Soil and geography : in grasslands, the soil is red/yellow/grey or red/black clay. Grassland soil has rich fertile upper layers.
Flora : vegetation is mainly grass, but trees can grow on the hills and along river beds.
Fauna : many types of grass-eating herbivores can be found in this habitat, such as black wildebeest, blesbok and eland. Rodents are also common in grasslands which makes this biome an ideal hunting ground for birds of prey. The diverse plant species also support many plant-eating insects such as butterflies, grasshoppers, crickets and ants.

Figure 8.6: Grasslands are regions where the vegetation is dominated by grasses.

Figure 8.7: Grasslands are regions where the vegetation is dominated by grasses.
Burning of grassland
Compare and analyse the advantages and disadvantages of burning grassland.
Instructions
Using these resources, tabulate the advantages and disadvantages of burning grassland.
- Remember to cite your references correctly.
Activity: Burning of grassland
Below are a couple of advantages and disadvantages related to the burning of grassland. However, learners may also come up with different examples, and as long as they can cite their source, their answers should be accepted.
|
|
| Some types of seeds actually germinate better after a fire, as their hard seed pods crack open. | Valuable seeds may be destroyed and then they cannot germinate. |
| Species that were being crowded or overwhelmed by other species have a chance to recolonise. | Animals and plants are injured, damaged or killed. Fire kills indiscriminately and may kill endangered plant / animal species. |
| Plants that grow aggressively (weeds) are restricted. | Organisms in the soil are destroyed, humus is reduced. Loss of nitrogen and sulphur from the soil. |
| Younger plants provide better nutrition (green grass after a severe winter). Fire destroys unpalatable grass types and allows new palatable (better tasting) grass to grow out. | Grasses are weakened if burning is practised or occurs at the wrong time. |
| Blackened soil heats up quickly in the sun and speeds up the growth of new plants. | Destroys the soil cover and can lead to erosion. |
| Destroys insect pests, ticks and parasitic worms. | Fires may get out of control and burn useful farmland or endanger lives and property. |
| Phosphate in the ash acts as soil fertiliser and improves plant growth. | Air pollution and smoke may irritate the lungs of people and animals. |
2. Savannah biome
Location : the Savannah biome is the largest biome in Southern Africa. It is found mainly in the western parts of Limpopo, the northern parts of the Northern Cape and Free State, the North West Province and KwaZulu Natal.
Climate : summers are hot and wet and the winters are cool with little or no rain. Frost occurs in winter.
Soil and geography : the soil consists of red/black clay or red/ yellow/ grey soil and is often sandy.
Flora : this biome is also known as the bushveld, where grasses are mainly found and regular fires prevent the trees from dominating. Herbaceous plants and woody plants can be found in different areas. Plants are able to withstand fire.
Fauna : big game species such as kudu and Springbok, lion, buffalo and elephant are found in the Savannah Biome. This is also a malaria-prone area.

Figure 8.8: Savannah biome.

Figure 8.9: Savannah biome.
3. Succulent Karoo biome
Location : the Succulent Karoo biome can be found along the west coast of the Northern Cape Province and the northern parts of the Western Cape Province.
Climate : this biome is hot in summer and cold in winter and the rainfall in this area is very low. Fog is common, and frost is seldom severe enough to cause damage.
Soil and geography : lime-rich, weakly developed soils, rocks and sand that is easily eroded.
Flora : forty percent of plant species found here are endemic to this biome. The Namaqualand region of this biome is famous for its colourful wild flowers. Succulent plants are able to live through dry seasons by using water stored in their leaves or stems.
Fauna : insects are common and the plants provide grazing for sheep and goats.
The word 'Karoo' comes from the Khoi word Karusa , which means dry, barren, thirstland. Karoo is an apt description for this arid region.
4. Nama Karoo
Location : the Nama Karoo is the second largest biome in South Africa. It forms the major part of the Northern Cape Province and the Free State.
Climate : it is regarded as a semi-desert area receiving very little rain. The summers are very hot and the winters are very cold and frost often occurs.
Soil and geography : soil occurring on rocks is weakly developed. The area is also characterised by sands and rocky and red clay, making erosion occur easily.
Flora : it is characterised by grassy dwarf shrub land.
Fauna : the flora provides good grazing for sheep and goats.

Figure 8.10: Nama Karoo found in the Northern Cape province.
5. Forest Biome
Trees are not only producers, but as a result of their size they also create a habitat for other species. The leaf cover of trees provides shelter for animals, while the bark and fissures in the trees also provide a habitat for insects. The leaf cover also creates a shady environment in which shade-loving, low-growing plants can flourish.
Location : the forest biome in South Africa occurs in patches, in areas such as Knysna of the Western Cape as well as KwaZulu Natal, the Eastern Cape, Limpopo and Mpumalanga.
Climate : some of these forests experience rain only in winter, while others get rainfall throughout the year.
Soil and geography : forests range in altitude from sea level to above 2000 metres, soil is drained and virtually all soil types are present.
Flora : forests are dominated by trees of which the Yellowwood is the largest. There are many herbaceous and bulbous plants that also occur.
Fauna : numerous insect species, birds ans small mammals such as bushpig, bushbuck and monkeys. The canopy is a perfect habitat for birds such as the Knysna Loeries, pigeons and eagle.

Figure 8.11: Forest biome.

Figure 8.12: Knysna Forest.
When leaves or fruit fall from the trees and collect at the feet of the trees, another series of organisms can appear. By breaking down organic material, decomposers such as microorganisms return the organic nutrients to the soil. Humus is formed in this way. Humus is dead organic material. Other creatures that live off decayed organic material, namely the detritivores, also promote this process of decomposition by breaking up dead plant matter into its component nutrients.
Poster project to illustrate the role players in a forest ecosystem
Bring pictures of animals, trees and other plants to class.
- The teacher will divide the class into groups.
Each group will prepare a poster to illustrate the mutual dependence of the trees, other plants and animals.
- Each group must present their poster to the rest of the class.
Answer the following questions / follow the instructions arising from the class discussion:
Supposing the tree on your poster was to fall over.
Which organisms would die?
Which organisms would move away?
Which organisms would increase in number?
Describe the role played by trees in an ecosystem.
Ecologically speaking, why is it bad practice to rake up leaves under trees?
Name three more examples where humans harm ecosystems.
Identify components of the ecosystem, including each trophic level. Represent this in the form of a diagram.
Project: Create a poster to illustrate the key role-players in a Forest Ecosystem
The answers to the questions will depend on the poster the learners have done. Each poster should have different answers to the questions.
If the tree falls:
The following organisms would die:
- Probably the tree itself, if it was still alive.
- Also young birds in nests in the tree.
- Anything on which the tree falls.
The following organisms would move away:
- Adult birds that were nesting or sleeping in the tree.
- Any animals that used the tree as a food source or habitat.
- Animals in the immediate vicinity of the tree (for a short while, anyway).
The following organisms would increase in number:
- Fungi and bacteria that would decompose the fallen tree.
- Ants and termites, that may use the dead tree as food source.
- Probably mosses and lichens that may grow on the surface of the tree.
- Smaller green plants in the area, as they would now get more sunlight.
The role played by trees in an ecosystem:
- Play a role in the gas balance of the atmosphere (remove \(\text{CO}_{2}\) and produce \(\text{O}_{2}\)).
- Form a habitat for many animals, e.g. squirrels, insects and birds.
- Many trees form fruits that act as a food source for animals, while leaves are eaten by herbivores. Nectar is eaten by bats, birds and insects.
- Provide shade and shelter against hail, sleet, snow and rain.
- Prevent soil erosion by holding back soil when it rains
- Thick stands of mangrove trees prevent much of the damage caused by tsunamis.
- They provide a screen against harsh winds
- Fallen leaves decompose to form compost, enriching the soil.
- Trees absorb much water and reduce surface runoff, allowing soil water sources to replenish slowly.
- There are several other learner-dependent answers.
Leaves should be left to decompose slowly, releasing nutrients into the soil. This provides fertiliser to enrich the soil with humus and recycles nutrients in nature.
Examples where humans harm ecosystems:
- Uncontrolled burning / accidental damage to ecosystems.
- Deliberate burning as a form of deforestation.
- Pollution in all its forms, including litter.
- Noise due to people, machines and vehicles unsettles animals.
- Planting invasive alien plants / not removing them from ecosystems destroys the natural species balance of an area.
- Monoculture removes biodiversity, e.g. on farms / golf estates
Learner dependent answer. This may be shown as a food pyramid.
The Fynbos contains approximately \(\text{75}\%\) of South Africa's rare and threatened plants.
Location : fynbos is the natural shrub found in the Western Cape of South Africa.
Climate : characterised by cold, wet winters and hot, dry summers (Mediterranean climate conditions).
Soil and geography : poor, acid and coarse-grained soil.
Flora : fynbos is widely known for its widespread biodiversity. Important plant types found in the fynbos include proteas, 'silver trees' and 'pincushions'. Plants growing here do not lose their leaves. Proteas have striking flowers. It has the highest fynbos variety in the world, with over 9000 species of fynbos found here.
Fauna : fynbos is home to many bird species, insects and small mammals.

Figure 8.13: Mountain Fynbos found in Western Cape.
The flora of the fynbos has a high degree of endemism . This is the ecological state of being specific to a geographic location such as an island, country or in this case, a defined biome such as the fynbos.
Fire is a necessary stage in the life-cycle of nearly all fynbos plants, and is common during the dry summer months. Many of the seeds germinate only after the intense heat of a fire. As proteas 'prepare' for the fire, they retain their seeds on the bush for at least a year, a habit known as serotiny .

The lowlands of the fynbos have been developed for agriculture and wine farming. Due to this, various species of fynbos have been threatened. For this reason, the fynbos region must be protected and preserved. It is a major tourist destination.
Discovering fynbos in South Africa
The astonishing richness and diversity of the Western Cape's natural resources is matched only by the resourcefulness and diversity of its many people. Historical patterns of unsustainable use of resources have led to the Cape Floristic Region (CFR) being listed as one of the world's threatened bioregions, and the scars are deeply etched in the land and its people.
Western Cape residents are exploring new and sustainable ways to value and benefit from these globally important assets.
South Africa's Cape Floristic region is legendary, and the unique nature of the fynbos biome has been celebrated by biologists, conservationists, development experts, and ecologist worldwide.
(Adapted from speech by Tasneem Essop the Western Cape Provincial Minister for Environment, Planning and Economic Development)
Write an essay on the fynbos biome and discuss the following aspects:
- What is the meaning of the term “fynbos”?
- Identify features of families/ indicator species that make up this vegetation type.
- Describe its ecological role in the environment.
- Describe the environmental impacts of destroying this type of vegetation.
- Describe the economical importance of fynbos for the people of the Western Cape.
- Describe management strategies involved in protecting it.
Your essay may be written or typed . Marks will be awarded for originality and own interpretation. Include a bibliography of three of more resources. No marks will be awarded for plagiarism.
Project: Learners to write an essay on the fynbos using the source information provided
Write an essay on the 'Fynbos' biome and discuss the following aspects.
Essay may be written or typed . Marks will be awarded for originality and own interpretation. Include a bibliography of three of more resources.
The exact content of such an essay cannot be specified and is up to the individual teacher. The following may serve as a guideline only:
Fynbos Biome
Fynbos is the natural shrubland or heathland vegetation occurring in a small belt of the Western Cape of South Africa, in coastal and mountainous areas with a Mediterranean climate. The soil is acidic and nutrient-poor, while the climate is marked by cold wet winters and hot dry summers. It is the smallest of the world's six floral kingdoms.
Indicator Species
Small, fine-leafed, low-growing and tough evergreen plants. Fynbos includes legumes and bulbous plants (like Watsonia spp. and chincherinchees), but the three main indicator groups are Ericas, Restios and Proteas .
Restios Restios are reed or rush-like plants. They are found in dense stands in areas of poor drainage. Being hardy, they are not grazed, but locals harvest them for thatching.
Ecological role
This biome can’t support large animals due to lack of enough nitrogen, but the area has many smaller animals like baboons, klipspringers, grysbok, dassies, mongooses and mice. Many endemic sunbirds are found here, also highly endangered butterfly species, like those whose larvae actually eat ants and live inside the ant colonies. The geometric tortoise, the world’s second rarest tortoise, is found only here, as well as several endangered frog species.
Biological impact of destruction
Again, possible answers vary widely. They should mention the loss of biodiversity, the threat to our natural heritage, possible cures for disease in plants that have not yet been studied scientifically, the loss of ecotourism and the jobs / income associated with this, the fact that extinction is forever and cannot be reversed, etc.
Economic impacts of this area for people
- Products such as rooibos tea and honeybush tea are grown here.
- Buchu plants provide oil for medicines and perfume.
- Many fynbos flowers are exported in fresh / dry form, as they last so long.
- Area provides recreational and relaxation opportunities.
- Huge numbers of research opportunities exist in the fynbos area.
- The natural beauty of the area cannot be overemphasised.
- There are several others – accept anything relevant.
Management strategy and Protection
- Removal of alien plants like pine trees, prickly pears, Australian blue gums and Acacia species.
- No developments like roads / power lines may happen here without approval.
- No agriculture will be allowed.
- No picking of flowers or removal of any plant parts.
- Encourage ecotourism to generate income and create jobs.
- Discourage the sale of curios, like shells of geometric tortoises for key rings.
- Several other relevant points may be accepted.
Below is a list of other miscellaneous facts that students may choose to include in their essays:
- Fynbos, meaning "fine bush", is a unique and strikingly beautiful group of flora endemic to a small section of the Western Cape of South Africa.
- Fynbos grows in a 100-to-200-km-wide coastal belt stretching from Clanwilliam on the West coast to Port Elizabeth on the Southeast coast. It forms part of the Cape floral kingdom, where it accounts for half of the surface area and \(\text{80}\%\) of the plant varieties.
- The fynbos in the western regions is richer and more varied than in the eastern regions of South Africa.
- Of the world's six floral kingdoms, this is the smallest and richest per area unit. The diversity of fynbos plants is extremely high, with over 9000 species of plants occurring in the area, around 6200 of which are endemic, i.e. they do not grow anywhere else in the world.
- Soil is made of rock and sandstone.
- Fire is required for seed germination and is also important to clear accumulated growth.
- Ants are important for seed dispersal and birds assist in pollination.
- Other animals found in the fynbos biome are the cape golden mole, geometric tortoise and ostrich.
No marks should be awarded for plagiarism.
Location : the thicket biome occurs along the coasts of KwaZulu Natal and the Eastern Cape.
Climate : thickets develop in areas where the rainfall is fairly high; however, there may be dry periods that prevent the vegetation from developing into forests.
Soil and geography : most thickets occur in river valleys.
Flora : the vegetation of this biome includes short trees, low intertwining shrubs and vines. There are no distinct layers of trees and shrubs, with many large open spaces found in the thicket biome. Thickets in the Eastern Cape are comprised of dense impenetrable vegetation dominated by spiny, often succulent trees and shrubs.
Fauna : examples of fauna found in thicket include kudu, monkey, bushbuck and elephant.

8. Desert Biome
Location : the Desert Biome is found largely in the Namib Desert along the coast of Namibia. The transition regions between deserts and grasslands are sometimes called semi-arid deserts.
Climate : deserts are dry areas where evaporation usually exceeds precipitation. Rainfall is low, less than 25 centimetres per year, and can be highly variable and seasonal. The low humidity results in temperature extremes between day and night. Deserts can be hot or cold. Hot deserts (e.g. the Namib and Kalahari) are very hot in the summer and have relatively high temperatures throughout the year and have seasonal rainfall. This combination of low rainfall and high temperatures keeps the air very dry, increasing its evaporating power.
Soil and geography : the soil consists mostly of sand, gravel or rocks.
Flora : deserts have relatively little vegetation.
Fauna : many insects and reptiles (lizards and snakes) occur in the desert biome.

Figure 8.15: Kalahari desert.
Did you know that most of the animals in the desert can live without water for a long time? They have adapted in many ways to do this. For instance, they can store water internally, take water out of their prey, or peck at succulents and suck out the water stored inside them.
Biomes Advertisement
Getting to know the biomes of South Africa.
- reference books
Study some advertisements for ideas.
You work for an Advertising Agency that is bidding for the account of a top travel agency. The bid includes designing a full page advert (A4) for the Getaway Magazine. Presentation, appeal and accuracy will therefore be of top priority.
The travel agency has specified that they would like the following to be included in the ad, which is geared towards people looking for a different and fascinating holiday in a specific biome :
- A region in the biome of your choice, including cities and/or towns worth a visit
- Climate (of interest to tourists)
- Well-known geographical features in the region
- Mention of some interesting wildlife (i.e. birds, animals, plants) that may be seen
- The name of the travel agency, with contact information
This is an optional activity that teachers may include if they want to. Assessment will be based on the fulfilment of the criteria given and may require the assistance of a member of the Language and /or Art department of the school, to ensure that the adverts are assessed for scientific accuracy, as well as artistic appeal and visual impact. It is recommended that a rubric be drawn up to guide assessment, such as the example given below. The teacher just ticks the relevant box and allocates the mark above that column:
Rubric to asses the biome poster
Criteria to be assessed | 3 | 2 | 1 | 0 |
Assess for impact and relevance (2) | X | |||
Clearly and accurately described (3) | ||||
Accuracy and relevance taken into account (3) | ||||
Accuracy, not overly detailed, flora AND fauna (3) | ||||
Assess colour, layout, clarity, size, relevance, captions given (6) | ||||
Clearly indicated, all details present, correct size (3) | ||||
All given? Check carefully! (3) | ||||
(2) | X |
Total out of 25 marks:
Biome Poster
The following activity is to be done in groups of four
Brainstorm a suitable set of criteria for assessment for poster and verbal report
Select one biome from the list given and do the following:
Use suitable references to obtain as much information as possible on the plants and animals found in your selected biome.
Make notes about the climate, landscape, flora and fauna, stating how some of these are adapted to their environment.
Design an attractive poster to illustrate the landscape as well as the dominant plants and animals that make up a food chain.
Display your poster on the classroom wall and each person of the group is to give a verbal presentation on an aspect of the biome you studied.
Biome Project
It is suggested that a rubric similar to the above be drawn up for assessment of the project. Such rubrics go a long way in standardising the assessment for different projects and they ensure that learners are given adequate feedback about what was correct / wrong about their project.
All teachers who have learners in this grade should come together and have a brainstorming session themselves, deciding on what the criteria are they will assess and what aspects will be taken into account within each criterion. The more clearly the criteria and sub-topics are defined in the beginning, the better. This cannot be over-emphasised.
It should be made clear to learners that group work DOES NOT involve one or two members doing all the work and others sitting back as spectators. ALL of them have to get involved and make a contribution to the project. The teacher may ask that they allocate specific tasks, e.g.
ALL: Decide on which biome to select and compile the food chain at the end.
Person 1: Collect pictures and information on plants of the area.
Person 2: Collect pictures and information on animals of the area.
Person 3: Collect pictures and information on climate and landscape.
Person 4: Design the poster and put it together.
ALL: Each member gives a verbal report on one aspect of this biome (max 1-2 minutes each).
Academia.edu no longer supports Internet Explorer.
To browse Academia.edu and the wider internet faster and more securely, please take a few seconds to upgrade your browser .
Enter the email address you signed up with and we'll email you a reset link.
- We're Hiring!
- Help Center

A Summer in South Africa: Reflections and Research on Race, Schools, and Humanity

This colloquium explored issues of race through the reflections and ethnographic notes taken during Barry’s two-month trip to Cape Town, South Africa. These reflections on race draw on literature that discuss the “white/non-white” binary and various tenets of critical race theory. In addition, Barry’s pictures, videos, and notes document the changes in his racial framework that occurred from the beginning to the end of the trip, offering a critique of traditional liberalism. This colloquium also discussed the merits of community-centered and student-focused schools in the United States using an international context.
Related Papers
Education as Change
Crispin Hemson
In the World Yearbook of Education 2023: Racialization and Educational Inequality in Global Perspective. Edited By Janelle Scott and Monisha Bajaj, published November 7, 2022, by Routledge. ISBN 9781032148434. Chapter 11, pp 189-203.
ENVER MOTALA
This chapter begins with an overview of the genesis and history of racialization in education in South Africa before discussing contemporary forms of "race" and class stratification in education. The chapter argues that present-day educational segregation in post-apartheid South Africa must be examined with reference to the history of racial capitalism and to contemporary socioeconomic and political disadvantage and patterns of inequality in society. The chapter asserts that racism in education does not constitute an autonomous form of oppression but is inextricably linked to power relations and reproduced in conjunction with class, gender, and other inequalities. Education is embedded in social class relations and largely reflects and reinforces the inequalities in a racial capitalist society. The chapter also documents resistance in South African education by focusing on the "Fees Must Fall" protests by university students.
Edmund ' T E D ' Hamann
As much as there are reasons for optimism as one thinks about changes in South Africa, Africa, and the United States in relation to the transcendence of racial differentiation and hierarchy, this book is a reminder of how both harrowing and incomplete that journey is. This book, a crucial addition from the Global South to the scholarship on immigrant students\u27 schooling, depicts how salient and fraught racial identity, both asserted and ascribed, continues to be for the negotiation of school in South Africa. Immigrant students are loathed and marginalized for their accents and \u27foreign\u27 ways, and yet they are also stereotyped and viewed jealously as more serious and committed students than their native-born Black peers
King Ndebele
Sharlene Swartz
Beginning in early 2000, the South African Department of Education (DoE) embarked on a Values in Education initiative, which has subsequently been renamed Race and Values in Education (RVIE). Broadly stated, the aim of the initiative is to articulate common values which should be central to the new curriculum and the educational experience of all South African children, youth and educators. The initiative was largely participatory and attempted to gain wide spread support and participation from various sectors of society. Much of the criticism of the process has centred around the theoretical construction of the term values, the dangers of nation-building in a global society, and the lack of consultation with educators. These criticisms were addressed through responses to the initial report (Values in Education) by the public, a conference in January 2000 (Saamtrek) and a final report issued in August 2001 (Manifesto). In this paper I will describe the process which lead to the creation of a directorate in the national department of education (Race and Values in Education) in South Africa and highlight the importance of citizenship education in a nascent democracy such as South Africa. I will then provide a critique of the process, the outcomes to date and future plans of the DoE, and finally I will articulate some of the inherent areas of challenge for the implementation of citizenship education and its intersect with both moral and religious education.
Derek Catsam
In both South Africa and the United States South, education stands and has stood historically as a vital cultural and economic center for its people. In both cases school integration has proved to be profoundly contentious. Certainly much of the Civil Rights Movement in the U.S. was centered on integrating schools from the elementary school playground to the university campus.
crain soudien
dzunisani sydney
Clarence George III
"Social cohesion is our primary objective; through learning about the cultures of other nations, we can discover the common roots of humanity and break down social barriers to achieve a powerful and unified identity," (Dr. Mathole Motshekga, Chair, Kara Heritage Institute and School, Pretoria, South Africa). I am a first-year doctoral student in African American and African Studies with a concentration in African Studies at Michigan State University. I recently earned a Master’s Degree in Ethnic Studies with an African American Studies minor at San Francisco State University in California.
Journal of Social Issues
Loading Preview
Sorry, preview is currently unavailable. You can download the paper by clicking the button above.
RELATED PAPERS
Rakgadi Phatlane
Africa Education Review
Lynne Damons , Doria Daniels
TPCS Tilburg Papers in Culture Studies
University of Cape Town
Dr. Wanelisa Xaba
Perspectives in Education
Christina Amsterdam , Mokubung Nkomo
Dolapo Adeniji-Neill
Salim Vally
Sadia Rajput
Mlamuli Hlatshwayo
Nomalanga Grootboom
Jacob R Boersema
Teaching in Higher Education
Carolyn McKinney
Power and Education
Nuraan Davids
IDS Bulletin
Crain Soudien
Zimitri Erasmus
Critical Education
doug selwyn
Kenton Special Edition
jacqui dornbrack
Transformation
Phila M Msimang
Thabang Dladla
Social Work/Maatskaplike Werk
Vishanthie Sewpaul
- We're Hiring!
- Help Center
- Find new research papers in:
- Health Sciences
- Earth Sciences
- Cognitive Science
- Mathematics
- Computer Science
- Academia ©2024

- Change Font Size A A
- Change Language हिंदी | Hindi
- Focus on Story
- हिंदी | Hindi
- T20 World Cup
- T20 WC Points Table
- Euro Schedule
- Table tennis
- Othersports
- Points table
- Cricket Matches
- Football Matches
- All Matches
India vs South Africa Live Streaming T20 World Cup 2024 Final Live Telecast: Where To Watch Match Live

India vs South Africa Live Streaming T20 World Cup Final Live Telecast : India take on South Africa in the T20 World Cup 2024 final in Barbados on Saturday. The Rohit Sharma -led side has been impressive with the competition till now with comfortable victories over tough opponents like Pakistan and Australia. On the other hand, South Africa are also unbeaten in the competition (like India) and this is the first time that they have ever qualified for a World Cup final in their cricketing history.
When will the India vs South Africa, T20 World Cup Final match be played?
The India vs South Africa, T20 World Cup Final match will be played on Saturday, June 29.
Where will the India vs South Africa, T20 World Cup Final match be played?
The India vs South Africa, T20 World Cup Final match will be played at Kensington Oval in Barbados.
What time will the India vs South Africa, T20 World Cup Final match start?
The India vs South Africa, T20 World Cup Final match will start at 8 PM IST.
Which TV channels will telecast the India vs South Africa, T20 World Cup Final match?
The India vs South Africa, T20 World Cup Final will be telecasted on the Star Sports Network.
Where to follow the live streaming of the India vs South Africa, T20 World Cup Final match?
The India vs South Africa, T20 World Cup Final will be streamed on Disney+Hotstar (Free for mobile users).
(All the details are as per the information provided by the broadcaster)
India: Rohit Sharma, Virat Kohli , Rishabh Pant , Suryakumar Yadav , Hardik Pandya , Shivam Dube , Ravindra Jadeja , Axar Patel , Kuldeep Yadav , Arshdeep Singh, Jasprit Bumrah .
South Africa: Quinton de Kock , Reeza Hendricks , Aiden Markram , Tristan Stubbs , Heinrich Klaasen , David Miller , Marco Jansen , Keshav Maharaj , Kagiso Rabada , Anrich Nortje , Tabraiz Shamsi .
Advertisement
The Daily Show Fan Page

Explore the latest interviews, correspondent coverage, best-of moments and more from The Daily Show.
Extended Interviews

The Daily Show Tickets
Attend a Live Taping
Find out how you can see The Daily Show live and in-person as a member of the studio audience.
Best of Jon Stewart

The Weekly Show with Jon Stewart
New Episodes Thursdays
Jon Stewart and special guests tackle complex issues.
Powerful Politicos

The Daily Show Shop
Great Things Are in Store
Become the proud owner of exclusive gear, including clothing, drinkware and must-have accessories.
About The Daily Show
- News for Kids
- Dominican Republic
- Netherlands
- New Zealand
- Papua New Guinea
- Philippines
- Puerto Rico
- South Africa
- South Korea
- Switzerland
- United Arab Emirates
- United Kingdom
- United States of America
- 7 Continents
- Australia/Oceania
- North America
- South America
- Chinese New Year
- Elections 2024
- Olympics 2024
- European Union
- Trivia & Quizzes
- Solar System Quiz
- Travel Reviews
- Travel Health
- Travel Links
Competition 2024
- Winners 2023
- Winners 2022
- Winners 2021
- Winners 2020
- Winners 2019
- Request A Correction
South Africa for Kids
Here are some interesting facts about South Africa for kids which were chosen and researched by kids especially for kids.
South Africa Facts | Country Facts

- Population : 60 million people live in South Africa (2023)
- Capital city : Pretoria has 3 million inhabitants. Pretoria's nickname is "The Jacaranda City" due to the many jacaranda trees. In South Africa, the city is referred to as 'Tshwane' as the municipality changed its name in 2007. However, South Africa has three capital cities: Pretoria, Cape Town and Bloemfontein
- Name : Republic of South Africa

- Government : Democracy
- Languages : 12 official languages: isiZulu, Afrikaans, English, isiXhosa, siSwati, Sesotho, Xitsonga, Sepedi, isiNdebele, Setswana, Tshivenda, sign language
- Religion : mainly Christians and Muslims, but also Jews and other faiths.
- Currency : 1 South African Rand (ZAR) = 100 Cents
- History : South Africa was colonised by British, Dutch suffered from Apartheid. Nelson Mandela came free from prison and became the first democratically elected leader in South Africa in 1994.

- National Symbols : Protea (flower), Springbok (antelope) and others. Click here.
- Climate : Various climatic regions. Mostly mild to hot in summer, most of the days, cold nights, snowfall in winter. Read more.
- President : Cyril Ramaphosa
South Africa for Kids South Africa Geography
Sout h Africa is the southernmost country on the African continent.
South Africa shares borders with six countries: Namibia, Botswana and Zimbabwe in the North, and Mozambique in the East, as well as with Eswatini and Lesotho.
South Africa encloses two landlocked African countries which are Eswatini (formerly called Swaziland) and Lesotho . Both these countries are kingdoms.
South Africa is about as big as the land area of France and Spain combined or twice the size of the state of Texas/ USA.
It takes a 12-hour flight from London/England to fly to Johannesburg or a 12-hour flight from Singapore and it takes more than 17 hours from New York/USA.

In South Africa, the Atlantic Ocean and the Indian Ocean meet off the Southern Cape coast.
The southernmost point in South Africa is, however, at Cape Agulhas which is roughly 120 km/ 75 miles to the east of Cape Town.
In the image to the right, however, you see Cape Point, the southernmost point of the Cape Peninsula.
South Africa for Kids Geography
South Africa has various mountain ranges such as the uKhahlamba-Drakensberg, the Magaliesberg or the Cederberg mountains.
There is a narrow coastal stretch and in the interior of the country, you will find a high plateau.
The highest mountain in South Africa is Mafadi Peak, which is on the border to Lesotho. Mafadi Peak is 3,450 m/ 11,319 ft high.
In the north of the country, you will find the Kalahari Desert which borders neighbouring countries Namibia and Botswana. The Kalahari is part of the 'highveld', the highlands of the country.
Johannesburg is the largest city of South Africa and lies on this plateau at 1,700 m/ 5,577 ft above sea level. However, most of the bigger cities such as Cape Town and Durban are located near the coast in low-lying areas.
The longest river in South Africa is the Orange River which forms also part of the border between Namibia from South Africa.
In the mountain ranges of the Drakensberg region, there is snow in winter and you even could go skiing in South Africa. Tiffindell is the most popular winter resort town. Sometimes also the higher mountain tops in the Western Cape near Cape Town receive some snow in winter. But there is no snow in the city or in the towns along the coastline. Although the snow might be enough for a snowball fight, it certainly is not enough to go skiing.
Did you know, t hat the Tugela Falls in South Africa are the second-highest waterfalls in the world?
The country name is often displayed as ZA which means Z uid A frica and is Afrikaans for South Africa.
South Africa for Kids: South Africa Attractions

- Cape Town : Table Mountain, V&A Waterfront, beaches. More about Cape Town attractions here.
- Kruger National Park for safaris and great wildlife viewing
- Sun City near Pretoria for fun and games galore with amazing wave pool and lots of things to do for the whole family
- Addo National Park near Port Elizabeth for elephant-spotting

- Durban for surfing and great beaches and the KwaZulu-Natal midlands for some insights into South African history
- iSimangeliso (formerly St Lucia Wetlands) for wildlife viewing and unspoilt nature
- Drakensberg Mountains for hiking and nearby Lesotho for skiing in Africa
South Africa for Kids: Animals
Wildlife in South Africa is abunda nt. There are many species of antilope such as kudu, springbok, wildebeest, impala, oryx and gemsbok. When you go on safari you can see many other South African animals like lions, elephants, giraffes and monkeys.

South Africa is home t o the African Penguins and the Great White Sharks live in the ocean waters here too. Sometimes beaches are closed for bathing due to the sharks in the bays. Many beaches have shark nets. Remember to only swim in the sea where there are lifeguards on duty.
Whale watching is very popular in South Africa too and you will encounter an eno rmous sea life in South Africa when you go on a boat trip.
South Africa for Kids South Africa People
South Africa is often calle d the 'Rainbow Nation', because there are so many different languages are spoken and different cultural traditions are lived. Below you see people of the Ndebele tribe.

South Africa is often calle d "The Cradle of Humankind", because archaeological artefacts, tools and human remains proof that people lived in the area more than 100,000 years ago.
The ancient San and the Khoi were the early African civilizations settling the Cape. There are only few descendants of the local San people living in remote areas in the Karoo desert and near the Cape West Coast.

The biggest South African population group, the Zulu, mainly live in rural settlements in KwaZulu-Natal, they even have a king.
Portuguese, Dutch, British, French and German explorers have traveled and populated southern Africa in the 17 th and 18 th century and therefore you can find many European influences in South Africa too.
Read more about the 11 official languages in South Africa and learn some fun South African slang words here.

Nowadays most of the people in South Africa live in or near the big cities: Johannesburg, Pretoria, Durban and Cape Town. The rural areas are still very poor and also in the cities many people live in shantytowns or townships.
Johannesburg is the largest city and the main industrial and economic hub, while Cape Town and Durban are also large cities with more than 1 million inhabitants.
Did you know that in South Africa, cars drive on the left side of the road as they do in the UK and Australia? However, all distance and speed signs are in metres and kilometres and km/ hour and not as in England in yards and miles and miles/ hour!
South Africa for Kids Food in South Africa
The food the people eat in South Africa is very diverse. The South African staple food for the locals is 'mealies', which is corn, and seasonal vegetables, like potatoes, beans, carrots and peas.
South Africans love their meat and game meat such as kudu and springbok are common. However, most people eat beef, chicken, lamb and ostrich with their meals. The people in South Africa love a BBQ (here called 'braai') and almost every house has got a fireplace which is called 'braai-place'.

Typical South African food :
- bobotie : typica Cape Malay food containing rice, lamb and fragrant spices. Click to find a tasty easy-to-do bootie recipe.
- koeksisters : taste like donuts soaked in sticky sweet sauce. Yummy!
- mealie pap : corn porridge

- biltong : a cured and dried meat that is a typical South African snack and similar to beef jerky. Kids love to take this as snack to school.
- boerewors : Afrikaans for 'farmers sausage', is often put on the grill, formed in a curl (see image)
Read more about South African traditional food here.

Did you know that...?
… most people speak at least two or three languages, one of them usually English or Afrikaans.
… South Africa has really three capital cities. The powers are distributed between Pretoria, Cape Town and Bloemfontein. The parliament meets for six months in Pretoria and the other half of the year in Cape Town.
We hope you enjoyed our information on South Africa for Kids. Read also our top 15 South Africa facts even locals often do not know here.

Popular Pages
Resources: South Africa for Kids
- Central Intelligence Agency. "South Africa". WorldFactBook . Last updated 8 March 2022. Last accessed 30 March 2022
- Nelson Mandela Foundation. "Mandela Day." NelsonMandelaFoundation . Last accessed 30 March 2022
- South African History Online. People and Culture of South Africa. SAHistory.org. Last accessed 30 March 2022.
- South African Government. "Geography and Climate" gov.za. Last accessed 30 March 2022.
Join our 2022 competition and win

Go back from South Africa for Kids to Kids-World-Travel-Guide Homepage
Competition 2024 is open!

Would you prefer to share this page with others by linking to it?
- Click on the HTML link code below.
- Copy and paste it, adding a note of your own, into your blog, a Web page, forums, a blog comment, your Facebook account, or anywhere that someone would find this page valuable.
Events & Celebrations
Organisations, games & quizzes, travel tips, competition, recent articles, facts about bulgaria for kids | bulgaria facts | geography | travel.
Jun 28, 24 07:49 AM
Facts about Monaco for Kids | Europe facts for kids | Travel | Monaco
Jun 24, 24 10:23 AM
Mexico Facts for Kids | Mexico Attractions | Food | People | Geography
May 30, 24 04:44 AM
More about South Africa

More about Africa

More Countries in Africa

South Africa for Kids?
Make sure to visit these fascinating South African attractions and venues:
- Table Mountain
- Simons Town Penguins
- Umhlanga Sharks Board
- Mossel Bay Diaz Museum
- Sutherland Astronomical Observatory
- Gold Reef City
- Apartheid Museum
- Kruger National Park
- Addo Elephant Park

Temperature in Celsius
Temperature in fahrenheit, did you like what you read.

| Simply use the html code below. Copy and paste onto your website, blog or Facebook page: https://www.kids-world-travel-guide.com/south-africa-for-kids.html">Kids World Travel Guide: South Africa for Kids</a> |
Winning Essays 2021

Winning Essays 2022

Like us on Facebook
Kids World Travel Guide

Brilliantly
Content & links.
Verified by Sur.ly
©Kids-World-Travel-Guide.com 2010-2024 | Created by Regina Gräff and KidsWorldTravels
All rights reserved | Privacy Policy | Disclaimer

- Essay Samples
- College Essay
- Writing Tools
- Writing guide

Creative samples from the experts
↑ Return to Essay Samples
Descriptive Essay: My Country South Africa
South Africa is a country situated on the most southern tip of Africa cradled by the icy Atlantic Ocean on the west and the warm Indian Ocean on the south and east. The southernmost tip jutting out into the ocean is called Cape Aghulhas (Cape of Needles) and separates these two great oceans. To the north are Mozambique, Botswana, Namibia and Zimbabwe. South Africa encircles the independent kingdoms of Swaziland and Lesotho.
The 11 official languages are Afrikaans, English, Ndebele, Northern Sotho, Sotho, Swazi, Tswana, Tonga, Venda, Xhosa and Zulu. The three most spoken first languages are Zulu (22.7%), Xhosa (16%) and Afrikaans (13.5%). English is the language of commerce and science and is the first language of 9.6% South Africans. There are also semi-official and unofficial languages.
The unofficial languages of the Khoikhoi and San are in grave danger of extinction. They are the aboriginal peoples of South Africa and are physically distinct with an ancient culture founded on hunter-gatherer societies.
The country is made up of nine provinces Western Cape, Northern Cape, Eastern Cape, North West, Limpopo, Gauteng, KwaZulu-Natal, Mpumulanga and Free State. The main cities are Durban (coastal Kwazulu-Natal), Johannesburg (inland Gauteng), Cape Town (coastal Western Cape) and Pretoria (inland northern Gauteng).
Population and Size
South Africa takes up four percent of Africa’s landmass i.e. 1 221 040 square kilometers making it three times bigger than Texas and five times bigger than Britain. This area is populated by about 52 million people.
Our first peoples were the San and Khoikhoi established for thousands of years before the arrival of the Bantu-speaking tribes who were established hundreds of years before the Europeans. The Europeans were sent by the Dutch East India Company and arrived at the Cape in 1652 to build a fort and start a garden for ships on the eastern trading route. Eventually, this route also brought slaves to the Cape until slaves were freed in 1833.
The Afrikaans speaking Boers, descended mainly from Europeans, trekked north and east to establish the Boer republics of Orange Free State and Transvaal. They did not want to be under British rule. After diamonds were discovered in 1867, and gold in 1876, a war broke out between Boers and British. In 1902, the Boers were defeated and in 1910 South Africa became a Union of 2 British and 2 Boer colonies.
The ANC was founded in 1912 but in 1936 black South Africans were expunged from the voter rolls. In 1950, more than 1 million black South Africans were forced from cities to rural areas and poverty.
In 1961, South Africa became a republic under the white supremacist Nationalists and the country was gradually weakened by internal black opposition and international boycotts. In 1990, this led to de Klerk releasing Mandela from 27 years in prison. In a 1992 referendum, white South Africans voted to support de Klerk’s negotiations and in 1994 the ANC won the first democratic election and Mandela became president on 10 May.
My country has experienced a tragic history but today, all South Africans strive equally for a better future and to maintain democratic values e.g. the country is united in preventing current President Zuma from restricting freedom of speech. Our biggest challenge is reducing poverty but this is possible due to tourism, mineral wealth and Johannesburg being the thriving power-house of business on the African continent.
http://www.edusouthafrica.com/south-africa.html http://www.factmonster.com/ipka/A0107983.html http://www.factmonster.com/country/profiles/south-africa.html http://www.southafrica.info/about/history/521109.htm#.UzKlhqiSzxI

Follow Us on Social Media
Get more free essays

Send via email
Most useful resources for students:.
- Free Essays Download
- Writing Tools List
- Proofreading Services
- Universities Rating
Contributors Bio

Find more useful services for students
Free plagiarism check, professional editing, online tutoring, free grammar check.

COMMENTS
The Magic Of A South African Summer English Essay. The magic of a South African summer is something that cannot be put into words. From the beauty of the bushveld to the rolling hills of the Cape Winelands, the country is a stunning visual feast of natural beauty. The warmth of the sun, the sound of the birds singing, and the sound of the ocean crashing against the shore all combine to make a ...
Essay Writing. Niamh Walsh Vorster---- ... David Mann is a writer, editor and arts journalist who lives and works in Johannesburg, South Africa. Follow. More from David Mann and David Mann. David ...
Another fabulous reason to Visit South Africa this Summer is the Cederberg. It's quite usual to see hardened rock climbers, their odd mattresses on their backs, heading from the car park just inside the Cederberg Wilderness, less than three hours from the traffic jams of Cape Town. The Cederberg, most notable for its rooibos tea production ...
South African summer runs from November-February, so expect beautifully hot, dry, sunny days. With average highs of 29° it's too tempting to resist. 2. Waves. Let's be honest, the waves are what we're all here for. Due to Cape Town's unique peninsula, throughout the South African Summer months, south easterly winds create amazing ...
March marks the end of southern Africa's 2021/2022 summer wet-season. Since its onset in October, most summer rainfall zone regions have experienced wetter than normal conditions. Wetter ...
STATS SA said in its report, Poverty Trends in South Africa, an examination of absolute poverty between 2006 and 2015, that more than half of the South African population live under the poverty ...
Here are some of the top reasons this beautiful country during summertime: 1. Beach Beauties. Photo credit: Campdell Colledge. South Africa has some of the best beaches in the world and although Cape Town and Durban in particular are famed for their beaches, the Eastern Cape and Garden Route regions have some glorious offerings too.
South Africa's batting hasn't had the best time at this World Cup, averaging 21 and striking at 106. They're two-down early. 16 wickets for Arshdeep in this T20 World Cup, the joint-second most by ...
The Indian cricket team ended their 11-year wait for an ICC trophy after they defeated South Africa by seven runs in a thrilling T20 World Cup 2024 final at the Kensington Oval in Bridgetown ...
South Africa - playing in their first World Cup final in either T20 or 50-over cricket after winning a semi-final at the eighth attempt against Afghanistan - looked poised to reach their target of ...
The magic of south African Summer - Free download as Word Doc (.doc / .docx), PDF File (.pdf), Text File (.txt) or read online for free. South Africa offers a magical summer experience with its beautiful landscapes and wildlife. The document describes a family trip to Cape Town, where they enjoyed crystal blue waters and soft white beaches, saw where two oceans meet at Cape Agulhas, and ...
Here is a list of festivals that are actively celebrated in South Africa: September, 24 — heritage day. October, 10 — day Kruger; Summer in South Africa. At December hot tropical summer comes to the Republic of South Africa. Average daytime temperature is in the range between 26 °C (79 °F) in Cape Town and 28 °C (82 °F) in Pretoria and ...
India beat South Africa by seven runs in Barbados to win Mens T20 World Cup for a second time; Proteas denied a first World Cup title after heart-breaking defeat at Kensington Oval - watch England ...
There is much to love about the land at the southern tip of Africa but for me its biggest strength is its diversity. Having grown up under apartheid, I used to think that 'diversity' was only a good thing when it referred to the varied South African landscape. Of course the many worlds in one country is wonderful: You can drive only a ...
The World And South Africa Beyond 2023: The Latest Flags, Scenarios And Probabilities; The ANC Below 50%: What Is The Road Ahead? Anatomy Of State Capture; South Africa's Black Middle Class: Past, Present And Future; Philosophical Lives; A Voyage Of Discovery: The Trans-siberian Railway; Napoleon, The Rosetta Stone, And The Deciphering Of ...
2. Get Fit: Physical activity is the number one evidence-based way to reduce anxiety. Aim for at least 30 minutes of exercise a day, whether it's a morning run, yoga session, or playing a sport ...
Ogunbayo A, Mogotsi MT, Sondlane H, Nkwadipo KR, Sabiu S, et al. Metagenomic analysis of respiratory RNA virome of children with and without severe Acute Respiratory infection from the Free State, South Africa during COVID-19 pandemic reveals higher diversity and abundance in summer compared with Winter Period.
8. The magic of a South African summer!!! Summer yadi summer, summer yomuthi just to name a few phrases of a South African summer. The blazing hot sun, clear skies, ohhh the thunders that come with the rain then leaves a few moments after, it leaves the warmth and what l call the rainbow of hope. Lalela, listen let me tell you about the magic ...
Downtown Cape Town is beautiful, and the harbor area is one of the nicest I've seen with amazing shopping OOPS. It doesn't take a lot to understand why the most expensive real estate in Africa is here. On my first trip over to South Africa, Cape Town was the first stop after a long conference in Durban. And trust me, I was basically sobbing ...
The biome regions in South Africa are mapped in Figure 8.5. Figure 8.5: Biomes of South Africa. 1. Grassland Biome. Location: grasslands are found on the Highveld. Climate: they typically have summer rainfall of 400 mm to 2000 mm. Winters are cold, and frost can occur. Soil and geography: in grasslands, the soil is red/yellow/grey or red/black ...
This colloquium explored issues of race through the reflections and ethnographic notes taken during Barry's two-month trip to Cape Town, South Africa. These reflections on race draw on literature that discuss the "white/non-white" binary and various
Up to 15 Tories 'being investigated by Gambling Commission over betting scandal' Controversy has now cut through more with public than anger over PM's D-Day departure, poll shows
It was aptly coined the ''Rainbow Nation'' by national icon, Archbishop Desmond Tutu, describing the ''New South Africa'', post-apartheid, after the very first democratic elections way back when in 1994. Culturally and ethnically diverse as they come, a Rainbow Nation gave South Africans the long awaited chance to embrace their ...
2nd Semi-Final, ICC Men's T20 World Cup, 2024 at Guyana, Jun 27, 2024
The source for The Daily Show fans, with episodes hosted by Jon Stewart, Ronny Chieng, Jordan Klepper, Dulcé Sloan and more, plus interviews, highlights and The Weekly Show podcast.
Oceans moderate temperatures along the coast, causing summer to be less hot and winters less cold than in the interior. Places along the coast have a maritime climate (low temperature range) The influence of South Africa's latitudinal position South Africa is situated between the latitudes 22°S and 33°S. This means that
Cnn vs fox essay with essay about summer in south africa. The passage indicates that it is easier to rewrite the tale, beginning with what precision. Thus, provincializing europe would necessitate a shift in the reference in the. Still in search of yamamba a critique or at least once or more self-selected graduate students has conventionally ...
South Africa Facts | Country Facts. Population: 60 million people live in South Africa (2023) Capital city: Pretoria has 3 million inhabitants. Pretoria's nickname is "The Jacaranda City" due to the many jacaranda trees. In South Africa, the city is referred to as 'Tshwane' as the municipality changed its name in 2007.
Descriptive Essay: My Country South Africa. South Africa is a country situated on the most southern tip of Africa cradled by the icy Atlantic Ocean on the west and the warm Indian Ocean on the south and east. The southernmost tip jutting out into the ocean is called Cape Aghulhas (Cape of Needles) and separates these two great oceans.
The summer climate, while typically moderate, is changing, with rising temperatures and variable rainfall patterns affecting the delicate balance of South Africa's national parks and agriculture. Despite these climatic challenges, summer is a time of unity and striving for freedom, as echoed in the national call to come together, showcasing the ...