- Open access
- Published: 02 May 2022

A case management model for patients with granulomatous mastitis: a prospective study
- Yuan Deng 1 , 2 ,
- Ying Xiong 1 ,
- Ping Ning 1 ,
- Xin Wang 2 ,
- Xiao-Rong Han 1 ,
- Guo-Fang Tu 2 &
- Pei-Yu He 1
BMC Women's Health volume 22 , Article number: 143 ( 2022 ) Cite this article
3586 Accesses
5 Citations
1 Altmetric
Metrics details
Granulomatous mastitis (GM) is a chronic inflammatory mastitis disease that requires long-term treatment and has a high recurrence rate. Case management has been proven to be an effective mechanism in assisting patients with chronic illness to receive regular and targeted disease monitoring and health care service. The aim of this study was to investigate the application of a hospital-to-community model of case management for granulomatous mastitis and explore the related factors associated with its recurrence.
This was a prospective study on patients with granulomatous mastitis based on a case management model. Data on demographic, clinical and laboratory information, treatment methods, follow-up time, and recurrence were collected and analyzed. The eight-item Morisky Medication Adherence Scale (MMAS-8) was used to investigate patients' adherence to medications. Logistic regression models were built for analysis of risk factors for the recurrence of granulomatous mastitis.
By October 2021, a total of 152 female patients with a mean age of 32 years had undergone the entire case management process. The mean total course of case management was 24.54 (range 15–45) months. Almost all the patients received medication treatment, except for one pregnant patient who received observation therapy, and approximately 53.9% of the patients received medication and surgery. The overall recurrence rate was 11.2%, and “high” medication adherence (RR = 0.428, 95% CI 0.224–0.867, P = 0.015) was significantly associated with a lower rate of recurrence, while the rate of recurrence with a surgical procedure + medication was higher than that with medication alone (RR = 4.128, 95% CI 1.026–16.610, P = 0.046).
A case management model for patients with granulomatous mastitis was applied to effectively monitor changes in the disease and to identify factors associated with disease recurrence. “Low” medication adherence was a significant risk factor for the recurrence of granulomatous mastitis. Patients treated with medication and surgery were more likely to experience recurrence than those treated with medication alone. The optimal treatment approach should be planned for granulomatous mastitis patients, and patient medication adherence should be of concern to medical staff.
Peer Review reports
Granulomatous mastitis (GM) was first reported as a chronic inflammatory disease of the breast by Kessler and Wolloch in 1972 [ 1 ], and accounts for approximately 1.8% of benign breast diseases [ 2 ]. The main clinical presentation is a palpable, painful breast lump with concomitant skin erythema, nipple retraction, sinus tract formation, cellulitis changes, and axillary adenopathy formation [ 3 , 4 , 5 ], and in severe cases, there are usually multiple coexisting focal abscesses with skin inflammation and ulceration [ 5 ]. According to the severity of the disease, GM is clinically classified into mass, abscess, and refractory types [ 6 ]. Patients often endure a long disease course, as well as changes in breast appearance caused by the disease, which has serious physical and psychological effects on patients [ 7 ]. With only 2.4 per 100,00 incidences reported by the Centers for Disease Control and Prevention in 2009, most countries have not conducted large epidemiological surveys for GM due to the rarity of the disease [ 8 ]. To date, the etiology of GM is unknown and may be associated with a history of pregnancy, autoimmune disease, breast trauma, hyperprolactinemia, and infection [ 7 , 9 ]. The disease progresses rapidly with a recurrent or prolonged natural course, which has a high recurrence rate of 5%-50%, and is commonly seen in young women with a history of breastfeeding and childbirth [ 3 , 10 , 11 , 12 ]. As recently reported, there are racial differences in this disease, and the incidence of GM in Middle Eastern countries (Egypt, Turkey, Iran) and Spain is higher than that in Western countries (UK, USA, New Zealand) [ 13 , 14 , 15 ]. A large number of cases of GM have been described, mainly from Asian and Mediterranean countries, such as China, Iran, and Turkey [ 16 , 17 ]. However, there is no consensus on the management of GM and no gold standard regarding the diagnosis and treatment of the disease [ 4 ]. Currently, the main treatment include observation, medication therapy (steroids, antibiotics, methotrexate (MTX), and anti-molecular bacilli) and/or operative interventions (abscess incision and drainage, simple mass excision, enlarged mammary mass excision, etc.) [ 15 , 18 ], and medication therapy is the most commonly used treatment. The toxic side effects of long-term medication use have a significant impact on patients' quality of life, resulting in poor compliance with drug use, therefore, timely observation of medication use and changes in the breasts is essential to achieve good recovery rates for GM patients [ 11 , 18 , 19 , 20 ].
Recently, one approach to managing care that has gained wide popularity is case management [ 21 ], which promote access to provide patients with regular and targeted disease monitoring and health guidance through follow-up visits and WeChat consultations in China (WeChat is a mobile chat software by the Chinese company Tencent, in which patients can quickly consult with medical staff by sending voice messages, videos, pictures and texts over the internet quickly) [ 22 ]. Nurse specialists are responsible for the overall coordination, management, and continuity of care for a specific treatment or intervention to meet the health needs of an individual, reduce health care costs and improve the quality of service [ 23 , 24 ]. Currently, it is known that case management is widely applied for patients with breast disease, especially breast cancer [ 25 , 26 ], but it is rarely to applied for GM patients. Based on the characteristics of the disease, which is mostly treated and followed up in outpatients, a tailored model should be developed that it enables health providers monitor the condition changes of GM patients from outpatient to community to inpatient settings. A hospital-to-community model of case management, which allows cases managers to track and manage the treatment of GM patients from hospital to community settings, was described by Lamb in 1992, and includes the following five basic activities of case management: (1) assessment, (2) planning, (3) linking, (4) monitoring, and (5) advocacy [ 27 ]. Since January 2018, a tailored model for GM based on a hospital-to-community model, which can provide patients with full management and seamless health care services, has been explored and practiced in Chengdu Women's and Children's Central Hospital.
To better observe the development of this disease with treatment and identify some of the factors associated with its recurrence, we used a hospital-to-community-based model of case management to monitor the condition changes of GM patients. Prospective studies can provide more effective strategies and optimal approaches to prevent the recurrence of disease.
Materials and methods
Study design and participants.
A prospective study on patients with granulomatous mastitis based on the case management model was undertaken between January 2018 and November 2020 in the Breast Unit of Chengdu Women’s and Children’s Central Hospital. According to the characteristics of the disease, the whole case management process, presented in Fig. 1 , was divided into four key stages, including the diagnostic, conservative, perioperative, and follow-up periods. The entire process was led by case managers and tailored for patients, including the evaluation, planning, integration, implementation, and evaluation of treatment plans. Participants were followed up through the whole process. The case closure time was defined as the time when a patient was free of relapse during the 1-year follow-up period after the discontinuation of medication or surgery.
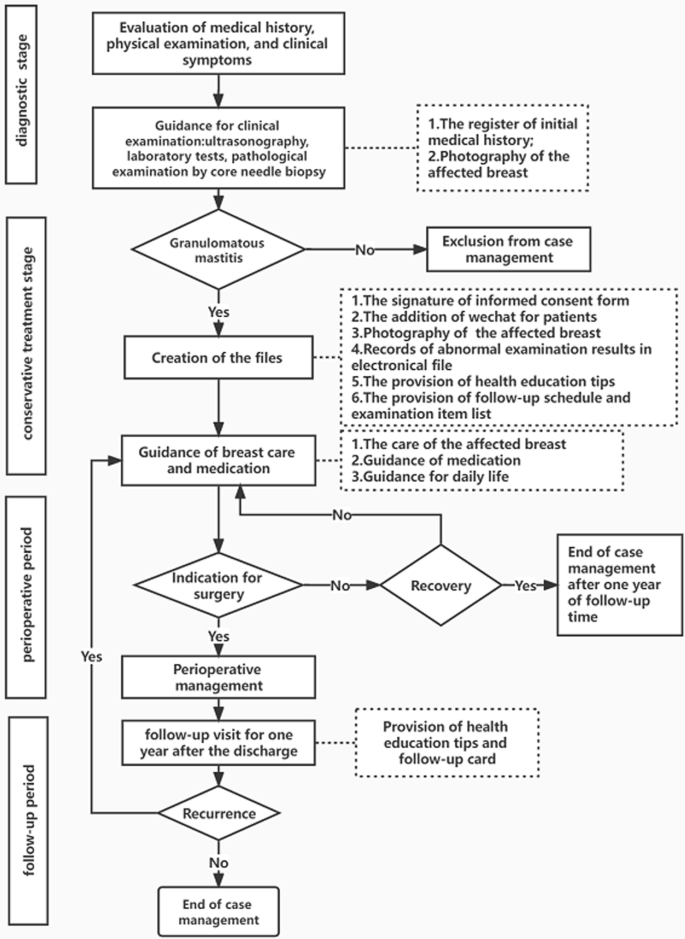
the algorithm for the case management of granulomatous mastitis
In the diagnostic stage, the case managers mainly based their decisions on clinical symptoms, regardless of whether a register of the initial medical history was created including age, pregnancy history, disease history, onset time, onset trigger, and contact phone number. A patient’s diagnosis of granulomatous mastitis was confirmed by the results of a pathological examination by core needle biopsy, and then a case management file was established. In the conservative treatment stage, case managers mainly performed the following: (1) followed up and recorded the results of ultrasounds, abnormal laboratory tests and breast signs, and explained the precautions and methods of medication administration according to a doctor's prescription; (2) surveyed GM patients for medication adherence at 2 months of drug use by the eight-item Morisky Medication Adherence Scale (MMAS-8) [ 28 ]; (3) distributed notes of disease considerations related to diet, sleep, behaviors, etc., as shown in Table 1 [ 6 , 29 , 30 ]; and (4) established a contact platform for GM patients to understand and observe the changes in their breasts during treatment, while being given psychological support and guidance at home. In the perioperative and follow-up periods, the case managers recorded the patients' surgery, medication, follow-up time and recurrence information.
In this study, qualifications for case managers were as follows: (1) nurses with bachelor's degree or above; (2) nurses with an intermediate title or above; (3) nurses with 5 years of experience or more in the breast department; (4) nurses who had received the training, which included the case management process, communication and health promotion skills; (5) nurses who were required to rotate through the breast clinic, ultrasound and pathology department, wound care unit and operating room, and (6) nurses who had passed the hospital examination for case management. All patients who received case management were eligible for inclusion if they were older than 18 years, had clinical breast symptoms, and had a confirmed diagnosis by core needle biopsy. Patients were ineligible if they had other complications of the breast and had been treated at other institutions. The study was approved by the Ethics Committee of Chengdu Women's and Children's Central Hospital (No. B2019 (13)). All participants signed an informed consent form.
Case definition
Histopathological examination is a necessary and gold-standard method for the diagnosis of granulomatous mastitis [ 31 ], so a definitive diagnosis of GM was largely accomplished with core needle biopsy in this study. The disease may be locally invasive with a risk of recurrence, and recurrence rates of 5 to 50% have been observed by various studies in recent years [ 10 , 11 , 12 ]. The following definition of recurrence was used in this study: the detection of new lesion (s) within the range of the primary location or any other part of the ipsilateral breast 1 month following the termination of therapy.
Medication adherence was measured using the eight-item Morisky Medication Adherence Scale (MMAS-8) [ 28 ], which was translated into a Chinese scale by Lin Chen et al. [ 32 ]; this scale has high reliability and validity and has been widely used in studies of various chronic diseases in China [ 32 , 33 ]. Three levels of adherence were considered based on the following scores: 0 to < 6 (low); 6 to < 8 (medium); and 8 (high). In a meta-analysis by Lei et al. [ 34 ], oral drug therapy was an effective treatment modality or GM patients in receiving both surgical and conservative treatment. According to the relevant literature, steroids are the most prominent drugs for GM, which usually lasts from 3 to 12 months, with a minimum of 2 months [ 35 , 36 , 37 , 38 ]. To survey as many patients as possible, we chose to conduct a survey of medication adherence at 2 months of medication use.
Statistical analysis
The statistical software package SPSS for Windows, version 19.0 (SPSS Inc., Chicago, IL) was used for statistical analysis. Clinical characteristics were described using the mean ± standard deviation, the mean (range) or numbers (and percentages) as appropriate. Continuous variables were compared between patients with and without recurrence using one-way ANOVA, while categorical variables were compared using the Chi-square test and Fisher's exact tests. Logistic regression models were built for the analysis of risk factors for the recurrence of GM.
Patient characteristics
In this study, 204 symptomatic patients with granulomatous mastitis were initially included in the diagnostic stage between January 2018 and November 2020. However, 4 patients were diagnosed with breast cancer, 8 dropped out, and 40 were still undergoing case management. Ultimately, 152 patients had completed case management by September 2021. Table 2 shows that the mean age of the patients was 32 years (range 22–48). It was observed that 71 (46.7%) patients had normal BMI, while 64 (47,4%) patients had a BMI higher than 25, and were considered overweight or obese. It was detected that the period in which GM was most frequently seen was the first 2–5 years after birth, with 94 patients (61.8%), followed by 30 patients (19.7%) diagnosed 0–2 years after birth (4 patients were breastfeeding), and 15 patients (9.9%) diagnosed during pregnancy. Accompanying diseases were found in only 28 (18.5%) patients, such as diabetes mellitus, thyroid disease, psychoses, hypertension, and hyperprolactinemia, accounting for the highest percentage of 13.8% of all comorbidities.
On physical examination, the most common finding was a palpable mass with pain (98.7%); 38.8% of the patients had a breast abscess, 75% suffered from skin lesions, and approximately 5% had fistulas and erythema nodosum (Table 2 ). Based on clinical symptoms, the disease was typed as the mass (74, 48.7%), abscess (66, 43.4%), and refractory types (12, 7.9%). Unilateral involvement was observed the most in 140 (92.1%) patients. In this study, 30.96% of the patients reported that they had bad behaviors a week before disease onset, including breast trauma (8.6%), excitant food (14.47%), and staying up all night (7.89%).
Patient treatments
Table 3 shows the different treatments that were administered. Of the 152 patients, only 1 (0.7%) recovered under observation without treatment, 82 (53.9%) recovered with medication and surgery, and 69 (45.4%) recovered with medical treatment alone. In the courses of medications, 65 (42.8%)patients chose systemic steroids alone, 21 (13.7%) patients chose tubercle bacillus drugs alone, and 65 (42.8%) patients required a combination or change of the drug regimen due to ineffective treatment or drug side effects including erythema nodosum (5.3%), skin rash (5.3%), abnormal index of liver function (7.2%), abnormal uric acid (2.0%) and edema on the lips and face (0.7%).
Patient follow-up visits
The mean follow-up time was 25.55 months (range 15–45) for the patients treated with medication and surgery, while it was 23.83 months (range 17–36) for the patients treated with medication alone. There was no statistically significant difference between the groups ( p = 0.570). The recurrence rate in the series was determined to be as 11.2% with 17 patients experiencing recurrence. At 2 months of initial medication use, the medication adherence outcome of the GM patients was “high” for 59 patients (39%), “medium” for 70 patients (46.4%), and “low” for 22 patients (14.6%), as shown in Table 4 .
Factors associated with recurrence
All statistically significant variables ( P < 0.05) related to BMI, treatments, medication use and medication adherence (Table 5 ) were included in the multivariable logistic regression model. The results of the multivariable analysis are shown in Table 6 . Surgical procedure and drug treatment (RR = 4.128, 95% CI 1.026–16.610, P = 0.046) were independently associated with an increased recurrence risk of granulomatous mastitis. In contrast, “high” medication adherence (RR = 0.428, 95% CI 0.224–0.867, P = 0.015) was associated with decreased recurrence risk.
Discussion and conclusion
This is the first study to report a case management model applied for GM patients. Although GM is a benign disease, its recurrence, one of the main challenges in the management of patients with the disease, has been reported to occur in 5%-50% of patients [ 10 , 11 , 12 ]. In our study, the recurrence rate of 11.2% is low in this range. Seventeen patients experienced recurrence, including ten with new lesions in the ipsilateral breast and seven with new lesions in the contralateral breast.
In recent years, the prevalence of granulomatous mastitis has been rapidly increasing, and the most affected patients are women of childbearing age [ 39 ]. In two studies, Freeman et al. reported that up to 86% of GM patients had a history of pregnancy in the past 5 years [ 38 ]. Prasad et al. reported that 73 patients with GM had a mean age of approximately 33 years and a history of childbirth 4.6 years before mastitis on average [ 40 ]. In our study, which had similar characteristics to previously reported studies, the median age of the patients was 32 years (range 22–48), 119 patients had a history of childbirth within the last 5 years, 15 patients had concurrent pregnancy, and 4 patients were currently breastfeeding. These findings indicated that hormones play an important role and may be related to the secretion theory, which has an important place in the pathophysiology of GM [ 12 ]. It has been postulated that GM results from a localized autoimmune response to the retained or extra vacated fat- or protein-rich secretions in the breast ducts in women of childbearing age due to previous hyperprolactinemia [ 41 ]. Therefore, the breast care for women of childbearing age deserves our attention.
GM patients mostly have mass and pain symptoms, and skin lesions and abscesses can be observed in mass localization. Findings such as fistula, erythema nodosum, and nipple or skin retraction can also be observed [ 1 , 2 , 35 ]. In many studies, the most common reported complaint at the time of the initial visit was a unilateral painful breast mass [ 35 , 42 ]. Similarly, 98.7% of the patients had mass and pain complaints, and 92.1% of the patients presented with a unilaterally affected breast. The case managers made initial judgments and provided tentative guidance based on clinical presentations. At the initial visit, there were mass (74, 48.7%), abscess (66, 43.4%), and refractory types (12, 7.9%), which were not associated with recurrence in the later stages ( P = 0.2). As the disease progressed, 10 mass type cases were actually abscess type cases, and 4 abscess type cases were actually refractory type cases. An important consideration for case managers is the care of the affected breast (shown in Fig. 2 and Fig. 3 ). Wound care should consist of managing drainage from fistulae with gauze and other nonadherent dressings. Tape should be avoided due to further abrasion and irritation of the skin [ 43 ]. Meanwhile, if a patient has a superficial abscess, a case manager should percutaneously perform puncture aspiration, and determine how deep the abscess is, while a mammographer, assisted by ultrasound guidance, performs puncture drainage, to create a path for the drainage of secretions and reduction of pressure in the inflamed area due to the accumulation of inflammatory fluid.
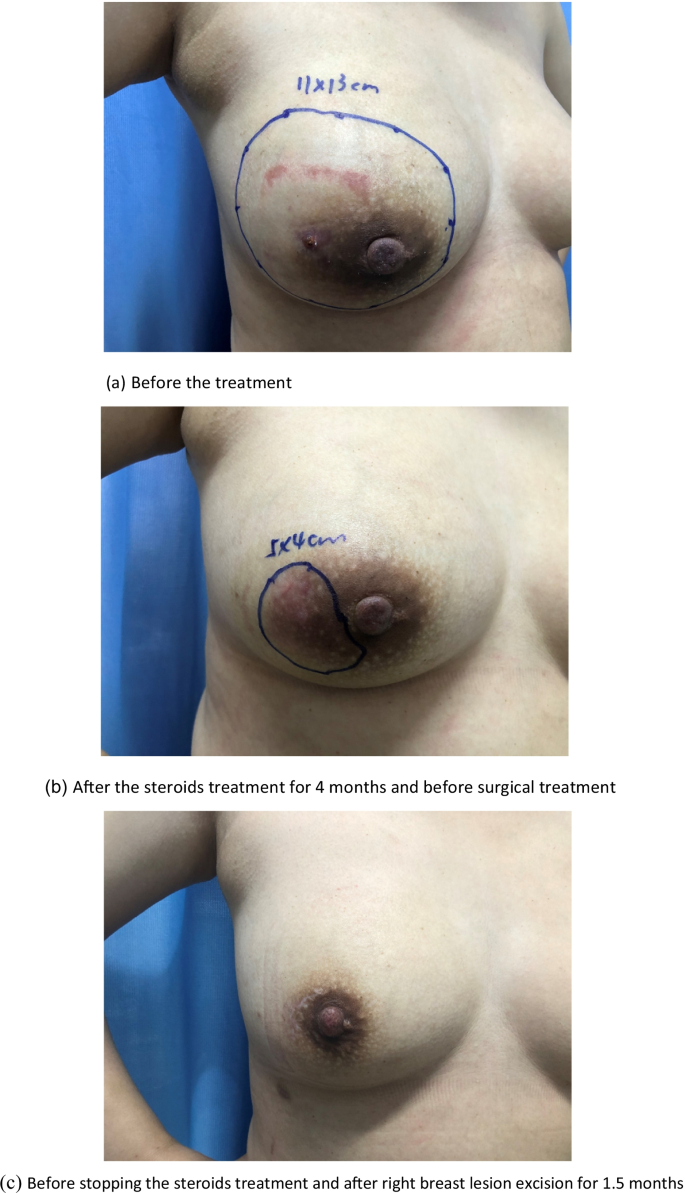
The effect of medical and surgical treatment in the case management. The underlined part of the figure shows the scope of the lesion located by ultrasound. a Before the treatment. b After the steroids treatment for 4 months and before surgical treatment. c Before stopping the steroids treatment and after right breast lesion excision for 1.5 months
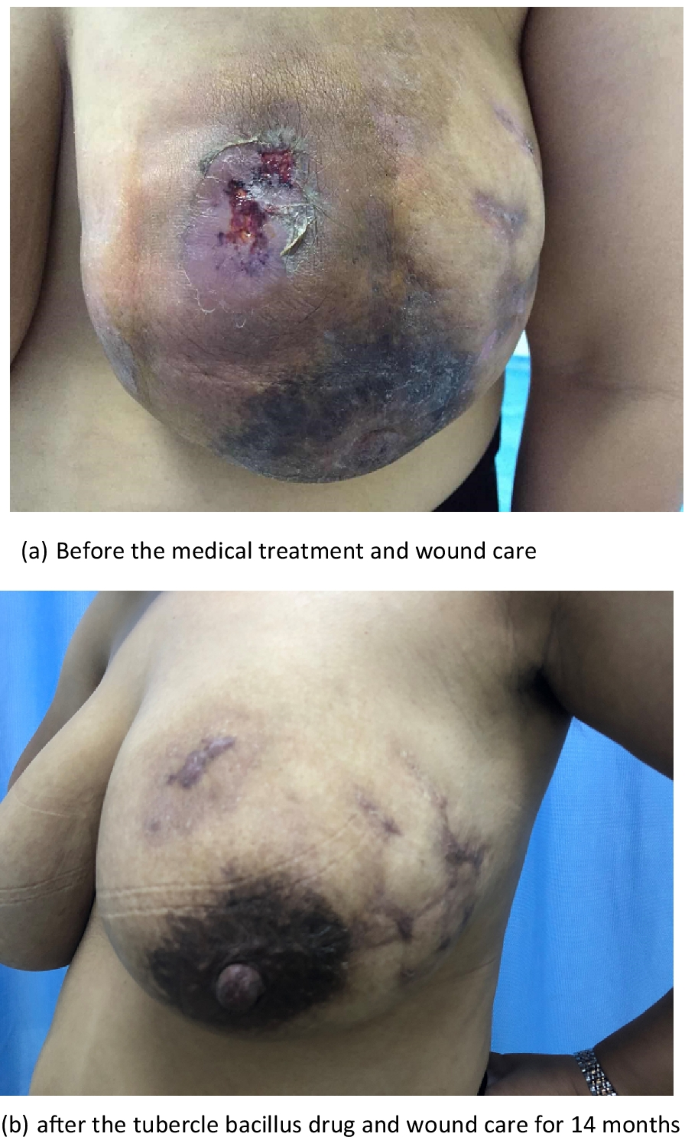
The effect of medical treatment in the case management. a Before the medical treatment and wound care. b After the tubercle bacillus drug and wound care for 14 months
Comparing the most recent publications on GM to older studies, there is no new information on this benign breast disease. Therefore, the best management of this disease is still unclear [ 11 , 12 ]. The usual treatment for GM is close observation, medical treatment, surgical management, or a combination of medication and surgery [ 3 , 15 , 44 ]. In the present study, only 1 (0.7%) patient recovered under observation, 82 (53.9%) recovered with medication and surgery (as shown in Fig. 2 ), and 69 (45.4%) recovered with medication alone (as shown in Fig. 3 ). Multivariate analysis revealed that medication and surgery was significantly associated with recurrence (RR = 4.128, 95% CI [1.026–16.610], P = 0.0046) (Table 6 ). Regarding the cause of recurrence, previous studies have ascribed the incompleteness of excision to the failure of surgical treatment, or inconsistent follow-up times. In this study, case managers assessed changes in the size of the breast mass and the proportion of the mass to the breast size and considered whether the patients could undergo surgical excision with minimal impact on the aesthetics of the breast. Breast lesion excision by minimally invasive surgery or open surgery was applied, which may have a risk of incomplete surgical excision. Akcan et al. and Yabanoğlu et al. reported that complete excision of the breast lesion or wide excision with or without medication achieved low recurrence rates [ 38 , 45 ]; however, it is possible to cause damage to the breast due to the excessive removal of tissues. Therefore, the biggest problem with surgical treatment is the contradiction between the surgical effect and the postoperative aesthetic effect. Whether the surgical procedure that is chosen which increases the recurrence rate of GM requires further investigation.
Our study demonstrated that medical treatment is the most prevalent treatment, regardless of whether it is coupled with surgical treatment. Drug therapies have numerous side effects, such as Cushion's syndrome, skin rash, abnormal liver enzymes and abnormal uric acid and [ 46 ]. In our study, 8 (5.3%) patients suffered from skin rash, 11 (7.2%) had abnormal liver enzymes, 3 (2.0%) had abnormal uric acid, and 1 (0.7%) had edema on the lips and face (as shown in Table 3 ). In this stage, case managers served as a treatment team by linking physicians, pharmacists, dermatologists, obstetricians, and general practitioners. They immediately communicated with the multidisciplinary team, and then guided patients regarding their medications, and finally, most of the side effects disappeared within 1 week.
To the best of our knowledge, there are no studies investigating medication adherence in GM patients. In our study, it shown that the MMAS-8 was completed by 154 patients, with 39% who had high adherence, 46.4% who had medium adherence, and 14.6% who had low adherence. As a result of case manager guidance, the “low” medication adherence rate of GM patients was much lower than that of 30% and 50% of reported for adults with chronic disease [ 47 , 48 ]. Furthermore, “high” medication adherence (RR = 0.428, 95% CI 0.224–0.867, P = 0.015) at 2 months after initial medication use was significantly associated with a lower rate of recurrence in multivariate analysis. At the initial stage, the case managers paid more attention to the changes in the patients’ breast symptoms than to patient medication adherence, and the guidance and supervision of medical staff to patient medication need improvement. Currently, several reports have demonstrated the importance of regular visits to a physician, adequate patient contact time in clinical practice, and patient education to improve medication adherence to treatment [ 49 , 50 ].
Recent evidence indicates that the occurrence and recurrence of GM is associated with the Corynebacterium species, especially Corynebacterium kroppenstedtii [ 39 , 51 ]. In our study, samples of C. kroppenstedtii were obtained by ultrasound guidance for the puncture or biopsy of breast abscesses or hypoechoic masses. Breast pus or tissues were used for bacterial culture, and the positive rate of C. kroppenstedti was only 23.69% (36/152). In different studies, the positive rate of C. kroppenstedtii varies considerably, mainly due to the detection techniques. Li et al. [ 52 ] reported that nanopore sequencing showed accurate C. kroppenstedti detection over the culture method in GM patients. Therefore, the need to improve detection techniques for the Corynebacterium species will facilitate the study of the relationship between GM and bacteria.
In this study, the results showed that 22 (14.47%) patients had excitant food before the onset of GM. The recent literature reports that bacterial interactions have been confirmed between the breast and gut [ 53 , 54 ]. Li et al. hypothesized that imbalances among the external environment, host, and microbiota lead to the occurrence of GM as follows: External factors disturb the balance between the immune microenvironment and breast flora and induce the release of inflammatory factors and milk secretion, resulting in damage to the mammary epithelium. The positive feedback between the immune and inflammatory reactions eventually induces GM [ 13 ]. The consumption of stimulating foods may disrupt the intestinal flora and induce inflammation. Therefore, patients with GM should be given information regarding disease considerations related to diet, sleep, behaviors, etc., as shown in Table 1 .
Our study has several limitations. First, it cannot be confirmed whether interesting factors such as dietary and lifestyle habits are related to the occurrence and recurrence of GM. Second, the effects of this case management model cannot be assessed by this study. Therefore, there are several directions for our next work, including developing targeted strategies based on the case management model and exploring the effectiveness of this model in GM patients.
In conclusion, this study identified some factors associated with the recurrence of the disease under a case management model. “Low” medication adherence was a significant risk factor for the recurrence of granulomatous mastitis. The patients treated with medication and surgery did not have a reduced recurrence rate compared to those treated with medication alone.
Availability of data and materials
The datasets generated and/or analyzed during the current study are not publicly available due to restrictions related to confidentiality i.e., they contain information that could compromise the privacy of research participants, but are available from the corresponding author on reasonable request.
Kessler E, Wolloch Y. Granulomatous mastitis: a lesion clinically simulating carcinoma. Am J Clin Pathol. 1972;58(6):642–6. https://doi.org/10.1093/ajcp/58.6.642 .
Article CAS PubMed Google Scholar
Baslaim MM, Khayat HA, Al-Amoudi SA. Idiopathic granulomatous mastitis: a heterogeneous disease with variable clinical presentation. World J Surg. 2007;31(8):1677–81. https://doi.org/10.1007/s00268-007-9116-1 .
Article PubMed Google Scholar
Li J. Diagnosis and treatment of 75 patients with idiopathic lobular granulomatous mastitis. J Investig Surg. 2019;32(5):414–20. https://doi.org/10.1080/08941939.2018.1424270 .
Article Google Scholar
Steuer AB, Stern MJ, Cobos G, Castilla C, Joseph KA, Pomeranz MK, Femia AN. Clinical characteristics and medical management of idiopathic granulomatous mastitis. JAMA Dermatol. 2020;156(4):460–4. https://doi.org/10.1001/jamadermatol.2019.4516 .
Article PubMed PubMed Central Google Scholar
Lai EC, Chan WC, Ma TK, Tang AP, Poon CS, Leong HT. The role of conservative treatment in idiopathic granulomatous mastitis. Breast J. 2005;11(6):454–6. https://doi.org/10.1111/j.1075-122X.2005.00127.x .
Wang Qi, Yu H. Precision diagnosis and treatment of granulomatous mastitis. Chin J Breast Dis (Electron Vers). 2017;03:129–31.
Google Scholar
Wang J, Xu H, Li Z, Li F, Yang Y, Yu X, Jiang D, Xing L, Sun H, Shao M. Pathogens in patients with granulomatous lobular mastitis. Int J Infect Dis. 2019;81:123–7. https://doi.org/10.1016/j.ijid.2019.01.034 .
Centers for Disease Control and Prevention (CDC). Idiopathic granulomatous mastitis in Hispanic women–Indiana, 2006–2008. MMWR Morb Mortal Wkly Rep. 2009;58(47):1317–21.
Al-Khaffaf B, Knox F, Bundred NJ. Idiopathic granulomatous mastitis: a 25-year experience. J Am Coll Surg. 2008;206(2):269–73. https://doi.org/10.1016/j.jamcollsurg.2007.07.041 .
Aghajanzadeh M, Hassanzadeh R, Alizadeh Sefat S, Alavi A, Hemmati H, et al. Granulomatous mastitis: Presentations, diagnosis, treatment and outcome in 206 patients from the north of Iran. Breast. 2015;24(4):456–60. https://doi.org/10.1016/j.breast.2015.04.003 .
Klipfel N, Grant E, Iyengar G. Granulomatous lobular mastitis: imaging, diagnosis, and treatment. AJR Am J Roentgenol. 2009;193(2):574–81. https://doi.org/10.2214/AJR.08.1528 .
Lin CH, Hsu CW, Tsao TY, Chou J. Idiopathic granulomatous mastitis associated with risperidone-induced hyperprolactinemia. Diagn Pathol. 2012;5(7):2. https://doi.org/10.1186/1746-1596-7-2 .
Article CAS Google Scholar
Barreto DS, Sedgwick EL, Nagi CS, Benveniste AP. Granulomatous mastitis: etiology, imaging, pathology, treatment, and clinical findings. Breast Cancer Res Treat. 2018;171(3):527–34. https://doi.org/10.1007/s10549-018-4870-3 .
Helal TE, Shash LS, Saad El-Din SA, Saber SM. Idiopathic granulomatous mastitis: cytologic and histologic study of 65 Egyptian patients. Acta Cytol. 2016;60(5):438–44. https://doi.org/10.1159/000448800 .
Sheybani F, Naderi HR, Gharib M, Sarvghad M, Mirfeizi Z. Idiopathic granulomatous mastitis: long-discussed but yet-to-be-known. Autoimmunity. 2016;49(4):236–9. https://doi.org/10.3109/08916934.2016.1138221 .
Al Manasra AR, Al-Hurani MF. Granulomatous mastitis: a rare cause of male breast lump. Case Rep Oncol. 2016;9(2):516–9. https://doi.org/10.1159/000448990 .
Velidedeoglu M, Kilic F, Mete B, Yemisen M, Celik V, Gazioglu E, Ferahman M, Ozaras R, Yilmaz MH, Aydogan F. Bilateral idiopathic granulomatous mastitis. Asian J Surg. 2016;39(1):12–20. https://doi.org/10.1016/j.asjsur.2015.02.003 .
Akbulut S, Yilmaz D, Bakir S. Methotrexate in the management of idiopathic granulomatous mastitis: review of 108 published cases and report of four cases. Breast J. 2011;17(6):661–8. https://doi.org/10.1111/j.1524-4741.2011.01162.x .
Hur SM, Cho DH, Lee SK, Choi MY, Bae SY, Koo MY, Kim S, Choe JH, Kim JH, Kim JS, Nam SJ, Yang JH, Lee JE. Experience of treatment of patients with granulomatous lobular mastitis. J Korean Surg Soc. 2013;85(1):1–6. https://doi.org/10.4174/jkss.2013.85.1.1 .
Mahlab-Guri K, Asher I, Allweis T, Diment J, Sthoeger ZM, Mavor E. Granulomatous lobular mastitis. Isr Med Assoc J. 2015;17(8):476–80.
PubMed Google Scholar
Beilman JP, Sowell RL, Knox M, Phillips KD. Case management at what expense? A case study of the emotional costs of case management. Nurs Case Manag. 1998;3(2):89–95.
CAS PubMed Google Scholar
Xing W, Huang J, Lu Z, et al. The effect of case management nursing practice on improving the quality of life of breast cancer patients. Chin J Nurs. 2011;4(6):1053–6.
Hisashige A. The effectiveness and efficiency of disease management programs for patients with chronic diseases. Glob J Health Sci. 2012;5(2):27–48. https://doi.org/10.5539/gjhs.v5n2p27 .
Tokem Y, Argon G, Keser G. Case management in care of Turkish rheumatoid arthritis patients. Rehabil Nurs. 2011;36(5):205–13. https://doi.org/10.1002/j.2048-7940.2011.tb00197.x .
Hubbard G, Gray NM, Ayansina D, Evans JM, Kyle RG. Case management vocational rehabilitation for women with breast cancer after surgery: a feasibility study incorporating a pilot randomised controlled trial. Trials. 2013;14:175. https://doi.org/10.1186/1745-6215-14-175 .
Scherz N, Bachmann-Mettler I, Chmiel C, Senn O, Boss N, Bardheci K, Rosemann T. Case management to increase quality of life after cancer treatment: a randomized controlled trial. BMC Cancer. 2017;17(1):223. https://doi.org/10.1186/s12885-017-3213-9 .
Lamb GS. Conceptual and methodological issues in nurse case management research. ANS Adv Nurs Sci. 1992;15(2):16–24. https://doi.org/10.1097/00012272-199212000-00004 .
Morisky DE, Ang A, Krousel-Wood M, Ward HJ. Predictive validity of a medication adherence measure in an outpatient setting. J Clin Hypertens (Greenwich). 2008;10(5):348–54. https://doi.org/10.1111/j.1751-7176.2008.07572.x .
Benson JR, Dumitru D. Idiopathic granulomatous mastitis: presentation, investigation and management. Future Oncol. 2016;12(11):1381–94. https://doi.org/10.2217/fon-2015-0038 .
Ozel L, Unal A, Unal E, Kara M, Erdoğdu E, Krand O, Güneş P, Karagül H, Demiral S, Titiz MI. Granulomatous mastitis: is it an autoimmune disease? Diagnostic and therapeutic dilemmas. Surg Today. 2012;42(8):729–33. https://doi.org/10.1007/s00595-011-0046-z .
Kaviani A, Vasigh M, Omranipour R, Mahmoudzadeh H, Elahi A, Farivar L, Zand S. Idiopathic granulomatous mastitis: looking for the most effective therapy with the least side effects according to the severity of the disease in 374 patients in Iran. Breast J. 2019;25(4):672–7. https://doi.org/10.1111/tbj.13300 .
Chen L, Chungee Z, Liya S, Rong C, Jian Wu. Current status and pharmacological monitoring of long-term oral glucocorticoid patients in rheumatology department. Chin J Hosp Pharm. 2020;24(09):1020–6. https://doi.org/10.13286/j.1001-5213.2020.09.13 .
Wu F, Zhao JX, Wang TS, Shao H, Shi LW. Reliability analysis of Chinese version of MMAS-8 to measure medication adherence in patients with rheumatoid arthritis. Chin Pharm. 2018;6(02):263–8. https://doi.org/10.6039/j.issn.1001-0408.2018.02.28 .
Lei X, Chen K, Zhu L, Song E, Su F, Li S. Treatments for idiopathic granulomatous mastitis: systematic review and meta-analysis. Breastfeed Med. 2017;12(7):415–21. https://doi.org/10.1089/bfm.2017.0030 .
Freeman CM, Xia BT, Wilson GC, Lewis JD, Khan S, Lee SJ, Lower EE, Edwards MJ, Shaughnessy EA. Idiopathic granulomatous mastitis: a diagnostic and therapeutic challenge. Am J Surg. 2017;214(4):701–6. https://doi.org/10.1016/j.amjsurg.2017.07.002 .
Keller K, Meisel C, Petzold A, Wimberger P. Granulomatöse Mastitis – möglicher diagnostischer und therapeutischer Ablauf anhand von Fallbeispielen. Senologie. 2018;15: e23.
DeHertogh DA, Rossof AH, Harris AA, Economou SG. Prednisone management of granulomatous mastitis. N Engl J Med. 1980;303(14):799–800. https://doi.org/10.1056/NEJM198010023031406 .
Akcan A, Öz AB, Dogan S, et al. Idiopathic granulomatous mastitis: comparison of wide local excision with or without corticosteroid therapy. Breast Care. 2014;9:111–5.
Tan QT, Tay SP, Gudi MA, Nadkarni NV, Lim SH, Chuwa EWL. Granulomatous mastitis and factors associated with recurrence: an 11-year single-centre study of 113 patients in Singapore. World J Surg. 2019;43(7):1737–45. https://doi.org/10.1007/s00268-019-05014-x .
Prasad S, Jaiprakash P, Dave A, Pai D. Idiopathic granulomatous mastitis: an institutional experience. Turk J Surg. 2017;33(2):100–3. https://doi.org/10.5152/turkjsurg.2017.3439 .
Agrawal A, Pabolu S. A rare case of idiopathic granulomatous mastitis in a nulliparous woman with hyperprolactinemia. Cureus. 2019;11(5): e4680. https://doi.org/10.7759/cureus.4680 .
Gupta N, Vats M, Garg M, Dahiya DS. Bilateral idiopathic granulomatous mastitis. BMJ Case Rep. 2020;13(8): e234979. https://doi.org/10.1136/bcr-2020-234979 .
Wang J, Zhang Y, Lu X, Xi C, Yu K, Gao R, Bi K. Idiopathic granulomatous mastitis with skin rupture: a retrospective cohort study of 200 patients who underwent surgical and nonsurgical treatment. J Investig Surg. 2019. https://doi.org/10.1080/08941939.2019.1696905 .
Gunduz Y, Altintoprak F, Tatli Ayhan L, Kivilcim T, Celebi F. Effect of topical steroid treatment on idiopathic granulomatous mastitis: clinical and radiologic evaluation. Breast J. 2014;20(6):586–91.
Yabanoğlu H, Çolakoğlu T, Belli S, Aytac HO, Bolat FA, Pourbagher A, et al. A comparative study of conservative versus surgical treatment protocols for 77 patients with idiopathic granulomatous mastitis. Breast J. 2015;21(4):363–9.
Kehribar DY, Duran TI, Polat AK, Ozgen M. Effectiveness of methotrexate in idiopathic granulomatous mastitis treatment. Am J Med Sci. 2020;360(5):560–5. https://doi.org/10.1016/j.amjms.2020.05.029 .
Briesacher BA, Andrade SE, Fouayzi H, Chan KA. Comparison of drug adherence rates among patients with seven different medical conditions. Pharmacotherapy. 2008;28(4):437–43. https://doi.org/10.1592/phco.28.4.437 .
Naderi SH, Bestwick JP, Wald DS. Adherence to drugs that prevent cardiovascular disease: meta-analysis on 376,162 patients. Am J Med. 2012;125(9):882–7. https://doi.org/10.1016/j.amjmed.2011.12.013 .
Gadallah MA, Boulos DN, Gebrel A, Dewedar S, Morisky DE. Assessment of rheumatoid arthritis patients’ adherence to treatment. Am J Med Sci. 2015;349(2):151–6. https://doi.org/10.1097/MAJ.0000000000000376 .
Marengo MF, Suarez-Almazor ME. Improving treatment adherence in patients with rheumatoid arthritis: what are the options? Int J Clin Rheumtol. 2015;10(5):345–56. https://doi.org/10.2217/ijr.15.39 .
Article CAS PubMed PubMed Central Google Scholar
Johnson MG, Leal S, Plongla R, Leone PA, Gilligan PH. The brief case: recurrent granulomatous mastitis due to Corynebacterium kroppenstedtii . J Clin Microbiol. 2016;54(8):1938–41. https://doi.org/10.1128/JCM.03131-15 .
Li XQ, Yuan JP, Fu AS, Wu HL, Liu R, Liu TG, Sun SR, Chen C. New insights of Corynebacterium kroppenstedtii in granulomatous lobular mastitis based on nanopore sequencing. J Investig Surg. 2022;35(3):639–46. https://doi.org/10.1080/08941939.2021.1921082 .
Li XQ, Wu HL, Yuan JP, Liu TG, Sun SR, Chen C. Bacteria associated with granulomatous lobular mastitis and the potential for personalized therapy. J Investig Surg. 2022;35(1):164–70. https://doi.org/10.1080/08941939.2020.1833262 .
Hu X, Li S, Fu Y, Zhang N. Targeting gut microbiota as a possible therapy for mastitis. Eur J Clin Microbiol Infect Dis. 2019;38(8):1409–23. https://doi.org/10.1007/s10096-019-03549-4 .
Download references
Acknowledgements
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Project of The Health Planning Committee of Sichuan [Grant No. 21PJ134].
Author information
Authors and affiliations.
Department of Breast, Chengdu Women’s and Children’s Central Hospital, School of Medicine, University of Electronic Science and Technology of China, No. 1617, Riyue Avenue, Qingyang District, Chengdu, 611731, Sichuan Province, People’s Republic of China
Yuan Deng, Ying Xiong, Ping Ning, Xiao-Rong Han & Pei-Yu He
Department of Nursing, Chengdu Women’s and Children’s Central Hospital, School of Medicine, University of Electronic Science and Technology of China, No. 1617, Riyue Avenue, Qingyang District, Chengdu, 611731, Sichuan Province, People’s Republic of China
Yuan Deng, Xin Wang & Guo-Fang Tu
You can also search for this author in PubMed Google Scholar
Contributions
PN, X-RH, and XW conceived and designed the study. YD, YX, and P-YH collected and analyzed the data. YD and YX drafted the paper. PN, XW, and G-FT read and revised the draft critically. YD and YX contributed equally to this work. All authors reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Correspondence to Ping Ning or Xin Wang .
Ethics declarations
Ethics approval and consent to participate.
This study was approved by the Institution Review Board of Chengdu Women and Children’s Central Hospital Ethical approval (Grant No. B2019 (13)). All participants signed an informed consent form before data collection. All procedures performed in this study were in accordance with the ethical standards.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Deng, Y., Xiong, Y., Ning, P. et al. A case management model for patients with granulomatous mastitis: a prospective study. BMC Women's Health 22 , 143 (2022). https://doi.org/10.1186/s12905-022-01726-w
Download citation
Received : 17 January 2022
Accepted : 20 April 2022
Published : 02 May 2022
DOI : https://doi.org/10.1186/s12905-022-01726-w
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Granulomatous mastitis
- Case management
- Medication adherence
BMC Women's Health
ISSN: 1472-6874
- General enquiries: [email protected]

ERIN F. MORCOMB, MD, CARMEN M. DARGEL, MD, AND SYDNEY A. ANDERSON, DO
Am Fam Physician. 2024;110(2):174-182
Related editorial: Supporting the Paradigm Shift: Watchful Waiting for Lactational Mastitis
Author disclosure: No relevant financial relationships.
Mastitis represents a spectrum of inflammatory conditions. Lactational mastitis is the most common, with an approximate incidence of 10% in the United States, and it usually occurs in the first 3 months postpartum. Diagnosis is made clinically based on the presence of symptoms such as fever, malaise, focal breast tenderness, and overlying skin erythema or hyperpigmentation without the need for laboratory tests or imaging. However, obtaining milk cultures should be considered to guide antibiotic therapy, and ultrasonography should be performed to identify abscesses in immuno-compromised patients or those with worsening or recurrent symptoms. Because most cases of mastitis are caused by inflammation and not a true infection, a 1- to 2-day trial of conservative measures (i.e., nonsteroidal anti-inflammatory drugs, ice application, feeding the infant directly from the breast, and minimizing pumping) is often sufficient for treatment. If there is no improvement in symptoms, narrow-spectrum antibiotics may be prescribed to cover common skin flora (e.g., Staphylococcus , Streptococcus ). Most patients can be treated as outpatients with oral antibiotics; however, if the condition worsens or there is a concern for sepsis, intravenous antibiotics and hospital admission may be required. Use of probiotics for treatment or prevention is not supported by good evidence. Factors that increase the risk of mastitis include overstimulation of milk production and tissue trauma from aggressive breast massage; therefore, frequent overfeeding, excessive pumping to empty the breast, heat application, and breast massage are no longer recommended because they may worsen the condition. The best prevention is a proper lactation technique, including a good infant latch, and encouraging physiologic breastfeeding rather than pumping, if possible.
- Immediate, unlimited access to all AFP content
- More than 130 CME credits/year
- AAFP app access
- Print delivery available
Issue Access
- Immediate, unlimited access to this issue's content
- CME credits
Article Only
- Immediate, unlimited access to just this article
- Blackmon MM, et al. Acute mastitis. StatPearls. Updated July 21, 2023. Accessed December 1, 2023. https://www.ncbi.nlm.nih.gov/books/NBK557782/
Mitchell KB, et al. Academy of Breastfeeding Medicine clinical protocol #36: the mastitis spectrum, revised 2022 [published correction appears in Breastfeed Med . 2022; 17(11): 977–978]. Breastfeed Med. 2022;17(5):360-376.
Spencer JP. Management of mastitis in breastfeeding women. Am Fam Physician. 2008;78(6):727-731.
Koziol KJ, et al. Predictive risk factors for childbirth-associated breast infections in the United States: a 10-year perspective. Int J Environ Res Public Health. 2023;20(14):6333.
Lin CH, et al. Descriptive study of mastitis in postpartum women in Taiwan: incidence and related factors. J Womens Health (Larchmt). 2023;32(5):616-622.
Louis-Jacques AF, et al. Risk factors, symptoms, and treatment of lactational mastitis. JAMA. 2023;329(7):588-589.
Mitoulas LR, et al. Breast pumps and mastitis in breastfeeding women: clarifying the relationship. Front Pediatr. 2022;10:856353.
Grzeskowiak LE, et al. Incidence, antibiotic treatment and outcomes of lactational mastitis: findings from the Norwegian Mother, Father and Child Cohort Study (MoBa). Paediatr Perinat Epidemiol. 2022;36(2):254-263.
Wilson E, et al. Incidence of and risk factors for lactational mastitis. J Hum Lact. 2020;36(4):673-686.
Lai BY, et al. Risk factors for lactation mastitis in China. PLoS One. 2021;16(5):e0251182.
Westerfield KL, et al. Breastfeeding: common questions and answers. Am Fam Physician. 2018;98(6):368-373.
Couvillion SP, et al. Interrogating the role of the milk microbiome in mastitis in the multi-omics era. Front Microbiol. 2023;14:1105675.
Castro I, et al. Interactions between human milk oligosaccharides, microbiota and immune factors in milk of women with and without mastitis. Sci Rep. 2022;12(1):1367.
Deng Y, et al. Maternal risk factors for lactation mastitis. West J Nurs Res. 2021;43(7):698-708.
Johnson HM, et al. Academy of Breastfeeding Medicine clinical protocol #32: management of hyperlactation. Breastfeed Med. 2020;15(3):129-134.
Oikonomou G, et al. Milk microbiota: what are we exactly talking about?. Front Microbiol. 2020;11:60.
Mastitis: what you should know. Am Fam Physician. 2008;78(6):732.
Afeiche MC, et al. The Dietary Inflammatory Index is associated with subclinical mastitis in lactating European women. Nutrients. 2022;14(22):4719.
Li D, et al. Risk factors and prognosis of acute lactation mastitis developing into a breast abscess. PLoS One. 2022;17(9):e0273967.
Douglas P. Re-thinking benign inflammation of the lactating breast: classification, prevention, and management [published correction appears in Womens Health (Lond) . 2023; 19: 17455057231157916]. Womens Health (Lond). 2022;18:17455057221091349.
Anderson L, et al. Effectiveness of breast massage for the treatment of women with breastfeeding problems. JBI Database System Rev Implement Rep. 2019;17(8):1668-1694.
Witt AM, et al. Therapeutic breast massage in lactation for the management of engorgement, plugged ducts, and mastitis. J Hum Lact. 2016;32(1):123-131.
Mitchell KB, et al. Management of common complications of lactation: the breast surgeon's role in examining the science and debunking old myths. Surg Clin North Am. 2022;102(6):973-987.
Betts RC, et al. It's not yeast: retrospective cohort study of lactating women with persistent nipple and breast pain. Breastfeed Med. 2021;16(4):318-324.
Amir LH, et al. Identifying the cause of breast and nipple pain during lactation. BMJ. 2021;374:n1628.
Pandey TS, et al. Idiopathic granulomatous mastitis—a prospective study of 49 women and treatment outcomes with steroid therapy. Breast J. 2014;20(3):258-266.
Chang CM, et al. Risk of breast cancer in women with non-lactational mastitis. Sci Rep. 2019;9(1):15587.
Schwarz J, et al. Case report of recurrent bilateral mastitis in a woman who is exclusively pumping breast milk for an infant in the NICU. J Obstet Gynecol Neonatal Nurs. 2021;50(6):765-773.
Crepinsek MA, et al. Interventions for preventing mastitis after childbirth. Cochrane Database Syst Rev. 2020(9):CD007239.
Dagla M, et al. Do maternal stress and depressive symptoms in perinatal period predict the lactation mastitis occurrence? A retrospective longitudinal study in Greek women. Diagnostics (Basel). 2021;11(9):1524.
Yu Q, et al. The preventive and therapeutic effects of probiotics on mastitis. PLoS One. 2022;17(9):e0274467.
Zhang Y, et al. Oral Lactobacillus fermentum CECT5716 in the patients with lactational abscess treated by needle aspiration. Medicine (Baltimore). 2022;101(26):e29761.
Barker M, et al. Probiotics and human lactational mastitis. Women Birth. 2020;33(6):e483-e491.
Zhang Y, et al. Breast microecology improvement using probiotics following needle aspiration in patients with lactational breast abscess. Sci Rep. 2022;12(1):16692.
Amir LH, et al. Probiotics and mastitis: evidence-based marketing?. Int Breastfeed J. 2016;11:19.
Arroyo R, et al. Treatment of infectious mastitis during lactation: antibiotics versus oral administration of Lactobacilli isolated from breast milk. Clin Infect Dis. 2010;50(12):1551-1558.
Lai ECH, et al. The role of conservative treatment in idiopathic granulomatous mastitis. Breast J. 2005;11(6):454-456.
Konishi T, et al. Impact of the National Action Plan for Antimicrobial Resistance on antibiotic use for mastitis using a Japanese nationwide database. Breast Care (Basel). 2023;18(2):122-129.
Mitchell KB, et al. Challenges in the management of breast conditions during lactation. Obstet Gynecol Clin North Am. 2022;49(1):35-55.
Abdelhadi MSA, et al. Breast infections in non-lactating women. J Family Community Med. 2005;12(3):133-137.
Martinez-Ramos D, et al. Idiopathic granulomatous mastitis. Breast J. 2019;25(6):1245-1250.
Hovanessian Larsen LJ, et al. Granulomatous lobular mastitis: imaging, diagnosis, and treatment. AJR Am J Roentgenol. 2009;193(2):574-581.
Jahanfar S, et al. Antibiotics for mastitis in breastfeeding women. Cochrane Database Syst Rev. 2013(2):CD005458.
Long T, et al. Maintaining breastfeeding during the treatment of breast abscesses is not contraindicated. Breastfeed Med. 2022;17(9):753-757.
Johnson HM, et al. Low incidence of milk fistula with continued breastfeeding following radiologic and surgical interventions on the lactating breast. Breast Dis. 2021;40(3):183-189.
Zakarija-Grkovic I, et al. Treatments for breast engorgement during lactation. Cochrane Database Syst Rev. 2020(9):CD006946.
Continue Reading

More in AFP
More in pubmed.
Copyright © 2024 by the American Academy of Family Physicians.
This content is owned by the AAFP. A person viewing it online may make one printout of the material and may use that printout only for his or her personal, non-commercial reference. This material may not otherwise be downloaded, copied, printed, stored, transmitted or reproduced in any medium, whether now known or later invented, except as authorized in writing by the AAFP. See permissions for copyright questions and/or permission requests.
Copyright © 2024 American Academy of Family Physicians. All Rights Reserved.
When viewing this topic in a different language, you may notice some differences in the way the content is structured, but it still reflects the latest evidence-based guidance.
Mastitis and breast abscess
- Overview
- Theory
- Diagnosis
- Management
- Follow up
- Resources
Breast infections (including infectious mastitis and breast abscess) more commonly affect women aged 15-45 years, especially those who are lactating. However, mastitis and breast abscess can occur at any age.
Staphylococcus aureus is the most frequent pathogen isolated.
Prompt and appropriate management of mastitis usually leads to a timely resolution and prevents complications, such as a breast abscess.
Breast abscess requires both the removal of pus and antibiotic therapy. Interventions can include aspiration and incision and drainage procedures.
It is imperative to identify and treat any underlying co-existent causes of infection to facilitate resolution and prevent recurrence. It is also necessary to exclude breast carcinoma.
Mastitis is defined as inflammation of the breast with or without infection. Mastitis with infection may be lactational (puerperal) or non-lactational (e.g., duct ectasia). Non-infectious mastitis includes idiopathic granulomatous inflammation and other inflammatory conditions (e.g., foreign body reaction). A breast abscess is a localised area of infection with a walled-off collection of purulence. It may or may not be associated with mastitis.
This topic covers mastitis in adults. Neonates and infants with suspected mastitis should be referred to a paediatric consultant for evaluation and management.
History and exam
Key diagnostic factors.
- decreased milk outflow
- breast warmth
- breast tenderness
- breast firmness
- breast swelling
- breast erythema
- flu-like symptoms, malaise, and myalgia
- breast pain
- breast mass
Other diagnostic factors
- nipple discharge
- nipple inversion/retraction
- lymphadenopathy
- extra-mammary skin lesions
Risk factors
- poor breastfeeding technique
- milk stasis
- nipple injury
- previous mastitis
- prolonged mastitis (breast abscess)
- women aged >30 years (breast abscess)
- prior breast abscess (breast abscess)
- shaving or plucking areola hair
- anatomical breast defect, mammoplasty, or scar
- other underlying breast condition
- nipple piercing
- foreign body
- skin infection
- Staphylococcus aureus carrier
- immunosuppression
- hospital admission
- breast trauma
- primiparity (breast abscess)
- multiparity
- overabundant milk supply
- complications of delivery
- maternal stress
- tight clothing
- antifungal nipple cream
- fibrocystic breast disease
- cigarette smoking
- vaginal manipulation (breast abscess)
- antiretroviral therapy
Diagnostic investigations
1st investigations to order.
- breast ultrasound
- diagnostic needle aspiration drainage
- cytology of nipple discharge or sample from fine-needle aspiration
- milk, aspirate, discharge, or biopsy tissue for culture and sensitivity
Investigations to consider
- pregnancy test
- blood culture and sensitivity
- histopathological examination of biopsy tissue
- milk for leukocyte counts and bacteria quantification
Treatment algorithm
Lactational mastitis, non-lactational mastitis, breast abscess, breast abscess post acute intervention, recurrence of mastitis and/or breast abscess, contributors, jesse casaubon, do, fsso, facs.
Breast Surgical Oncologist
Baystate Health
Springfield
Disclosures
JC declares that he has no competing interests.
Acknowledgements
Dr Jesse Casaubon would like to gratefully acknowledge Dr Holly S. Mason, Dr Jose A. Martagon-Villamil, Dr Daniel Skiest, Dr Gina Berthold, and Dr Liron Pantanowitz, previous contributors to this topic.
HSM, JAMV, DS, and GB declare that they have no competing interests. LP is a co-author of references cited in this topic.
Peer reviewers
Edward sauter, md, phd.
Program Officer
National Institutes of Health
National Cancer Institute
ES declares that he has no competing interests.
Justin Stebbing, MA, MRCP, MRCPath, PhD
Consultant Medical Oncologist/Senior Lecturer
Department of Medical Oncology
Imperial College/Imperial Healthcare NHS Trust
Charing Cross Hospital
JS declares that he has no competing interests.
William C. Dooley, MD
The G. Rainey Williams Professor of Surgical Oncology
University of Oklahoma
Oklahoma City
WD declares that he has no competing interests.

Differentials
- Breast engorgement
- Nipple sensitivity
- Galactocele
- ABM clinical protocol #36: the mastitis spectrum
- Clinical practice guideline. Breastfeeding challenges: Mastitis and breast abscess management
Patient information
Mastitis in breastfeeding women
Mastitis: breastfeeding advice
Use of this content is subject to our disclaimer
Log in or subscribe to access all of BMJ Best Practice
Log in to access all of bmj best practice, help us improve bmj best practice.
Please complete all fields.
I have some feedback on:
We will respond to all feedback.
For any urgent enquiries please contact our customer services team who are ready to help with any problems.
Phone: +44 (0) 207 111 1105
Email: [email protected]
Your feedback has been submitted successfully.
- Case report
- Open access
- Published: 25 April 2013
A lactating woman presenting with puerperal pneumococcal mastitis: a case report
- Barbara Miedzybrodzki 1 &
- Mark Miller 2
Journal of Medical Case Reports volume 7 , Article number: 114 ( 2013 ) Cite this article
7255 Accesses
4 Citations
1 Altmetric
Metrics details
Introduction
Streptococcus pneumoniae is an uncommon etiologic agent in soft-tissue infections.
Case presentation
We report the case of a 35-year-old Caucasian woman who presented to our facility with puerperal pneumococcal mastitis, and review the only other three cases of pneumococcal mastitis described in the medical literature.
Conclusions
The roles of the various pneumococcal vaccines in preventing this disease are discussed.
Peer Review reports
Puerperal mastitis occurs most commonly during the first three to six months post-partum in breastfeeding mothers. Up to 25 percent of breastfeeding women have experienced at least one episode of mastitis, and recurrent mastitis has been reported in four to eight-and-a-half percent of breastfeeding women [ 1 ]. The most common causative organism of mastitis is Staphylococcus aureus . Other less common organisms include coagulase-negative staphylococci , beta-hemolytic streptococci (Lancefield groups A or B) , Escherichia coli , and Corynebacterium species [ 2 ]. Streptococcus pneumoniae is an extremely rare cause of mastitis. In this paper, we present the case of a healthy 35-year-old woman who presented to our facility with puerperal pneumococcal mastitis, and review the only three other cases of pneumococcal mastitis described in the medical literature.
A literature review using a MEDLINE search from 1950 to July 2010 revealed only two cases of puerperal mastitis and one case of non-puerperal mastitis caused by S. pneumoniae (Table 1 ). The first case of pneumococcal mastitis was described by DiNubile et al . in 1991 in a 23-year-old woman with systemic lupus erythematosus who was being treated with prednisone but was not lactating [ 3 ]. She presented with an abscess of the left breast and the aspirate revealed S. pneumoniae and Bacteroides fragilis. The second case, described by Wüst et al . in 1995, was in a healthy 38-year-old woman breastfeeding her nine-month-old daughter [ 4 ]. In that case, serotyping was performed on a nasal and throat swab taken from the child as well as from the breast. All three cultures revealed S. pneumoniae serotype 6B, which the authors reported as being the second most frequent type found in the region at that time. The third case was published by Kragsbjerg et al . in 1995, concerning a 38-year-old woman who presented with purulent secretions from the breast while she was breastfeeding her four-month-old child [ 5 ]. Cultures taken from the breast and from the nasopharynx of the child revealed the same serotype of S. pneumoniae.
A 35-year-old Caucasian woman who was breastfeeding her eight-month-old twins presented to our facility with a three-day history of fronto-parietal headache, fever, general malaise, and two episodes of syncope on the day of admission. On further questioning, she also reported increasing pain in her right breast over the last 24 hours.
She appeared toxic and was febrile (39.0°C axillary temperature). A physical examination revealed an exquisitely tender right breast that was erythematous and indurated in the right lower lateral quadrant. There was, however, no area of fluctuance although purulent milky secretions could be expelled from the right nipple with mild peri-areolar pressure. These secretions were cultured. Slightly tender right axillary adenopathy was also present.
The results of laboratory investigations were unremarkable, including a normal blood count, except for the presence of a left shift with 80 percent neutrophils (total white blood cell count of 9.8×10 9 cells/L). Several diagnostic investigations were performed, including a lumbar puncture, cerebral computed tomography (CT) and magnetic resonance imaging (MRI) scans, and blood cultures, all of which yielded normal results. A clinical diagnosis of puerperal mastitis was made, and treatment with intravenous vancomycin and cefazolin was initiated. Our patient continued pumping her breast milk. On the day after admission, increased amounts of pus were noted draining from the right nipple with each breast pumping. Our patient’s fever and rigors resolved within 48 hours. Culture of the breast secretions at the time of admission revealed heavy pure growth of S. pneumoniae , polysaccharide serotype 19A, which was susceptible to penicillin, cephalosporins, macrolides, tetracyclines and vancomycin. Her hospital course was uncomplicated and she was discharged home on day three post-admission with a 10-day course of oral cefadroxil. Neither of her babies showed any evidence of a respiratory tract infection prior to our patient’s illness; nasopharyngeal culture tests from the babies were not performed as they were at home with the father and unavailable for culture sampling.
Pneumococcal mastitis is an extremely rare entity and, to the best of our knowledge, there have been only three other case reports in the literature, two of which were puerperal. S. pneumoniae is a leading cause of respiratory tract infections and meningitis in both children and adults. It is, however, a rare cause of skin and soft-tissue infections and the cases reported are mostly described in patients who have some degree of immunosuppression [ 6 ]. Our patient, whose case we present here, was a healthy 35-year-old immunocompetent woman and there were no signs of any connective tissue diseases or other coincidental health issues.
Although neither of her babies showed any evidence of a respiratory tract infection prior to our patient’s illness, and testing of the babies was not undertaken due to their unavailability, it appears that the most probable way in which the mother became infected with S. pneumoniae serotype 19A was from one or both of the nasopharyngeal tracts of the babies during breastfeeding. In both of the previous case reports [ 4 , 5 ], the breastfed babies had tested positive on nasopharyngeal swabs and showed symptoms of mild respiratory tract infections, which is consistent with our interpretation of the mode of transmission of the S. pneumoniae in mastitis. Our patient's twin babies were both routinely vaccinated at two and four months of age with Prevnar-7® (Wyeth, Collegeville, PA, USA), which contains capsular antigens of S. pneumoniae serotypes 4, 6B, 9V, 14, 18C, 19F, and 23F. Thus, the serotype 19A S. pneumoniae isolated in our patient was not part of the seven-valent pneumococcal conjugate vaccine administered to children in the province of Quebec, where our patient resided at the time of her illness. Current Quebec immunization guidelines recommend vaccination of healthy babies with a pneumococcal seven-valent conjugate vaccine (Prevnar-7®) to be given in three doses administered at two, four and 12 months of age [ 7 ]. However, since the introduction of Prevnar-7®, there has been growing concern of the development and spread of the pneumococcal serotypes not covered in the vaccine. A recent review by Reinert et al . describes global indicators showing that serotype 19A is now the most prevalent as well as the most increasingly resistant S. pneumoniae serotype in invasive infections [ 8 ]. The most prevalent serotypes involved in invasive disease in Canada at the time of our patient’s presentation were (in descending order): 19A, 7F, 18C, 6A, 22F, 4, 5, 3 and 23B [ 9 ].
Given these findings, the new 13-valent vaccine (Prevnar-13®) that has recently been licensed in Canada, will likely reduce the increasingly prevalent infection rate from the 19A strain of S. pneumoniae . This new vaccine contains the same antigens as Prevnar-7® with six additional capsular antigens of serotypes 1, 3, 5, 6A, 7F and 19A [ 10 ], which together comprise 13 of the 91 S. pneumoniae serotypes described thus far [ 8 ].
This case report highlights the fact that puerperal mastitis may be caused by unusual bacteria, including S. pneumoniae. Immunization of babies with effective pneumococcal vaccines should decrease the incidence of pneumococcal puerperal infections even further, as well as other invasive pneumococcal infections that may be similarly transmitted from baby to mother.
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Foxman B, D'Arcy H, Gillespie B, Bobo JK, Schwartz K: Lactation mastitis: occurrence and medical management among 946 breastfeeding women in the United States. Am J Epidemiol. 2002, 155: 103-114. 10.1093/aje/155.2.103.
Article PubMed Google Scholar
World Health Organization: Mastitis: Causes and Management. WHO/FCH/CAH/00.13. 2000, Geneva, Switzerland: WHO
Google Scholar
DiNubile MJ, Albornoz A, Stumacher RJ, Van Uitert BL, Paluzzi SA, Bush LM, Nelson SL, Myers AR: Pneumococcal soft-tissue infections: possible association with connective tissue diseases. J Infect Dis. 1991, 163: 897-900. 10.1093/infdis/163.4.897.
Article CAS PubMed Google Scholar
Wüst J, Rutsch M, Stocker S: Streptococcus pneumoniae as an agent of mastitis. Eur J Clin Microbiol Infect Dis. 1995, 14: 156-157. 10.1007/BF02111883.
Kragsbjerg P, Noren T, Soderquist B: Deep soft-tissue infections caused by Streptococcus pneumoniae . Eur J Clin Microbiol Infect Dis. 1995, 14: 1002-1004. 10.1007/BF01691383.
Garcia-Lechuz JM, Cuevas O, Castellares C, Perez-Fernandez C, Cercenado E, Bouza E: Streptococcus pneumoniae skin and soft tissue infections: characterization of causative strains and clinical illness. Eur J Clin Microbiol Infect Dis. 2007, 26: 247-255. 10.1007/s10096-007-0283-7.
Public Health Aganecy of Canada: Publicly funded Immunization Programs in Canada - Routine Schedule for Infants and Children (including special programs and catch-up programs). http://www.phac-aspc.gc.ca/im/ptimprog-progimpt/table-1-eng.php ,
Reinert RR, Jacobs MR, Kaplan S: Pneumococcal disease caused by serotype 19A: review of the literature and implications for future vaccine development. Vaccine. 2010, 28: 4249-4259. 10.1016/j.vaccine.2010.04.020.
Griffith A, Demczuk W, Martin I, Shane A, Tyrrell G, Gilmour MW, the Canadian Public Health Laboratory Network: Distribution of invasive pneumococcal serotypes in Canada: 2010–2011. Abstract #K4. 2012, Vancouver, Canada: AMMI Canada - CACMID Annual Conference
Bryant KA, Block SL, Baker SA, Gruber WC, Scott DA, PCV13 Infant Study Group: Safety and immunogenicity of a 13-valent pneumococcal conjugate vaccine. Pediatrics. 2010, 125: 866-875. 10.1542/peds.2009-1405.
Download references
Acknowledgements
We would like to thank our patient for allowing use of her clinical and laboratory information for publication. No financial support has been given for this report.
Author information
Authors and affiliations.
Division of Internal Medicine, Jewish General Hospital, 3755 Cote-Ste-Catherine, Montreal, Quebec, H3T 1E2, Canada
Barbara Miedzybrodzki
Division of Infectious Diseases, Jewish General Hospital, 3755 Cote-Ste-Catherine, Montreal, Quebec, H3T 1E2, Canada
Mark Miller
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Mark Miller .
Additional information
Competing interests.
The authors declare that they have no competing interests.
Authors’ contributions
EM performed the literature review. Both authors collected, analyzed and interpreted the clinical and microbiologic data from our patient. Both authors wrote the manuscript and read and approved the final version.
Rights and permissions
This article is published under license to BioMed Central Ltd. This is an Open Access article distributed under the terms of the Creative Commons Attribution License ( http://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Reprints and permissions
About this article
Cite this article.
Miedzybrodzki, B., Miller, M. A lactating woman presenting with puerperal pneumococcal mastitis: a case report. J Med Case Reports 7 , 114 (2013). https://doi.org/10.1186/1752-1947-7-114
Download citation
Received : 25 November 2012
Accepted : 26 March 2013
Published : 25 April 2013
DOI : https://doi.org/10.1186/1752-1947-7-114
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Pneumococcal infection
- Puerperal mastitis
- Streptococcus pneumoniae
- Pneumococcal vaccine
Journal of Medical Case Reports
ISSN: 1752-1947
- Submission enquiries: Access here and click Contact Us
- General enquiries: [email protected]
- Case Report
- Open access
- Published: 15 September 2020
Case report: characterization of a persistent, treatment-resistant, novel Staphylococcus aureus infection causing chronic mastitis in a Holstein dairy cow
- Ellie J. Putz 1 , 2 ,
- Mitchell V. Palmer 1 ,
- Eduardo Casas 2 ,
- Timothy A. Reinhardt 2 &
- John D. Lippolis ORCID: orcid.org/0000-0003-2314-4384 2
BMC Veterinary Research volume 16 , Article number: 336 ( 2020 ) Cite this article
4048 Accesses
12 Citations
1 Altmetric
Metrics details
Mastitis is the most common health concern plaguing the modern dairy cow and costs dairy producers estimates of two billion dollars annually. Staphylococcus aureus infections are prevalent, displaying varied disease presentation and markedly low cure rates. Neutrophils are considered the first line of defense against mastitis causing bacteria and are frequently targeted in the development of treatment and prevention technologies. We describe a case of naturally occurring, chronic mastitis in a Holstein cow (1428), caused by a novel strain of S. aureus that was not able to be cleared by antibiotic treatment.
Case presentation
The infection was identified in a single quarter, 2 months into the cow’s first lactation. The infection persisted for the following 20 months, including through dry off, and a second calving and lactation. This case of mastitis was associated with a consistently high somatic cell count, however presented with no other clinical signs. This cow was unsuccessfully treated with antibiotics commonly used to treat mastitis, consisting of two rounds of treatment during lactation and an additional round at the beginning of dry off. The chronic infection was also unchanged through an experimental mid-lactation treatment with pegylated granulocyte-colony stimulating factor (PEG-gCSF) and an additional periparturient treatment with PEG-gCSF. We isolated milk neutrophils from 1428 and compared them to two cows challenged with experimental S. aureus, strain Newbould 305. Neutrophils from 1428’s milk had higher surface expression of myeloperoxidase compared to experimental Newbould challenged animals, as well as increased presence of Neutrophil Extracellular Traps. This suggests a heightened activation state of neutrophils sourced from 1428’s naturally occurring infection. Upon postmortem examination, the affected quarter revealed multifocal abscesses separated by fibrous connective tissues. Abscesses were most common in the gland cistern and collecting duct region. Microscopically, the inflammatory reaction was pyogranulomatous to granulomatous and consistent with botryomycosis. Colonies of Gram-positive cocci were found within the eosinophilic matrix of the Splendore-Hoeppli reaction within granulomas and intracellularly within the acinar epithelium.

Conclusions
Collectively, we describe a unique case of chronic mastitis, the characterization of which provides valuable insight into the mechanics of S. aureus treatment resistance and immune escape.
Mastitis is estimated to cost the US dairy industry $2 billion per year [ 1 ]. One of the most common mastitis causing pathogens remains Staphylococcus aureus ( S. aureus ), which can appear in both chronic and acute varieties, with markedly low cure rates [ 2 , 3 ]. S. aureus is known to escape immune clearance by adhering and infiltrating epithelial cells of the mammary gland which contributes to the difficulty to treat an infection [ 4 , 5 ]. S. aureus can also be associated with walled-off aggregates seen histologically as Splendore-Hoeppli phenomena [ 6 ]. Strain specific phenotypes are also associated with S. aureus infections in cattle, including varying degrees of epithelial invasiveness, and inflammatory responses [ 5 , 7 , 8 ].
Neutrophils are a primary immune effector cell in response to an intramammary infection [ 9 , 10 , 11 ]. Circulating neutrophils express the cell adhesion molecule CD62L (L-selectin) on their cell surface. In response to an infection, local vascular signaling molecules interact with CD62L. This activation causes CD62L to be cleaved and shed from the cell surface, which facilitates cell migration into the tissue and helps target CD62L expressing immune cells, such as neutrophils, to the site of infection [ 10 , 12 , 13 , 14 ]. An additional adhesion molecule, CD62E (E-selectin), is differentially expressed on vascular endothelial cells at the site of infection. Neutrophils have been shown to upregulate their surface expression of myeloperoxidase (MPO) in response to stimulus [ 15 , 16 ]. Glycovariants of surface MPO are thought to bind to E-selectin [ 17 ] and maybe part of the mechanism that allows the movement of the neutrophils from the circulation into the mammary gland. Treatment of cows with PEGg-CSF can cause shedding of surface CD62L and up-regulation of cell surface MPO in neutrophils [ 18 ]. Experimental infection of the mammary gland has resulted in the appearance of neutrophils in the milk with the high surface level of CD62L and MPO, suggesting their translocation from the blood into the mammary gland of infected cows [ 18 ].
When they encounter a pathogen, neutrophils have multiple antimicrobial mechanistic weapons at their disposal. They can produce reactive oxygen species, phagocytose the bacteria, or eject their genomic material to capture the bacterium in what are called Neutrophil Extracellular Traps (NETs) that contain antimicrobial proteins [ 19 , 20 , 21 ]. The presence of neutrophil NETs in milk from infected cows can be observed by DNA stains of the milk fat. Of the three antimicrobial mechanisms employed by neutrophils, NETs have been shown to have a longer efficacy in milk than the others [ 20 ].
While antibiotics are the most common treatment for mastitis cases, alternative approaches do exist including preventative cytokine therapeutics such as pegylated granulocyte-colony stimulating factor (PEG-gCSF) (Imrestor/Pegbovigrastim, Elanco, IN USA). These alternative approaches have been shown to boost circulating neutrophil numbers, lower disease severity against mastitis challenge, and reduce the naturally occurring incidence of mastitis when administered during the periparturient period [ 22 , 23 ].
We describe the case of a three-year old Holstein dairy cow (1428) who presented with a naturally-occurring, subclinical mastitis infection in her left hindquarter, approximately two months into her first lactation. Milk samples from cows in our research herd are periodically monitored for bacterial growth and changes in SCC (Somatic Cell Count) to monitor animal and udder health. Additional samples are collected if naturally occurring mastitis is suspected or for various scientific uses. Cow 1428 was born and raised on the USDA campus within the Holstein research herd. Mastitis was observed at a routine daily milking and culture of the sample was performed. Milk from quarters of interest was aseptically collected, by hand milking, and SCC sample values were determined by Dairy Lab Services (IA, USA). For bacterial counts, aseptically collected milk samples were plated on Trypticase Soy Agar with 5% sheep blood plates (BD Biosciences, CA, USA Cat. No.221261), and incubated overnight at 37 °C, prior to colony counting. An isolated colony was typed by the Iowa State University Veterinary Diagnostic Laboratory (ISU VDL) and identified as S. aureus . The S. aureus strain was sequenced and designated as SA1428 [ 24 ]. The infection remained subclinical, with no identifiable drop in milk yield, no visual signs of inflammation including teat hardening, redness, or milk chunkiness, but was continuously identifiable by moderately increased SCC and bacterial culture. Multiple SCC and bacterial counts were determined over the course of several months. At the initial detection of the infection SCC in the infected quarter were 3.8 × 10 6 cells/ mL with bacteria counts > 3000 cfu/mL. Other quarters had no detectable bacteria and SCC below 7.5 × 10 4 cells/ mL. Cow 1428 was not isolated from the herd, however, no other cows became naturally infected with the novel S. aureus pathogen to our knowledge.
Cow 1428 was treated with antibiotics, daily for five days, with cephapirin sodium (ToDAY, Boehringer Ingelheim, MO, USA) twice daily, and additionally pirlimycin hydrochloride (PIRSUE, Zoetis, NJ, USA) once daily. In our herd, this treatment has been successful at clearing experimentally induced S. aureus infections (Newbould 305 strain). When antibiotics did not clear the infection (as confirmed by bacterial culture) an additional round of antibiotics was completed two months later, which also failed to clear 1428’s infection. Numerous rounds of antibiotic treatment may not be a common commercial practice, but was appropriate within our research herd where previously we have been able to clear experimental S. aureus infections with this specific treatment and where milk is not used for human consumption. Interestingly, susceptibility testing of SA1428 by the ISU VDL, revealed susceptibility to several antibiotics (Ampicillin, Ceftiofur, Cephalothin, Erythromycin, Oxacillin, Penicillin, Penicillin/Novobiocin, Pirlimycin, Sulfadimethoxine, and Tetracycline). Mid-lactation, cow 1428 was treated off-label with a cytokine-based, PEG-gCSF therapy (Imrestor/Pegbovigrastim, Elanco, IN, USA), which was administered in two subcutaneous doses of 2.7 mL of 15 mg PEG-gCSF 7 days apart. While on-label use is designed for periparturient administration, our group was interested if the PEG-gCSF targeted neutrophil expansion would have an effect on 1428’s chronic infection. After PEG-gCSF treatment, circulating blood neutrophils increased from 2× 10 9 cells per liter of blood to 54 × 10 9 cells per liter of blood at their peak, 2 days post the second PEG-gCSF injection. In her infected quarter, cow 1428’s SCC also increased, from 1.11 × 10 6 cells per milliliter of milk to 5.17 × 10 6 cells per milliliter of milk, peaking 4 days post the second PEG-gCSF injection. Despite the increased presence of circulating neutrophils and milk SCC, cow 1428 did not clear the S. aureus infection. After being bred and confirmed pregnant, cow 1428 was dried off approximately 60 days prior to calving. In accordance with general dry-off practice, she was treated with cephapirin benzathine (ToMORROW, Boehringer Ingelheim, MO, USA). Seven days prior to cow 1428’s calving date she was treated again with an injection of PEG-gCSF, and again on the day of calving, as directed by on-label use of the product. Despite this treatment, 1428’s S. aureus infection presented immediately with the start of her second lactation.
We sought to characterize the infected mammary gland environment to identify phenotypes associated with SA1428 infection as compared to experimentally infected S. aureus . For the experimentally infected samples, we utilized resident Holsteins five weeks post experimental infection with S. aureus Newbould 305. For another ongoing study on the USDA National Animal Disease Center campus, eight Holstein cows were challenged by intramammary infusion in a single quarter with 150 CFU of S. aureus (Newbould). Subclinical, chronic infections developed in all cows. Five weeks after challenge, the Newbould infected cow with consistently high SCC values, and the Newbould infected cow with consistently low SCC values had milk samples collected for comparison along with milk from 1428’s naturally occurring infection. Over three consecutive days, 1428’s SCC for her infected quarter averaged 3.02 ± 0.78 × 10 6 cells per milliliter of milk, the high SCC cow averaged 12.59 ± 6.55 × 10 6 cells per milliliter of milk, and the low SCC cow averaged 0.24 ± 0.08 × 10 6 cells per milliliter of milk.
From S. aureus infected quarters 100–150 mL of milk was collected into 50 mL conical tubes. Samples were spun for 40 min, at 10,000 x g, at 4 °C to separate for pelleted milk cells for flow analysis and milk fat. Top milk fat layers were scraped into separate tubes, washed with PBS and protease inhibitor, and frozen for subsequent NET analysis.
Milk from the centrifuged samples was poured off, and cell pellets were placed on ice and resuspended in 1 mL media (L-glutamine, 10% FBS supplemented complete RPMI). Cell suspensions were layered over density gradients (Histopaque 1077, Sigma Aldrich, MO, USA, Cat No. 10771-500ML) spun for 20 min at 1500 x g, and had buffy coats removed leaving a highly neutrophil enriched cell pellet. Cell pellets were washed once with PBS and live cell counts were determined by cell counter (TC20 automated cell counter, BioRad, CA, USA). We used flow cytometry to evaluate the surface expression of MPO and L-selectin on neutrophils sourced from milk from infected quarters. To compare MPO and L-selectin expression over a range of SCC, we sampled milk from the Newbould infected cows with the highest and lowest SCC to compare with milk from cow 1428. Live milk cells were washed and resuspended in flow buffer (BioLegend, CA, USA, Cat. No. 420201). Individual primary, secondary, and directly conjugated antibodies were added to cell suspensions and incubated at room temperature for 15 min in the dark, with a flow buffer wash step between each antibody set. Samples were run on a Becton Dickinson LSR II flow cytometer and all analyses were performed with FlowJo software (FlowJo LLC, Ashland, OR, USA). Neutrophil gating was determined by forward and side scatter. Live, singlet milk cells were gated for CD45 (Monoclonal Antibody Center, Washington State University, USA. Cat. No. BOV2039). CD45 + cells were separately assessed for MPO (BioRad, Hercules, CA, USA Cat. No. VPA00193) and CD62L (BioLegend, San Diego, CA, USA Cat. No. 304824) surface expression. Flow cytometry of milk derived neutrophils from the three cows showed that cow 1428 had the greatest MPO surface expression (Fig. 1 a). Between the Newbould infected cows, the high SCC cow also showed higher surface MPO expression compared to the low SCC cow (Fig. 1 a). Surface expression of L-selectin revealed comparable levels on 1428 and the high SCC Newbould infected cow, but both were reduced compared to the low SCC Newbould infected cow (Fig. 1 b).
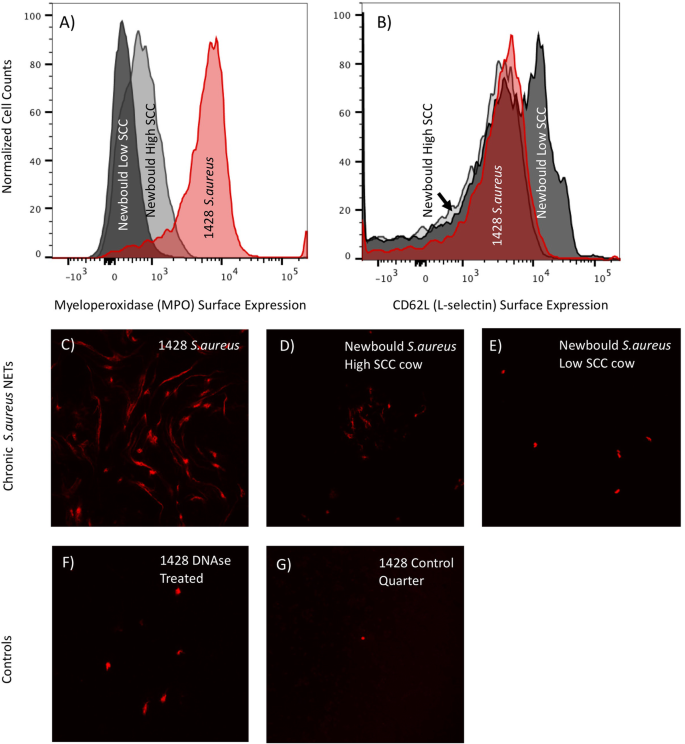
Flow cytometry and microscopy of NETs sourced from chronically S. aureus infected milk from 1428 and two Newbould infected cows (high and low somatic cell responders). Flow Cytometry of neutrophils isolated from infected milk was analyzed for surface expression of ( a ) myeloperoxidase and ( b ) CD62L (L-selectin). The red histogram depicts cell isolated from 1428 milk, dark gray is from a chronic Newbould challenged low SCC cow, and light gray represents a chronic Newbould challenged high SCC cow. Milk fat was additionally evaluated for the presence of Neutrophil Extracellular Traps (NETs). ( c ) 1428 S. aureus infected milk fat shows increased NET presence compared to both high ( d ) and low ( e ) SCC cows challenged with Newbould. Control NET staining shows DNAse treatment of 1428’s infected milk fat ( f ), and staining of clean healthy quarter milk fat from cow 1428
Neutrophils are known to produce NETs with activation which results in cell death. To capture information about NET-forming neutrophils, we stained milk fat for DNA as described previously [ 16 ], which is indicative of NET formation from all three cows. Microscopy showed that 1428 had the greatest NET presence (Fig. 1 c), compared to both Newbould infected cows (Fig. 1 d, e). DNAse treated and healthy milk fat controls are shown in Fig. 1 f, g. Samples were analyzed via confocal microscopy imaging using a Nikon A1R+ laser scanning microscope and NIS-Elements imaging software. Slide images are shown at the 20X objective, 75 numerical aperture, as imaged using a GaASP detector, 561 laser.
Cow 1428 was euthanized approximately 20 months after the first identification of infection by lethal injection of barbiturates by our institutional veterinarian. Gross pathology of the infected quarter of the mammary gland can be seen in Fig. 2 a, b . The infected quarter was systematically sampled by obtaining samples from 12 different sites; 3 each from the proximal and distal gland body of the gland, 3 from the collecting duct region and 1 each from the gland cistern, teat cistern and streak canal as illustrated in Fig. 2 c. Gross examination revealed multifocal abscesses and increased amounts of fibrous connective tissue, most notably in the collecting duct and gland cistern regions. Tissue samples (≤0.5 cm thick) were fixed by immersion in 10% neutral buffered formalin for 24 h, then transferred to 70% alcohol followed by standard paraffin embedding techniques. Paraffin embedded samples were cut to 4 μm thick sections, transferred to Superfrost Plus™ charged microscope slides (Thermo Fisher, MA, USA) and stained with hematoxylin and eosin (H&E). Adjacent sections were stained by the Hucker-Twort technique for visualization of Gram-positive and Gram-negative bacteria. Microscopically, samples from the teat sphincter and teat cistern were normal, with minimal if any inflammation (Fig. 2 c) and no bacteria present, confirmed by Gram stain. Samples from the gland cistern and collecting duct regions contained multifocal suppurative to pyogranulomatous infiltrates. Some regions contained distinct infiltrates of only neutrophils surrounding colonies of Gram-positive cocci embedded in a brightly eosinophilic, homogenous matrix, which radiated outward; interpreted to be Splendore-Hoeppli reaction (Fig. 3 a, b). In these same regions, there were also pyogranulomatous to granulomatous infiltrates arranged in nodules separated by prominent bands of fibrous connective tissue (Fig. 3 c). These nodular infiltrates contained variable numbers of extracellular Gram-positive cocci. In the alveolar duct and body regions of the gland, numerous acini contained infiltrates of large numbers of neutrophils (Fig. 3 d). In such acini, Gram-positive cocci were found both individually and in small colonies (Fig. 3 e). Some glands were absent of inflammatory infiltrates, but one to several Gram-positive cocci could still be found adhered to or within epithelial cells (Fig. 3 f).
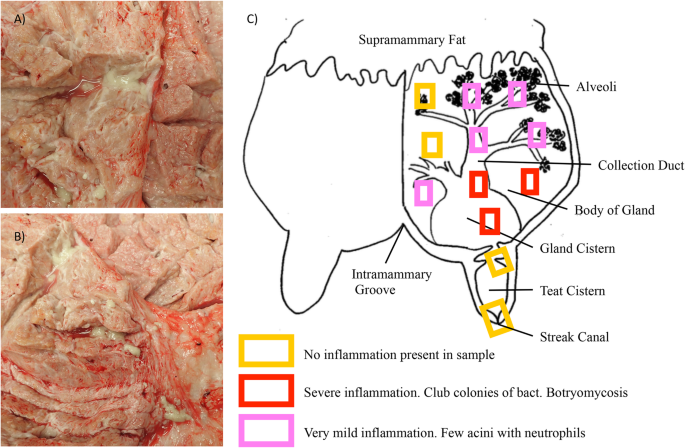
Gross pathology and necropsy collection outline detailing levels of inflammation. Gross pathology of 1428’s infected quarter ( a , b ). Note multifocal purulent exudate found in abscesses surrounded by fibrous connective tissue. The mammary gland was sampled as detailed in ( c ), capturing tissue from physiologically meaningful regions of the mammary gland. Also depicted, general levels of inflammation found at each section level ( c ). Diagram in ( c ) adapted from [ 25 ]
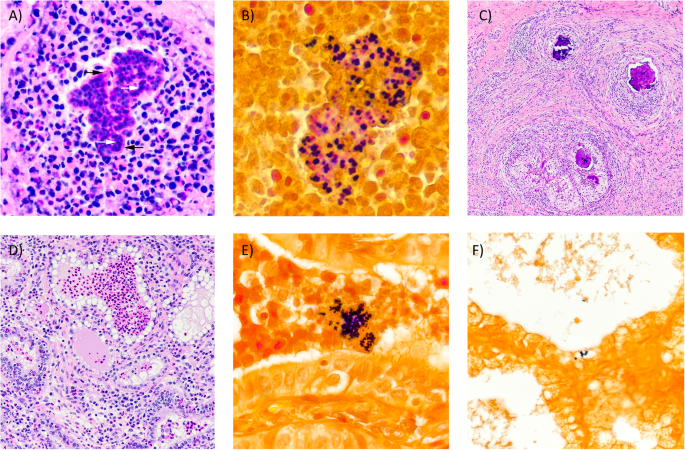
Photomicrographs of mastitic quarter from 1428. (A) Pyogranulomatous infiltrate. Note colonies of basophilic cocci (white arrows) within brightly eosinophilic and radiating matrix of Splendore-Hoeppli reaction (black arrows). H&E 20X. (B) Cocci within eosinophilic matrix are Gram-positive. Gram stain 40X. (C) Granulomas with abundant peripheral fibrosis and central areas of dystrophic mineralization. H&E 4X. (D) Acini containing numerous neutrophils. H&E 10X. (E) Intraluminal Gram-positive cocci within acinus. Gram stain 40X. (F) Intracellular Gram-positive cocci. Gram stain 20X
To further document S. aureus strain SA1428, its genome was sequenced. The genome sequencing data have been deposited in NCBI Sequence Read Archive under accession number PRJNA609126. The de novo genome assembly is available at NCBI with the accession number CP048431-CP048432.
Discussion and conclusions
This unique case report sheds light on important host and pathogen interactions that should be further investigated to be utilized in the development of mastitis treatment and preventative technologies. The presence of heightened immune cell activation in the mammary gland identified by present neutrophils, but lack of bacterial clearance, raises questions about the mechanisms of inflammation regulation and immune escape strategies of S. aureus as a mastitis causing pathogen. Low cure rates of S. aureus mastitis have been attributed to components of host and pathogen genetics, environmental exposure, and antibiotic resistance [ 26 ]. Additionally, while it’s known that S. aureus can evade neutrophil killing and gain intracellular access to epithelial cells [ 26 ], much more needs to be understood about why the immune response is ineffective and how S. aureus escape mechanisms function. In this work we demonstrate a correlation between infections caused by two strains of S. aureus and differences in surface expression of proteins of interest on milk neutrophils. Milk neutrophils sourced from cow 1428 had substantially increased surface expression of MPO (Fig. 1 a), which has been associated with cell activation [ 16 ] as well as accumulation within mastitic mammary glands [ 18 ]. Also consistent with neutrophil activation, levels of surface CD62L were comparable between 1428 cells and the high SCC Newbould cow, but both appeared to have increased shedding of CD62L compared to the low SCC cow (Fig. 1 b). These observations are consistent with the role of CD62L as an adhesion molecule important for the targeting cells to the site of infection. These observations are also supportive of the hypothesis that MPO expressed on the cell surface may be a ligand for E-selectin and potentially plays a role in cell migration to localized infections. Lastly, imaging showed more NETs present in 1428’s milk fat than in either of the Newbould challenged cows (Fig. 1 c, d, e). Collectively these findings suggest that cow 1428 had heightened neutrophil activation within the mammary gland compared to cells from experimentally infected animals. It is also clear that this activation is not driven strictly by accumulated cell numbers as the high SCC Newbould infected cow had four-fold higher SCC numbers than 1428. Of interest, between the two Newbould experimentally-infected animals, the high SCC Newbould infected cow had higher surface expression of MPO compared to the low SCC Newbould infected cow (Fig. 1 a), reduced surface expression of CD62L (Fig. 1 b), and increased NET presence (Fig. 1 d,e), which is supportive that these parameters are capturing biological activation. These findings should be further validated in the context of hypothesis driven experimental studies.
Many different strains of S. aureus exist that are capable of causing mastitis in dairy cattle. Variations in mastitis-causing strains include differences in the genotypic expression of virulence factors, biofilm production, cellular infiltration, and antimicrobial activity [ 27 ]. Comparison of an infection by SA1428 and SA Newbould highlights the phenotypic variation between strains that can be observed, both in terms of host immune cell response and response to antibiotic treatment; Newbould being successfully cleared and SA1428 persisting. The antibiotic susceptibility results of S. aureus SA1428, but failure to treat in vivo, suggests that the persistence of 1428’s infection may be contributed to physical escape by the bacteria. Histologically, inflammation of 1428’s infected quarter can be considered chronic in nature with pyogranulomatous and granulomatous lesions and increased fibrosis. The largest numbers of Gram-positive cocci were seen within the eosinophilic matrix of the Splendore-Hoeppli reaction associated with pyogranulomatous lesions and the nodular granulomatous infiltrates surrounded by large bands of fibrous connective tissue. Both settings provide protection from antibiotic treatment.
The naturally occurring, chronic case of S. aureus mastitis of cow 1428 describes the identification of strain SA1428, and describes an associated treatment-resistant phenotype. Our unique evaluation of activation levels of mammary sourced neutrophils, and detailed look at weakly characterized histology phenomena, contributes to the general knowledge of the behavior of chronic S. aureus infections, and offers several opportunities for hypothesis driven research to explore these findings.
Availability of data and materials
The genome sequencing data for SA1428 has been deposited in NCBI Sequence Read Archive (accession number PRJNA609126), and the de novo genome assembly is available at NCBI (accession number CP048431-CP048432). All additional datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
Somatic Cell Count
- Myeloperoxidase
Pegylated granulocyte-colony stimulating factor
Neutrophil extracellular trap
Hematoxylin and eosin
USDA-APHIS. Highlights of Dairy2007 part I: reference of dairy health and Management in the United States; 2007. p. 1–2.
Google Scholar
Ismail ZB. Mastitis vaccines in dairy cows: recent developments and recommendations of application. Veterinary World. 2017;10(9):1057–62.
Article CAS Google Scholar
Linder M, Paduch JH, Grieger AS, Mansion-de Vries E, Knorr N, Zinke C, et al. Cure rates of chronic subclinical Staphylococcus aureus mastitis in lactating dairy cows after antibiotic therapy. Berl Munch Tierarztl Wochenschr. 2013;126(7–8):291–6.
PubMed Google Scholar
Hensen SM, Pavicic MJ, Lohuis JA, de Hoog JA, Poutrel B. Location of Staphylococcus aureus within the experimentally infected bovine udder and the expression of capsular polysaccharide type 5 in situ. J Dairy Sci. 2000;83(9):1966–75.
Hensen SM, Pavicic MJ, Lohuis JA, Poutrel B. Use of bovine primary mammary epithelial cells for the comparison of adherence and invasion ability of Staphylococcus aureus strains. J Dairy Sci. 2000;83(3):418–29.
Bianchi RM, Schwertz CI, de Cecco BS, et al. Pathological and microbiological characterization of mastitis in dairy cows. Trop Anim Health Prod. 2019;51(7):2057–66.
Kim Y, Atalla H, Mallard B, Robert C, Karrow N. Changes in Holstein cow milk and serum proteins during intramammary infection with three different strains of Staphylococcus aureus. BMC Vet Res. 2011;7:51.
Kozytska S, Stauss D, Pawlik MC, Hensen S, Eckart M, Ziebuhr W, et al. Identification of specific genes in Staphylococcus aureus strains associated with bovine mastitis. Vet Microbiol. 2010;145(3–4):360–5.
Paape MJ, Bannerman DD, Zhao X, Lee JW. The bovine neutrophil: structure and function in blood and milk. Vet Res. 2003;34(5):597–627.
Burton JL, Erskine RJ. Immunity and mastitis. Some new ideas for an old disease. Vet Clin North Am Food Anim Pract. 2003;19(1):1–45.
Article Google Scholar
Kehrli ME Jr, Harp JA. Immunity in the mammary gland. Vet Clin North Am Food Anim Pract. 2001;17(3):495–516.
Ivetic A. A head-to-tail view of L-selectin and its impact on neutrophil behaviour. Cell Tissue Res. 2018;371(3):437–53.
Diez-Fraille A, Mehrzad J, Meyer E, Duchateau L, Burvenich C. Comparison of L-selectin and mac-1 expression on blood and milk neutrophils during experimental Escherichia coli-induced mastitis in cows. Am J Vet Res. 2004;65(8):1164–71.
Wang Y, Zhang AC, Ni Z, Herrera A, Walcheck B. ADAM17 activity and other mechanisms of soluble L-selectin production during death receptor-induced leukocyte apoptosis. J Immunol. 2010;184(8):4447–54.
Kindzelskii AL, Clark AJ, Espinoza J, Maeda N, Aratani Y, Romero R, et al. Myeloperoxidase accumulates at the neutrophil surface and enhances cell metabolism and oxidant release during pregnancy. Eur J Immunol. 2006;36(6):1619–28.
Powell EJ, Reinhardt TA, Casas E, Lippolis JD. The effect of pegylated granulocyte colony-stimulating factor treatment prior to experimental mastitis in lactating Holsteins. J Dairy Sci. 2018;101(9):8182–93.
Silvescu CI, Sackstein R. G-CSF induces membrane expression of a myeloperoxidase glycovariant that operates as an E-selectin ligand on human myeloid cells. Proc Natl Acad Sci U S A. 2014;111(29):10696–701.
Putz EJ, Eder JM, Reinhardt TA, Sacco RE, Casas E, Lippolis JD. Differential phenotype of immune cells in blood and milk following pegylated granulocyte colony-stimulating factor therapy during a chronic Staphylococcus aureus infection in lactating Holsteins. J Dairy Sci. 2019;102(10):9268–84.
Brinkmann V, Reichard U, Goosmann C, Fauler B, Uhlemann Y, Weiss DS, et al. Neutrophil extracellular traps kill bacteria. Science (New York, NY). 2004;303(5663):1532–5.
Lippolis JD, Reinhardt TA, Goff JP, Horst RL. Neutrophil extracellular trap formation by bovine neutrophils is not inhibited by milk. Vet Immunol Immunopathol. 2006;113(1–2):248–55.
Reinhardt TA, Sacco RE, Nonnecke BJ, Lippolis JD. Bovine milk proteome: quantitative changes in normal milk exosomes, milk fat globule membranes and whey proteomes resulting from Staphylococcus aureus mastitis. J Proteomics. 2013;82:141–54.
Canning P, Hassfurther R, TerHune T, Rogers K, Abbott S, Kolb D. Efficacy and clinical safety of pegbovigrastim for preventing naturally occurring clinical mastitis in periparturient primiparous and multiparous cows on US commercial dairies. J Dairy Sci. 2017;100(8):6504–15.
Ruiz R, Tedeschi LO, Sepulveda A. Investigation of the effect of pegbovigrastim on some periparturient immune disorders and performance in Mexican dairy herds. J Dairy Sci. 2017;100(4):3305–17.
Lippolis JD, Putz EJ, Ma H, Alt DP, Casas E, Reinhardt TA. Genome Sequence of a Staphylococcus aureus Strain Isolated from a Dairy Cow That Was Nonresponsive to Antibiotic Treatment. Microbiol Resour Announc. 2020;9(20):e00206–20.
Alany RG, Bhattarai S, S. P Devarajan PV. Intramammary delivery technologies for cattle mastitis treatment. In: Rathbone M, McDowell A, editors. Long acting animal health drug products. Advances in delivery science and technology. Boston: Springer; 2013. p. 295–327.
Chapter Google Scholar
Barkema HW, Schukken YH, Zadoks RN. Invited review: the role of cow, pathogen, and treatment regimen in the therapeutic success of bovine Staphylococcus aureus mastitis. J Dairy Sci. 2006;89(6):1877–95.
Côté-Gravel J, Malouin F. Symposium review: features of Staphylococcus aureus mastitis pathogenesis that guide vaccine development strategies. J Dairy Sci. 2019;102(5):4727–40.
Download references
Acknowledgements
Our greatest appreciation goes to Duane Zimmerman, Tera Nyholm, Adrienne Shircliff, and Judi Stasko of the USDA Microscopy Service team, as well as to the animal and veterinary care staff for the excellent care of animals. Mention of trade names, proprietary products, or specified equipment do not constitute a guarantee or warranty by the USDA and does not imply approval to the exclusion of other products that may be suitable. USDA is an Equal Opportunity Employer.
This research was funded solely by the USDA.
Author information
Authors and affiliations.
USDA-ARS-National Animal Disease Center, Infectious Bacterial Diseases Research Unit, Ames, IA, USA
Ellie J. Putz & Mitchell V. Palmer
USDA-ARS-National Animal Disease Center, Ruminant Disease and Immunology Research Unit, Ames, IA, USA
Ellie J. Putz, Hao Ma, Eduardo Casas, Timothy A. Reinhardt & John D. Lippolis
You can also search for this author in PubMed Google Scholar
Contributions
EJP wrote the manuscript and performed neutrophil characterization. MP was the case pathologist, responsible for necropsy sample collection, interpretation of histological results, and helped write and edit the manuscript. HM was responsible for the processing of sequence data. JDL and TAR helped interpret results, contributed to writing and editing the manuscript, and aided in designing characterization methods. The author(s) read and approved the final manuscript.
Corresponding author
Correspondence to John D. Lippolis .
Ethics declarations
Ethics approval.
All animal procedures were done with animals owned by the USDA and approved by the National Animal Health Center Animal Care and Use Committee.
Consent for publication
Not applicable.
Competing interests
EC is editorial board member of BMC Veterinary Research. The other authors declare that they have no competing interests.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Putz, E.J., Palmer, M.V., Ma, H. et al. Case report: characterization of a persistent, treatment-resistant, novel Staphylococcus aureus infection causing chronic mastitis in a Holstein dairy cow. BMC Vet Res 16 , 336 (2020). https://doi.org/10.1186/s12917-020-02528-8
Download citation
Received : 19 May 2020
Accepted : 18 August 2020
Published : 15 September 2020
DOI : https://doi.org/10.1186/s12917-020-02528-8
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Staphylococcus aureus
- Antibiotic resistant
- Splendore-Hoeppli
BMC Veterinary Research
ISSN: 1746-6148
- General enquiries: [email protected]
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
- My Bibliography
- Collections
- Citation manager
Save citation to file
Email citation, add to collections.
- Create a new collection
- Add to an existing collection
Add to My Bibliography
Your saved search, create a file for external citation management software, your rss feed.
- Search in PubMed
- Search in NLM Catalog
- Add to Search
Granulomatous mastitis: a case series
Affiliation.
- 1 Department of General Surgery, Dow University of Health Sciences/Civil Hospital, Karachi. [email protected]
- PMID: 20642963
- J Coll Physicians Surg Pak. 2010 Oct;20(10):706. Memon, Aysha [corrected to Memon, Aisha]
Objective: To find out the clinical and management profile of granulomatous mastitis (GM).
Study design: A case.
Place and duration of study: This study was carried out at Civil Hospital and Dow University of Health Sciences, Karachi and Tuberculosis Clinic at Bantwa Hospital, Karachi, from January 1999 to January 2009.
Methodology: Histopathologically diagnosed cases of GM were studied. Data included, detailed history, examination, clinical diagnosis, ultrasound, mammogram and fine needle aspiration cytology (FNAC). All patients were initially given antibiotics and corticosteroid for a period of 10 days. All patients were followed for a period of 6 months. Recurrent cases were managed by wide excision and followed for another period of 6 months. Percentage, mean and range values were determined on SPSS version 10.
Results: The mean age of the 22 patients was 37.6 years (range=22-52 years). Right breast was affected in 54.5% and left breast in 45.5% patients. Eighteen (82%) were non-lactating. Patients presented clinically with painful breast lump in 54.5%, painless lump in 45.5% and additional discharging sinuses in 18%. Clinical diagnosis was chronic mastitis in 40.9%, tuberculosis of breast in 36.3% and malignancy in 22.7% patients. Mammography showed an ill-defined mass in 45.5% and asymmetrical density in 31.8%. Diagnosis was confirmed in all patients on histopathology. Recurrence was noted in 2 patients, managed by wide excision, and followed for another period of 6 months without evidence of complications.
Conclusion: GM is an uncommon chronic inflammatory disease of the breast. Usually involving a single non-lactating breast in reproductive age group. It clinically mimics tuberculosis and carcinoma. Mammography remains non-conclusive. Excision and wide excision biopsy are both diagnostic and therapeutic in majority of cases. Treatment includes short course of steroids and antibiotic along with close regular surveillance.
PubMed Disclaimer
Similar articles
- Spectrum of breast tuberculosis. Afridi SP, Memon A, Rehman SU, Memon A, Baig N. Afridi SP, et al. J Coll Physicians Surg Pak. 2009 Mar;19(3):158-61. J Coll Physicians Surg Pak. 2009. PMID: 19268014
- Feasibility of surgical management in patients with granulomatous mastitis. Asoglu O, Ozmen V, Karanlik H, Tunaci M, Cabioglu N, Igci A, Selcuk UE, Kecer M. Asoglu O, et al. Breast J. 2005 Mar-Apr;11(2):108-14. doi: 10.1111/j.1075-122X.2005.21576.x. Breast J. 2005. PMID: 15730456
- Idiopathic granulomatous mastitis: time to avoid unnecessary mastectomies. Bani-Hani KE, Yaghan RJ, Matalka II, Shatnawi NJ. Bani-Hani KE, et al. Breast J. 2004 Jul-Aug;10(4):318-22. doi: 10.1111/j.1075-122X.2004.21336.x. Breast J. 2004. PMID: 15239790
- [Idiopathic granulomatous mastitis. Review of the literature illustrated by 4 cases]. Ayeva-Derman M, Perrotin F, Lefrancq T, Roy F, Lansac J, Body G. Ayeva-Derman M, et al. J Gynecol Obstet Biol Reprod (Paris). 1999 Dec;28(8):800-7. J Gynecol Obstet Biol Reprod (Paris). 1999. PMID: 10635482 Review. French.
- Tubercular mastitis - a great masquerader. Gon S, Bhattacharyya A, Majumdar B, Kundu S. Gon S, et al. Turk Patoloji Derg. 2013;29(1):61-3. doi: 10.5146/tjpath.2013.01150. Turk Patoloji Derg. 2013. PMID: 23354799 Review.
- Granulomatous mastitis: changing clinical and imaging features with image-guided biopsy correlation. Handa P, Leibman AJ, Sun D, Abadi M, Goldberg A. Handa P, et al. Eur Radiol. 2014 Oct;24(10):2404-11. doi: 10.1007/s00330-014-3273-z. Epub 2014 Jun 25. Eur Radiol. 2014. PMID: 24962828
- Idiopathic granulomatous lobular mastitis - report of 43 cases from iran; introducing a preliminary clinical practice guideline. Omranipour R, Mohammadi SF, Samimi P. Omranipour R, et al. Breast Care (Basel). 2013 Dec;8(6):439-43. doi: 10.1159/000357320. Breast Care (Basel). 2013. PMID: 24550752 Free PMC article.
- Search in MeSH
Related information
Linkout - more resources.
- MedlinePlus Health Information
Research Materials
- NCI CPTC Antibody Characterization Program
- Citation Manager
NCBI Literature Resources
MeSH PMC Bookshelf Disclaimer
The PubMed wordmark and PubMed logo are registered trademarks of the U.S. Department of Health and Human Services (HHS). Unauthorized use of these marks is strictly prohibited.
- Research article
- Open access
- Published: 11 October 2006
A case-control study of mastitis: nasal carriage of Staphylococcus aureus
- Lisa H Amir 1 ,
- Suzanne M Garland 2 &
- Judith Lumley 1
BMC Family Practice volume 7 , Article number: 57 ( 2006 ) Cite this article
10k Accesses
28 Citations
3 Altmetric
Metrics details
Mastitis is a common problem for breastfeeding women. Researchers have called for an investigation into the possible role of maternal nasal carriage of S. aureus in the causation of mastitis in breastfeeding women.
The aim of the study was to investigate the role of maternal S. aureus nasal carriage in mastitis. Other factors such as infant nasal S. aureus carriage, nipple damage, maternal fatigue and oversupply of milk were also investigated. A case-control design was used. Women with mastitis (cases, n = 100) were recruited from two maternity hospitals in Melbourne, Australia (emergency departments, breastfeeding clinics and postnatal wards). Breastfeeding women without mastitis (controls, n = 99) were recruited from maternal and child health (community) centres and the rooms of a private obstetrician. Women completed a questionnaire and nasal specimens were collected from mother and baby and placed in charcoal transport medium. Women also collected a small sample of milk in a sterile jar.
There was no difference between nasal carriage of S. aureus in breastfeeding women with mastitis (42/98, 43%) and control women (45/98, 46%). However, significantly more infants of mothers with mastitis were nasal carriers of S. aureus (72/88, 82%) than controls (52/93, 56%). The association was strong (adjusted OR 3.23, 95%CI 1.30, 8.27) after adjustment for the following confounding factors: income, private health insurance, difficulty with breastfeeding, nipple damage and tight bra. There was also a strong association between nipple damage and mastitis (adjusted OR 9.34, 95%CI 2.99, 29.20).
We found no association between maternal nasal carriage of S. aureus and mastitis, but nasal carriage in the infant was associated with breast infections. As in other studies of mastitis, we found a strong association between nipple damage and mastitis. Prevention of nipple damage is likely to reduce the incidence of infectious mastitis. Mothers need good advice about optimal attachment of the baby to the breast and access to skilled help in the early postpartum days and weeks.
Peer Review reports
Mastitis is a common problem for breastfeeding women [ 1 – 3 ] yet it is a poorly researched topic [ 4 ]. Mastitis may be a noninfective inflammation, resolving with heat and increased breast drainage [ 5 , 6 ], or an infective process which may lead to a breast abscess [ 7 ].
The most commonly isolated organism in lactating women with mastitis is Staphylococcus aureus ( S. aureus ): present in 32% to 44% of breast milk samples [ 8 – 11 ]. S. aureus is a commensal which may colonise the nostrils, axillae, vagina and pharynx of 30 to 50% of adults; as well as damaged skin, such as traumatised nipples of lactating women [ 12 , 13 ]. When S. aureus are present in the nostrils, they may act as a reservoir of S. aureus for clinical infections in the host or may facilitate spread to other people [ 14 ]. A review has concluded that four studies conducted in the 1990s found that S. aureus nasal carriers had a relative risk of 7.1 (95%CI 4.6, 11.0) of surgical-site infections, due to wound colonisation by the patient's endogenous flora [ 15 ].
Researchers have called for an investigation into the possible role of maternal nasal carriage of S. aureus in the causation of mastitis [ 16 ]. In clinical practice, there is anecdotal evidence that maternal or infant nasal carriage may be linked to recurrent mastitis in lactating women [ 17 ]. Our literature review did not identify any studies investigating the role of maternal or infant nasal carriage of S. aureus in mastitis. Medline searches (via PubMed) were conducted using keywords "staph* AND (lactation OR breastfeeding OR postpartum OR mastitis)" limited to human studies. A recent update (31 May 2006) has identified one case study of a mother of premature quadruplets who had symptoms of mastitis; methicillin-resistant S. aureus (MRSA) was isolated in expressed breast milk and nasal cultures from mother and infants [ 18 ].
We conducted a case-control study to examine the possible role of maternal nasal carriage of S. aureus in the development of mastitis. The role of S. aureus nasal carriage in the infant and other factors reputed to predispose women to mastitis, such as nipple damage, maternal fatigue and oversupply of milk, were also assessed.
Cases were women with mastitis attending the Emergency Department or the Breastfeeding Clinic at the Royal Women's Hospital or Mercy Hospital for Women in Melbourne, Australia. Mastitis was defined as at least two breast signs or symptoms (pain, redness or lump) and one systemic symptom (fever or 'flu-like symptoms) present for at least twelve hours.
Women in the control group were lactating women (with babies aged six weeks or less) attending Maternal and Child Health (MCH) centres in metropolitan Melbourne (community clinics attended by new mothers). Also, women attending a private obstetrician for their six-week postnatal visit were invited to join the study.
After women gave written consent, they completed a questionnaire about nipple damage, oversupply of milk and other factors identified as possible predisposing factors for mastitis. Maternal fatigue was assessed using the four Vitality questions from the SF-36 [ 19 ]. (The SF-36 Health Survey is one of the most widely used health-related quality of life questionnaires, measuring eight health concepts, including Physical functioning, Social functioning and Vitality) [ 19 ]. At the end of the questionnaire for women with mastitis, participants were asked to describe "how you have been feeling and how mastitis has affected you"; the results of these open-text comments have been published separately [ 20 ].
Nasal specimens were collected from mother and baby. Saline-moistened swabs were rotated around the inside of the nasal vestibule, then placed in Amies charcoal transport medium. Women used a sterile water wipe to cleanse her nipple then expressed a small sample of milk in a sterile jar. Specimens were labelled and delivered to the microbiology laboratory at the Royal Children's Hospital, where the swabs were inoculated on Mannitol Salt Agar plates, a medium selective for staphylococci. An aliquot of expressed breast milk was also placed on a Mannitol Salt Agar plate. S. aureus was confirmed with DNase test (thermostable nuclease) and antibiotic susceptibility was conducted using standard microbiological methods.
Using Epi-Info 6.0 for an unmatched case-control study with 95% confidence and 80% power, if 20% of controls were nasal carriers and 40% of cases, we would need 91 women in each group. We planned to recruit 100 cases (women with mastitis) and 100 controls (women in the community). The data were analysed by Stata 8.0 computer program. The comparability of cases and control groups were described. Odds Ratios were calculated to compare exposures in each group, and Mantel-Haenszel Weighted Odds Ratios where appropriate. Logistic regression was used to determine factors predictive of mastitis.
Four infants over 6 weeks age were inadvertently recruited for the control group (two at 7 weeks, one 9 and one 11 weeks). A sensitivity analysis was conducted by repeating the analysis without these infants and the results were found to be almost identical: therefore the records of these four mothers and babies were retained in the sample.
Ethics approval:
La Trobe University Human Ethics Committee (21/8/02 Project 02-61)
Research and Ethics Committees, Royal Women's Hospital (23/7/02, Project 02/22)
Research Ethics Committee, Mercy Hospital for Women (17/7/02, Project R02/32)
Department of Human Services, Victoria (5/6/02, Project 36/02).
One hundred women with mastitis (cases) and ninety-nine breastfeeding women (controls) were recruited between August 2002 and April 2004. Recruiting was stopped as the database incorrectly indicated that 100 controls had been recruited.
Fifty-four women with mastitis were recruited at the Mercy Hospital for Women and 46 women at the Royal Women's Hospital; from the breastfeeding clinics (n = 38), readmitted with mastitis in the wards (n = 32) and through the emergency departments (n = 25; 5 missing data). Most of the women for the control group were recruited through MCH centres (n = 70), whilst 29 women were from the private obstetrician.
Characteristics of study population
The background characteristics are displayed in Table 1 . Women with mastitis were older than women in the control group (34 years and 32 years, p < 0.05), while the median age of the babies was 16 days for cases and 36 days for controls. Family income was lower in the women with mastitis and they were less likely to have private health insurance.
Table 2 shows the health of women and their infants. After a question to determine if women had experienced mastitis in the past, women were asked "Have you ever had any other staph bacterial infections? For example: boils, abscesses, sores inside your nose?" Women with mastitis were more likely to have self-reported a past history of staphylococcal infection(s), 23% compared to 12%.
The sum of the scores from the four Vitality questions in the SF36 ranged from 4 (low) to 17 (high), with a mean of 10.7 (n = 195). Women were dichotomised into two groups scoring above or below the mean, and the groups compared. Sixty-one percent of women with mastitis scored below the mean vitality score, compared to 26% of controls. However, it is possible that some women were reporting lethargy associated with the onset of mastitis.
The breastfeeding characteristics of the sample can be seen in Table 3 . Cases were more likely to have had nipple pain (71%) compared to controls (35%), and more likely to be using purified lanolin on their nipples (64%) than controls (35%). Thirteen cases used topical antifungal cream/ointment/gel, while nine controls used topical antifungal treatment (not significant). More cases used nipple shields (16%) than controls (2%). Cases were also more likely to have experienced breast engorgement in the previous week (51%) than controls (19%), were more likely to say that they had missed a feed (54%) than the controls (36%) and that they had too much milk: 29% compared to 17%. Women were asked about pressure on their breasts in the previous week. More cases reported pressure from a tight bra (37%) than controls (20%).
Women with mastitis were more likely to report that their infant was having difficulty with breastfeeding, 57%, than the control group, 14%. Ten women with mastitis were currently feeding their infant expressed breast milk only, while none of the women in the control group was expressing only.
Microbiological results
There was no difference between nasal carriage of S. aureus in women with mastitis (43%) and women in the control group (46%) (Table 4 ). The overall proportion of women with a positive nasal culture for S. aureus was 44.4% (Binomial Exact 95% CI 37.3, 51.6). (MRSA was not isolated in any specimens in this study).
As expected, the expressed breast milk of women with mastitis was more likely to be positive for S. aureus (45/99, 46%), than the milk of controls (15/83, 18%). Most of the S. aureus isolated from the milk of the controls was reported as 'sparse" (11/15, 73%). Only one specimen in the control group was reported as "profuse" (1/15, 7%) compared to 14/45 (31%) in the mastitis group.
Significantly more infants of mothers with mastitis were nasal carriers of S. aureus (72/88, 82%) than infants in the control group (52/93, 56%, OR 3.55, 95%CI 1.80, 7.00). A high proportion of S. aureus in both groups was reported as "profuse", 66% of cases and 50% of controls, 59% in total. Overall, 68.5% of infants were nasal carriers of S. aureus (Binomial Exact 95% CI 61.2, 75.2).
The youngest infants were most likely to be nasal carriers (91% of infants in the first two weeks in the mastitis group), compared to 78% of infants aged 5–6 weeks. A stratified analysis of S. aureus nasal carriage in the infants was conducted to examine the results in babies at different ages. The Mantel-Haenszel weighted Odds Ratio was 3.49 (95%CI 1.38, 8.83) for infants of mothers with mastitis to be nasal carriers compared to infants in the control group (Table 5 ).
A statistically significant association was found between women with a cracked nipple and nasal carriage of S. aureus in their infants. Eighty-four percent (38/45) of women with a cracked nipple had a baby with nasal S. aureus , compared to 63% (85/135) of women without a cracked nipple, OR 3.19 (95%CI 1.33, 7.69). However, there was no association between nasal carriage in the mother and a cracked nipple: carriage in women with a cracked nipple was 39% (19/49) compared to 46% (67/146) of women without a cracked nipple.
Multivariate analysis
A logistic regression model was developed to look at factors predictive of mastitis. The independent variables of interest were tested individually against the dependent variable and were entered in the model if the p-value of the Wald statistic was ≤ 0.25 [[ 21 ], p95]. Where there were small numbers of missing values, records were deleted (seven records). Fifteen women had missing values for income, and sixteen babies did not have a result for nasal swab. These records were retained with the missing values coded accordingly. This left 192 records for analysis.
The initial model included the following variables: mother's age, income (2 levels), private health insurance, past history of staphylococcal infection, baby having difficulty with breastfeeding, nipple cracked, engorged breast/s, missed feed/s, tight bra, too much milk, using lanolin on nipple/s, baby positive for nasal S. aureus , mother anaemic, baby prefers one breast. Variables were eliminated one at a time using logistic regression. Only those with a p-value of the Wald statistic ≤ 0.05 were retained in the model. The process was repeated until only significant variables remained. Then all independent variables eliminated in the original univariate analysis were added back into the model one at a time to check that none was now significant given the reduced model. The lroc test identified that the area under ROC curve was 0.8778, that is a high sensitivity, and the lstat test showed 80.73% correctly classified. The final model (Model 1) is presented in Table 6 .
The adjusted Odds Ratio for infants of mothers with mastitis to be nasal carriers was 3.23 (95%CI 1.30, 8.27) after adjusting for possible confounding factors (Model 1). In order to explore the effects of breastfeeding factors and baby nasal carriage without including demographic factors, a second model was developed (Model 2). Without including the demographic variables (income and private health insurance), the second model is very similar to Model 1. In a third model (not shown), the demographic variables were included while limiting the analysis to private patients (n = 126), and the results were also similar. A fourth model (not shown, n = 184), excluding all babies over 7 weeks also found that infant nasal carriage was significant (adjusted OR 4.08, 95%CI 1.44, 11.67).
Summary of main findings and comparison with existing literature
The study showed that there was no difference in the proportion of women with mastitis and without mastitis who were nasal carriers (43% and 46% respectively). The overall proportion of women with a positive nasal culture for S. aureus was 44.4% (95% CI 37.3, 51.6). This is consistent with the mean of 37.2% in general populations calculated by Kluytmans and colleagues from eighteen studies in 13,873 people [ 15 ], but seems higher than other studies published in 2004: 29% [ 22 ], 24% [ 23 ] and 33% [ 24 ].
We found that a very high proportion of infants of mothers with mastitis were nasal carriers: 82% and this was statistically significantly higher than infants of other women (56%). Infant nasal carriage remained significant after adjusting for other variables. Younger infants were most likely to be nasal carriers than older infants in this study. In a similar manner, Peacock and colleagues found that 40–50% of infants were colonised with S. aureus in the first eight weeks, falling to 21% by six months [ 25 ].
An association was not found between parity or a history of mastitis and being a case in this study, in contrast to previous studies [ 2 , 16 ]. This may be related to the high proportion of primiparous women in both groups of our study. Also, women with a history of mastitis may have been more likely to volunteer to be a control than other women.
The presence of a cracked nipple was associated with a high odds for mastitis, 9.34 (95%CI 2.99, 29.20), after adjusting for other factors. Foxman and colleagues also found an association with "nipple cracks or sores" with an OR of 3.4 (95%CI 2.04, 5.51) on logistic regression [ 16 ]. Prevention of nipple damage is likely to reduce the incidence of infectious mastitis. New mothers need good advice about optimal attachment of the baby to the breast and access to skilled help in the early postpartum days and weeks.
This study found that using lanolin on nipples was significantly associated with mastitis on univariate analysis; however this was no longer significant on multivariate analysis. We expect that the association between creams on nipples and mastitis [ 26 ] is more likely to be related to the fact that nipple creams tend to be used when the nipples are damaged, and it is the nipple damage that probably is the route by which infection enters the breast rather than the creams themselves.
S. aureus was isolated in the milk of 46% of women with mastitis, a similar proportion to studies over the last thirty years [ 11 ]. S. aureus was also isolated in the milk of 17% of women without mastitis; mostly reported as "sparse" (11/15), probably reflecting contamination of the milk by bacteria on the skin of the nipple or the hands. In other studies between 0 and 20% of milk specimens from healthy women are positive for S. aureus [ 27 , 28 ].
Although it is not possible to conclude whether transmission occurred from the infant to the mother's breast or visa versa, it is likely that S. aureus was transmitted from the infant to the mother. In 1957, Wysham and colleagues demonstrated that 7 of 9 infants with positive throat cultures for S. aureus transmitted the organism to their bottle of formula milk [ 29 ]. Babies are born sterile and acquire their colonisation from their mother or the hands of health workers. Mothers and infants have been shown to be likely to carry the same strain of S. aureus [ 25 , 30 ]. Staphylococci may be transferred from the mother's nose to the infant's and then back to the mother's nipple, particularly if the nipple has been traumatised.
Strengths and limitations of this study
The diagnosis of mastitis relied on clinical signs and symptoms as there are no definitive tests for mastitis in women. The women in this study experienced either fever or systemic symptoms for at least 12 hours. Future studies could assess the usefulness of testing milk for the presence of leukocytes [ 31 ]. Molecular microbiology (eg pulsed field gel electrophoresis, PFGE) testing of isolates could have confirmed the clonality of S. aureus strains present in mothers with mastitis and in their infants. However, funding for this study was not sufficient to conduct PFGE.
It was originally planned to recruit women for the control group through MCH centres in the community (n = 25). However, we relied on women being referred to the study and recruitment was slow, so we started recruiting women attending a private obstetrician for their six-week postnatal check up. It would have been inappropriate to recruit women attending the hospital (Emergency Department or Breastfeeding clinic) as we were seeking women without problems for the control group. Therefore the controls were more likely to be private patients than cases, which resulted in more women from the higher income group as controls than cases. In order to assess if this had an effect on the study results, a logistic regression model was developed using only women with private insurance and similar results were obtained as when the model included all women.
A limitation of the case-control design is that any associations identified cannot be concluded to be causal. For example, private health insurance appears to be protective against mastitis (OR 0.27, 95%CI 0.14, 0.51), but this association is due to the selection bias that occurred during recruitment. The association between nasal S. aureus carriage in infants and mastitis in their mothers (adjusted OR 3.19, 1.23, 8.29) appears to be robust as it was significant in each logistic regression model. However, it does not tell us if this association proves a link between nasal carriage and mastitis nor in which direction the transmission is occurring.
Implications for clinical practice and future research
Mastitis is an acute painful illness, not limited to the breast, and often associated with a negative emotional response [ 20 ]. In order to prevent mastitis, clinicians could advice new mothers about the factors commonly associated with this problem, such as milk stasis caused by missed feeds, expressing and breast restriction [ 32 ]. Breastfeeding women can be alert for the early symptoms of mastitis when they have been extra busy, for example when travelling or when they have visitors staying. If women have anticipatory guidance they can overcome milk stasis in these situations by increasing breastfeeds or expressing more frequently.
Future studies need to aim to collect clinical specimens prospectively in order to determine the transmission dynamics between mother and infant. Molecular microbiology (e.g. PFGE) can be used to confirm that the same strain of S. aureus is present in mother and child, and the direction of transmission of organisms between mother and child.
It is not standard practice for mothers to wash hands before breastfeeding (less than 50% of women in both groups "always" washed hands). Future studies could focus on hand washing as S. aureus may be carried transiently on the hands [ 33 ] and can then be transferred to the breast. Hospitals should be aware of the possibility of transmission of potential pathogens on breast pumps; disinfection is particularly important after equipment is used by women with breast infections.
Another topic for future research is recurrent mastitis. Would it be possible to reduce recurrences of mastitis by reducing nasal carriage of S. aureus in mothers and infants where mothers have already experienced an episode of mastitis?
In conclusion, we found no association between maternal nasal carriage of S. aureus and mastitis, but nasal carriage in the infant was associated with breast infections. As in other studies of mastitis, we found a strong association between nipple damage and mastitis.
Kinlay JR, O'Connell DL, Kinlay S: Incidence of mastitis in breastfeeding women during the six months after delivery: a prospective cohort study. Med J Aust. 1998, 169: 310-312.
CAS PubMed Google Scholar
Vogel A, Hutchison BL, Mitchell EA: Mastitis in the first year postpartum. Birth. 1999, 26: 218-225. 10.1046/j.1523-536x.1999.00218.x.
Article CAS PubMed Google Scholar
Inch S: Incidence of mastitis among breastfeeding women from selected Oxfordshire general practitioners practices 1993-1994. 1996, Holloway, St Barts
Google Scholar
Renfrew MJ, Woolridge MW, Ross McGill H: Enabling Women to Breastfeed. A Review of Practices which Promote or Inhibit Breastfeeding - with Evidence-Based Guidance for Practice. 2000, London, The Stationery Office
World Health Organization: Mastitis: Causes and Management. 2000, Geneva, WHO/FCH/ CAH/00.13
Fetherston C: Mastitis in lactating women: physiology or pathology?. Breastfeed Rev. 2001, 9: 5-12.
Amir LH, Forster D, McLachlan H, Lumley J: Incidence of breast abscess in lactating women: report from an Australian cohort. BJOG. 2004, 111: 1378-1381.
Article PubMed Google Scholar
Thomsen AC, Espersen T, Maigaard S: Course and treatment of milk stasis, noninfectious inflammation of the breast, and infectious mastitis in nursing women. Am J Obstet Gynecol. 1984, 149: 492-495.
Matheson I, Aursnes I, Horgen M, Aabo O, Melby K: Bacteriological findings and clinical symptoms in relation to clinical outcome in puerperal mastitis. Acta Obstet Gynecol Scand. 1988, 67: 723-726.
Bertrand H, Rosenblood LK: Stripping out pus in lactational mastitis: a means of preventing breast abscess. Can Med Assoc J. 1991, 145: 299-306.
CAS Google Scholar
Osterman KL, Rahm VA: Lactation mastitis: bacterial cultivation of breast milk, symptoms, treatment and outcome. J Hum Lact. 2000, 16: 297-302.
Amir LH, Garland SM, Dennerstein L, Farish SJ: Candida albicans: is it associated with nipple pain in lactating women?. Gynecol Obstet Invest. 1996, 41: 30-34.
Livingstone VH, Willis CE, Berkowitz J: Staphylococcus aureus and sore nipples. Can Fam Physician. 1996, 42: 654-659.
CAS PubMed PubMed Central Google Scholar
von Eiff C, Becker K, Machka K, Stammer H, Peters G: Nasal carriage as a source of Staphylococcus aureus bacteremia. N Engl J Med. 2001, 344: 11-16. 10.1056/NEJM200101043440102.
Kluytmans J, Van Belkum A, Verbrugh H: Nasal carriage of Staphylococcus aureus: Epidemiology, underlying mechanisms, and associated risks. Clin Microbiol Rev. 1997, 10: 505-520.
Foxman B, D'Arcy H, Gillespie B, Bobo JK, Schwartz K: Lactation mastitis: occurrence and medical management among 946 breastfeeding women in the United States. Am J Epidemiol. 2002, 155: 103-114. 10.1093/aje/155.2.103.
Amir L: Breastfeeding and Staphylococcus aureus: three case reports. Breastfeeding Review. 2002, 10: 15-18.
PubMed Google Scholar
Gastelum DT, Dassey D, Mascola L, Yasuda LM: Transmission of community-associated methicillin-resistant Staphylococcus aureus from breast milk in the neonatal intensive care unit. Pediatr Infect Dis J. 2005, 24: 1122-1124. 10.1097/01.inf.0000189983.71585.30.
Ware JEJ, Gandek B, IQOLA Project Group: The SF-36 Health Survey: development and use in mental health research and the IQOLA project. Int J Ment Health. 1994, 23: 49-73.
Article Google Scholar
Amir LH, Lumley J: Women's experience of mastitis: 'I have never felt worse'. Aust Fam Physician. 2006, 35: 745-747.
Hosmer DW, Lemeshow S: Applied Logistic Regression. 2000, New York, John Wiley & Sons, Inc., 2nd Ed
Chapter Google Scholar
Bischoff WE, Wallis ML, Tucker KB, Reboussin BA, Sherertz RJ: Staphylococcus aureus nasal carriage in a student community: prevalence, clonal relationships, and risk factors. Infect Control Hosp Epidemiol. 2004, 25: 485-491. 10.1086/502427.
Berthelot P, Grattard F, Fascia P, Martin I, Mallaval FO, Ros A, Pozzetto B, Lucht F: Is nasal carriage of methicillin-resistant Staphylococcus aureus more prevalent among student healthcare workers? (Letter). Infect Control Hosp Epidemiol. 2004, 25: 364-365. 10.1086/503498.
Eveillard M, Martin Y, Hidri N, Boussougant Y, Joly-Guillou ML: Carriage of methicillin-resistant Staphylococcus aureus among hospital employees: prevalence, duration, and transmission to households. Infect Control Hosp Epidemiol. 2004, 25: 114-120. 10.1086/502360.
Peacock SJ, Justice A, Griffiths D, de Silva GD, Kantzanou MN, Crook D, Sleeman K, Day NP: Determinants of acquisition and carriage of Staphylococcus aureus in infancy. J Clin Microbiol. 2003, 41: 5718-5725. 10.1128/JCM.41.12.5718-5725.2003.
Article PubMed PubMed Central Google Scholar
Foxman B, Schwartz K, Looman SJ: Breastfeeding practices and lactation mastitis. Soc Sci Med. 1994, 38: 755-761. 10.1016/0277-9536(94)90466-9.
Wright KC, Feeney AM: The bacteriological screening of donated human milk: laboratory experience of British Paediatric Association's published guidelines. J Infect. 1998, 36: 23-27. 10.1016/S0163-4453(98)92946-2.
Eidelman AI, Szilagyi G: Patterns of bacterial colonization of human milk. Obstet Gynecol. 1979, 53: 550-552.
Wysham DN, Mulhern ME, Navarre GC, LaVeck GD, Kennan AL, Giedt WR: Staphylococcal infections in an obstetric unit. II Epidemiologic studies of puerperal mastitis. New Engl J Med. 1957, 257: 304-306.
Kawada M, Okuzumi K, Hitomi S, Sugishita C: Transmission of Staphylococcus aureus between healthy, lactating mothers and their infants by breastfeeding. J Hum Lact. 2003, 19: 411-417. 10.1177/0890334403257799.
Thomsen AC, Hansen KB, Moller BR: Leukocyte counts and microbiologic cultivation in the diagnosis of puerperal mastitis. Am J Obstet Gynecol. 1983, 146: 938-941.
Academy of Breastfeeding Medicine: Academy of Breastfeeding Medicine, Clinical Protocol Number 4: Mastitis. ABM News and Views. 30, 34-[ http://www.bfmed.org/ ]
Casewell MW: The nose: an underestimated source of Staphylococcus aureus causing wound infection. J Hosp Infect. 1998, 40: S3-S11. 10.1016/S0195-6701(98)90199-2.
Pre-publication history
The pre-publication history for this paper can be accessed here: http://www.biomedcentral.com/1471-2296/7/57/prepub
Download references
Acknowledgements
We would like to thank all the women who participated, the staff in the Breastfeeding Clinics, Maternal and Child Health Centres and Emergency Departments, in particular Colleen Stevens and Danielle Clifford.
Lisa Amir received a National Health and Medical Research Council Medical Public Health PhD Scholarship and a grant from the Medical Research Foundation for Women and Babies.
Author information
Authors and affiliations.
Mother & Child Health Research, La Trobe University, Melbourne, Australia
Lisa H Amir & Judith Lumley
Department of Microbiology and Infectious Diseases, The Royal Women's Hospital, Melbourne, Australia
Suzanne M Garland
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Lisa H Amir .
Additional information
Competing interests.
The author(s) declare that they have no competing interests.
Authors' contributions
The study was conducted by LHA as part of her PhD. SMG and JL supervised the project and contributed to the study design, analysis and writing.
Rights and permissions
This article is published under license to BioMed Central Ltd. This is an Open Access article distributed under the terms of the Creative Commons Attribution License ( http://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Reprints and permissions
About this article
Cite this article.
Amir, L.H., Garland, S.M. & Lumley, J. A case-control study of mastitis: nasal carriage of Staphylococcus aureus . BMC Fam Pract 7 , 57 (2006). https://doi.org/10.1186/1471-2296-7-57
Download citation
Received : 07 June 2006
Accepted : 11 October 2006
Published : 11 October 2006
DOI : https://doi.org/10.1186/1471-2296-7-57
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Private Health Insurance
- Nasal Carriage
- Express Breast Milk
BMC Primary Care
ISSN: 2731-4553
- General enquiries: [email protected]
Meet the 35-year-old CEO who wants to turn around bankrupt Red Lobster
- Former P.F. Chang's CEO Damola Adamolekun is set to take over Red Lobster as its CEO.
- Adamolekun moved to P.F. Chang's during the pandemic and pivoted its business to accommodate more to-go services.
- The 35-year-old's role at Red Lobster is pending a judge's approval of the company sale, set to take place next month.

Red Lobster has a new leader to right the company following its recent bankruptcy.
Damola Adamolekun, the former CEO at Asian-fusion restaurant chain P.F. Chang's, has been appointed Red Lobster's new CEO to guide the company through its restructuring process, a consortium of investors said Monday.
Red Lobster filed for Chapter 11 bankruptcy in May, after accruing over $1 billion in debt. The company faced a series of setbacks, including rising costs and shifting consumer preferences, Business Insider wrote in May.
The restaurant's previous owner, Thai Union Group, said it would exit the company, citing significant financial losses. The group had initially acquired a minority stake in 2016 and took majority control in 2020, but struggled to manage the brand's declining sales and rising costs — including big losses from its $20 "Endless Shrimp" promotion, BI reported in May.
The company secured $100 million from lenders in May, who took control of the business.
Enter Adamolekun, who was appointed by Red Lobster's lenders to take over the restaurant business. The 35-year-old most recently served as an operating partner at private equity firm Garnett Station Partners.
Earlier, Adamolekun helmed P.F. Chang's from June 2020 to August 2023. He oversaw the company through the difficulties posed by the COVID-19 pandemic.
The restaurant shifted its operations to focus on technology for "online optimization and putting our staff into new roles," Adamolekun told BI in 2022.
Under his leadership, P.F. Chang's pivoted to to-go services and resumed expansion efforts, adding about 225 locations in 2023, the company told the WSJ.
In a 2022 interview with BI, Adamolekun emphasized the importance of adaptability, customer focus, and transparent leadership.
Related stories
"From day one, I prioritized getting out in the field and talking to our teams in person and over company town halls," he said.
He also added that maintaining "long-term objectives and strategic direction" is the key to remaining resilient amid changing market conditions.
The incoming CEO now aims to revitalize Red Lobster's brand.
"Red Lobster is an iconic brand with a tremendous future," Adamolekun said in a statement .
The Harvard Business School alumnus said he plans to improve the customer experience and find new ways to engage with diners as he seeks to bring stability and growth back to the company.
Adamolekun is set to take over Red Lobster as CEO, pending a court approval next month of the company's sale to a buyer group led by Fortress Investment Group, the Wall Street Journal reported. The restaurant has about 550 locations across the US and Canada.
Watch: Red Lobster's fall is a case study on how to kill a business
- Main content
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- Animals (Basel)
- PMC10417731

Mastitis in Dairy Cattle: On-Farm Diagnostics and Future Perspectives
Associated data.
Not applicable.
Simple Summary
Mastitis is one of the most common diseases of the dairy industry and with it brings important economic losses. The most prevalent form of the disease is subclinical mastitis, which leads, in the absence of clinical signs, to decreased milk production, increased somatic cell count, and an increased risk of clinical mastitis during lactation. With the increasing public health concerns for antimicrobial use and its relationship with the development of antimicrobial resistance, nation-specific regulations and general pressure to reduce group-level prophylactic use of antimicrobials have been established. Selective dry therapy reserves antimicrobial treatment for cows or quarters suspected of having an intramammary infection. The treatment is administered after the last milking, while uninfected cows or quarters do not receive antibiotherapy. Since selective dry cow therapy was introduced, different methods of selecting infected cows or quarters have been reported. The aim of this article is to describe the management of mastitis in dairy cows and the main tools for its diagnosis, with a specific focus on on-farm instruments.
Mastitis is one of the most important diseases in dairy cattle farms, and it can affect the health status of the udder and the quantity and quality of milk yielded. The correct management of mastitis is based both on preventive and treatment action. With the increasing concern for antimicrobial resistance, it is strongly recommended to treat only the mammary quarters presenting intramammary infection. For this reason, a timely and accurate diagnosis is fundamental. The possibility to detect and characterize mastitis directly on farm would be very useful to choose the correct management protocol. Some on-field diagnostic tools are already routinely applied to detect mastitis, such as the California Mastitis Test and on-farm culture. Other instruments are emerging to perform a timely diagnosis and to characterize mastitis, such as Infra-Red Thermography, mammary ultrasound evaluation and blood gas analysis, even if their application still needs to be improved. The main purpose of this article is to present an overview of the methods currently used to control, detect, and characterize mastitis in dairy cows, in order to perform a timely diagnosis and to choose the most appropriate management protocol, with a specific focus on on-farm diagnostic tools.
1. Introduction
Mastitis, the inflammation of the mammary gland, is one of the most frequent diseases affecting dairy cows worldwide. It is responsible for approximately 60–70% of all antimicrobials administered on dairy farms [ 1 , 2 ]. It leads to severe economic losses, both direct and indirect. Direct costs associated with the treatment of the disease do contribute to overall total losses. Nonetheless, indirect costs such as discarding contaminated milk, losses in future milk production and quality, and increasing culling risk are substantially more consequential [ 3 ]. Several studies tried to estimate the losses due to mastitis infection. Two main different approaches have been used: observational and modelling or simulation approach. Indeed, several factors affect the estimation of costs, such as differences in the farming system, herd management and implementation rules of treatments, and differences both in the upper limit of milk production and in milk pricing according to tank SCC and other differences in prices of production factors [ 4 ]. Worldwide, published estimates of the economic losses of clinical mastitis range from EUR 61 to EUR 97 per cow on a farm, with large differences between countries; for example, in the Netherlands, losses due to clinical and subclinical mastitis varied between EUR 17 and EUR 198 per cow per year [ 5 ]. Underestimating the economic losses of mastitis can be regarded as a general problem in the dairy sector. To improve the adoption rate of advice and lower the incidence of mastitis, it is important to show farmers the real economic losses of mastitis on their farm [ 6 ].
It also represents a public health concern, considering that approximately 62% of isolated mastitis-causing agents are resistant to at least one antimicrobial agent, and that some of them are zoonotic agents [ 7 , 8 , 9 ]. For this reason, SCC has been set as a key component of national and international regulation for milk quality and commercialization. According to the US Food and Drug Administration, SCC official threshold for internal consumption is 750,000 cells/mL [ 10 ]. The US Pasteurized Milk Ordinance mandates that milk samples from all farms are officially tested at least four times in each six-month period. Under the current system, if two of four monthly tests exceed the 750,000 cells/mL, the producer is notified by the state regulators, and if three of five tests exceed the limit, then their milk permit is revoked [ 11 ]. However, recently, a European Union Health Certification Program has been introduced, specifying an SCC threshold of 400,000 cells/mL for companies exporting products into the EU [ 12 ]. Within the EU, the eligibility to supply raw milk for processing of dairy products for human consumption is governed by Regulations 853/2004 [ 13 ] and 854/2004 [ 14 ] using measurements of raw bulk tank milk SCC (BTSCC) to determine eligibility. According to these, BTSCC must be lower than 400,000 cells/mL, stated as a rolling geometric average over two or three months, with at least one sample collected per month. Moreover, in some countries, producers receive reimbursement for delivering milk with a low somatic count due to its more desirable technical characteristics and a longer shelf life [ 15 ].
Mastitis can be classified, according to its etiology, into environmental and contagious or, according to symptoms, into clinical and subclinical [ 2 ].
Being a multifactorial disease, mastitis susceptibility is given by multiple factors such as age, parity, lactation stage, milk yield, and udder anatomical dispositions. Two of the main factors are immunological condition and reactivity of the mammary gland. Consequently, the clinical manifestations as well as its further course depend on the interplay between the innate resistance and adaptive immunity of the dairy cow and the type, concentration, and virulence of udder pathogens [ 16 ].
Major contagious mastitis pathogens include Staphylococcus aureus , Streptococcus agalactiae , and Mycoplasma bovis . Environmental mastitis pathogens include a wide range of organisms, such as coliforms ( Escherichia coli , Klebsiella , Enterobacter , and Citrobacter ), environmental streptococci ( Streptococcus uberis and Streptococcus dysgalactiae ), Trueperella pyogenes , non- aureus staphylococci (NAS), and others such as Pseudomonas , Proteus , Serratia , Aerococcus , Listeria , yeast, and Prototheca [ 17 ].
A 2019 study [ 18 ] showed how pathogens were associated with specific histopathological patterns. Lymphoplasmacytic and suppurative mastitis were the main patterns observed with involvement of Streptococcus spp., coagulase-negative staphylococci (CNS), S. aureus , S. agalactiae , S. uberis , and Corynebacterium bovis . The pyogranulomatous pattern presented different forms depending on the agent involved and was primarily associated with S. aureus and Nocardia spp. The cases of abscedative mastitis were predominantly caused by T. pyogenes . The necrosuppurative pattern was predominantly associated with environmental bacteria producing endotoxins, such as E. coli . Granulomatous mastitis had the lowest frequency of cases and was occasionally associated with Mycobacterium spp.
In healthy lactating cows, macrophages are the dominant cell type found in milk and mammary tissues [ 19 ]. When bacteria enter the mammary gland through the teat canal, they multiply in the milk, enhancing an inflammatory response. Bacterial toxins, enzymes, and cell-wall components may have a direct effect on the function of the mammary epithelium. They also stimulate the production of numerous mediators of inflammation by inflammatory cells that may be directly involved in the pathogenesis of the disease [ 20 ]. These mediators include prostaglandins, leukotrienes, serotonin, histamine, complement components, and cytokines, such as tumor necrosis factor, interleukin-I, interleukin-6, interleukin-8, granulocyte colony-stimulating factor, and granulocyte-macrophage colony-stimulating factor. These bioactive compounds have various activities locally in infected tissues that direct movement and localization of neutrophils (and other leukocytes) from blood to milk [ 21 ]. The neutrophil recruitment into lacteal secretion results in the increase in the somatic cell count (SCC) [ 22 ].
Clinical mastitis is characterized by local and systemic clinical signs, alteration in milk composition and appearance, and elevated SCC. Subclinical mastitis manifests through production losses, lowered milk quality, and high SCC [ 23 ]. Dairy cows with subclinical mastitis might be overlooked because infected cows may not present any clinical signs. Moreover, available data demonstrate that subclinical mastitis is 15 to 40 times more prevalent than clinical mastitis. Thus, it is fundamental to find an accurate, effective, fast, and economic diagnostic tools in order to detect mastitis and apply an appropriate management protocol also in the absence of clinical signs [ 24 ].
The objective of this review is to describe the current approach to control, detect and characterize mastitis in the dairy cow, focusing on emerging diagnostic and on-farm tools, in order to promptly identify intramammary infection and to choose the most appropriate management protocol, also considering the increasingly concern for antimicrobial resistance.
2. Diagnosis and Control
2.1. diagnosis.
An efficient and effective mastitis control program requires the early detection of infection. This can be obtained by understanding the pathogenesis, discovering new sensitive tests for early screening, and adopting good managemental practices to reduce the chance of transmission of the infection from sick to healthy quarters [ 25 ].
Early detection of mastitis and identification of the causative agent are crucial for control and treatment [ 26 ]. These measures are fundamental in reducing costs, reducing losses in milk yield and milk quality, and increasing the cure rate of the infected animals [ 27 ]. Several studies demonstrated that a consequent early treatment significantly limited the severity of the disease and, in many cases, prevented the appearance of any visible signs of infection [ 28 ].
SCC and bacteria detection are the main tools to diagnose mastitis. In particular, bacteriological culture and PCR analysis are considered the gold standard.
Mammary infection is determined with a cell number above 200,000 cells/mL [ 29 ]. In addition, long-term elevated SCC levels suggest that the affected quarters are in a state of chronic inflammation and may interfere with the development of lactating tissues [ 30 ]. SCC varies during lactation, reaching a nadir at 30 DIM, coinciding with the peak of lactation, and gradually increasing in the remaining period [ 31 ]. Some authors have attributed high cell counts early in lactation to excessive shedding of epithelial cells in a small volume of milk. In fact, the mammary gland resumes function after a dormant period and in late lactation to a mere concentration of cells in a smaller volume of milk as milk production declines [ 32 ]. SCC has been proved to also be affected by parity, milking time, milking frequency, season, and udder infection [ 33 , 34 ]. A study has evaluated the variation in SCC patterns relating to the specific pathogen. In accordance with previous studies, for example, the authors found that intramammary infections (IMI) with S. aureus and S. uberis were often characterized by a long duration and high SCC; in the case of E. coli mastitis, instead, there was an association with short peaks [ 35 ].
New technologies, such as metabolomics, are emerging as tools for diagnosing and preventing mastitis. Metabolomics evaluates different changes in metabolites in affected cows in order to find effective biomarkers for timely and accurate prevention. Some interesting findings have been highlighted in two studies. The first has applied gas chromatography–mass spectrometry (GC-MS) on blood, finding as reliable the subclinical mastitis markers valine, serine, tyrosine, and phenylalanine 4–8 weeks pre-partum and valine, isoleucine, serine, and proline 4–8 weeks post-partum [ 36 ]. The second study has applied Nuclear Magnetic Resonance Metabolomics (NMR) on milk, finding that lactate, acetate, BHBA, butyrate, and isoleucine were in a higher concentration in high-SCC samples. On the other hand, for the same samples, lactose, hippurate, and fumarate were at lower levels than in milk with low SCC levels [ 37 ].
Also, recent molecular techniques such as MALDI-TOF and commercially available quantitative real time PCR (rtPCR) tests are becoming more common. MALDI-TOF is used to identify isolated organisms to genus and/or species level with a lower incidence of misclassification errors compared to traditional phenotypic speciation methods for some mastitis pathogens. Commercially available rtPCR tests reveal the advantages of being directly applied to milk samples containing preservatives: this procedure not only facilitates storage and shipment at room temperature, but it also enables the identification of organisms that are difficult to culture in a timely fashion [ 38 ].
Another group of widely investigated potential biomarkers are Acute Phase Proteins (APPs), already commonly employed as clinical biomarkers of inflammation in serum, such as milk isoforms of serum amyloid A (M-SAA) and haptoglobin (HP). Other proteins indicated as suitable mastitis markers are lactoferrin (LF) and cathelicidins (CATH) [ 25 , 39 ]. Some enzymes, such as Lactate dehydrogenase (LDH) and N-Acetyl-β-d-Glucosaminidase (NAGase), also seems to be reliable indicators of mastitis [ 40 ].
2.2. Control
The control program must include the strategic use of antimicrobials to curtail the problems of antibiotic residue in milk and antimicrobial resistance [ 41 ]. This implies the implementation of preventive measures. Most prevention activities focus on milking time and procedures. The use of management practices which reduce bacterial contamination of teat ends is a basic aspect of mastitis control. Pre-milking sanitation had usually been performed by washing udders, teats, pre-dipping, and post-dipping with water or disinfectants. Post-milking teat antiseptic was regarded as the single most effective practice to control IMI of lactating cows. This includes dipping or spraying the teat with an antiseptic formulation after milking. Antiseptic is sometimes associated with a filming agent in order to physically isolate the teat from the environment [ 23 , 42 ]. Furthermore, nutrition is an important factor in the resistance against disease. Deficiency of some trace substances and vitamins such as selenium, copper, zinc, and vitamin E have been found to be predisposing factors for mastitis. Dietary recommendation and possible supplementation of the lacking substances might significantly improve udder health [ 43 ]. Vaccination represents another preventive strategy. The use of vaccination, particularly with autogenous killed whole-cell vaccines, to try to control mastitis in dairy cattle is common. Several efforts have been made to develop a vaccine against mastitis, but few have claimed satisfactory outcomes, either in the field or on backyard farms. The main problem is that a single vaccine will not prevent mastitis caused by the multiple pathogens with different mechanisms of pathogenesis; if immunity is good against a certain agent, it does not protect from another one [ 44 ].
Nonetheless, the main strategy to treat mastitis still remains the administration of antibiotics. These can be given either by intramammary infusion or by intramuscular or intravenous injection [ 45 ]. For successful antibacterial mastitis therapy, the active medicine must achieve and conserve concentrations above the minimum inhibitory concentration (MIC) at the center of infection for long enough to breakdown the production and toxin-producing phase of the causal pathogen. This may be prohibited by several factors that include pathological changes in the udder parenchyma and mastitogenic bacterial and related factors [ 44 , 46 ]. Antibiotic administration can be performed either during lactation or the dry period. Treatment may be given during lactation for clinical mastitis and for some specific cases of subclinical mastitis. Treatment of clinical mastitis usually involves administering antibiotics to eliminate the causative organism from the quarter. A clinical response must be perceptible within 5–7 days [ 47 ]. Antibiotic therapy at drying-off has a double interest: first, to treat IMI in quarters contaminated at drying off (curative effect), and second, to prevent new infections at the beginning of the dry period [ 48 , 49 ]. For many years, blanket dry cow therapy (BDCT) has been the most common approach to mastitis control. This implied the infusion of all quarters of all cows with a long-acting intramammary antimicrobial at the end of the lactation. As concerns regarding the emergence of antimicrobial-resistant pathogens are on the rise, a protocol known as selective dry cow therapy (SDCT) has been applied. This establishes the treatment only of quarters with IMI at dry off [ 50 ]. Since SDCT was introduced, different methods of selecting infected cows or quarters have been reported: bacteriological culture in the laboratory, somatic cell counts (SCC), on-farm diagnostic tools and/or history of clinical mastitis. Many studies have demonstrated that the use of SDCT does not increase the risk of IMI at calving if internal teat sealants are used for all cows [ 51 ]. SDCT thus represents an effective treatment approach to reduce antimicrobial usage without affecting animal welfare or the farm economy [ 52 ].
3. On-Farm Diagnostic Tools
3.1. mammary ultrasound.
Ultrasonography is a non-invasive method for the diagnostics of various physiological and pathological conditions of the reproductive organs of ruminants [ 53 ]. It has also more recently been applied to the measurement of subcutaneous fat and for the diagnosis of respiratory diseases [ 54 , 55 ].
Many studies have demonstrated the utility of mammary ultrasound in identifying the presence of lesions or alterations of the mammary parenchyma and the teat, as well as in providing a valid indication of the outcome of treatment based on this evidence. Moreover, the possibility of pre-identifying animals for which intramammary antibiotic treatment could be ineffective would be an important element both in terms of antimicrobial resistance and for the economic impact of the disease itself [ 56 , 57 , 58 , 59 ]. The echographic examination of the mammary gland parenchyma in ruminant animals is performed primarily via the direct contact technique (transcutaneous echography). For this purpose, a linear, convex, or sector transducer can be used [ 56 ]. Ultrasonography of the normal udder parenchyma shows homogenous hypoechogenic parenchyma with interspersed anechoic blood vessel, milk alveoli, and lactiferous duct ( Figure 1 ).

Mammary ultrasound evaluation with longitudinal scanning (B mode). Frequency: 6.5 MHz. Depth: 150 mm. Focus: 50 mm.
The gland cisterns appear as a large homogenous anechoic area with few hypoechoic dots, which correspond to the milk [ 60 ]. A study performed on 52 dairy cattle proposed the classification of the parenchyma alterations in seven groups: physiological sonographic pattern (medium homogeneous echogenicity, isolated anechoic lactiferous ducts and vessels), non-homogeneous hyperechoic and few lactiferous ducts, abscesses (round, well-defined structures of varying size with a distinct capsule and hypoechoic content), non-homogeneous with hyperechoic zones and gas formation, hematoma (large septal spaces filled with anechoic up to hypoechoic fluids), non-homogeneous hypoechoic areas, and spotted hypoechoic areas. Consequently, a grading of the sonographic changes was also proposed as follows: physiological sonographic pattern, mild changes (only small percentages of the udder tissue are affected, with few obvious sonographic changes), moderate changes (larger areas of the udder tissue affected with more pronounced sonographic changes) and severe changes (normal udder structure is hardly extant anymore) [ 59 ]. Another study tried to examine whether ultrasonography could be used to predict the outcome of clinical mastitis. Udders were examined and classified according to on one or more of the following three signs: a hyperechoic spot in the parenchyma area, structural changes in the milk ducts, and non-homogenous parenchyma. According to the statistical analysis, only the presence of “non-homogeneous parenchyma” could be selected as a factor that predicts a marketable milk production [ 57 ].
An example of quarter with E. coli infection can be seen in Figure 2 , which shows the presence of hyperechoic spots and irregular aspect of the parenchyma.

Mammary ultrasound evaluation with longitudinal scanning (B mode) of a quarter with E.coli infection. Frequency: 6.5 MHz. Depth: 150 mm. Focus: 50 mm. Presence of multiple hyperechoic spots and irregular parenchyma.
Concerning B-mode echography of the teat in ruminants, instead, this is most often performed using the water bath technique and vertical scanning [ 56 ]. Under physiological conditions, the teat wall shows three layers; the skin appears as a thin, hyperechoic line, followed by the muscular/connective tissue layer containing blood vessels showing a thicker, homogenous, hypoechoic layer, and the innermost mucous membrane appears as a hyperechoic layer. A study evaluated the alterations of the teat structures in the case of subclinical mastitis. The teat cistern appeared as an anechoic area with few hypoechogenic dots according to the presence of milk content. There was an irregular outline of the teat canal and cistern, homogenous hypoechogenic contents, and loss of the three-layered appearance of the affected teat wall [ 60 ].
3.2. Blood Gas Analysis
The clinical status of the animal is equally crucial in assessing the success rate of treatment. Hematobiochemical testing is a useful and reliable indicator of the severity of syndromic conditions and is also a valuable tool in the diagnosis and prognosis of many diseases. Several studies have focused on the evaluation of the main alterations during mastitis, with the intention of also extending the use of blood parameters to this disease. The application of an Emogas analyzer would provide the possibility of obtaining prognostic index also in field condition and in real time [ 36 , 61 , 62 , 63 , 64 ]. Blood gas analysis is already extensively used in the evaluation and management of multiple pathologic condition. For example, in calves, it is useful to assess the acid–base status and to establish an appropriate treatment protocol in cases of respiratory disease and diarrhea [ 65 , 66 , 67 ].
Very recently, researchers investigated the variation of blood parameters at different milk production. Interestingly they found a direct correlation between pO 2 and milk production. This can probably be justified because the ability to transport oxygen is increased in cattle producing higher amounts of milk. Also, HCO 3 − was higher in high-producing cows. Finally, high glucose blood concentration was associated with low milk production, probably induced by the high rates of utilization needed to fulfill the requirements of the mammary gland for higher milk synthesis [ 68 ].
Several authors have investigated the application of ultrasound mammary evaluation in the case of mastitis. A study of 2022 tried to use the blood gas analyzer in cattle with high SCC to evaluate permeability in the mammary gland. The authors analyzed sodium (Na) and potassium (K) in particular, as they are considered to be important indexes for mammary permeability evaluation. The results showed higher levels of K, and a higher ratio of Na to K in animals with high SCC, suggesting the use of blood gas parameters for a rapid evaluation of the mammary gland health status [ 62 ]. Additionally, another article of 2016 showed levels of calcium, phosphorous, and zinc were significantly lower in the blood serum of infected cattle than in healthy cattle [ 69 ].
Finally, a study highlighted a decrease in glucose blood concentration in the case of clinical mastitis. Several authors explain this alteration with the concurrent presence of negative energy balance (NEB), very frequent in the transition period. Such NEB leads to significantly reduced serum glucose levels, thus affecting the energy supply of the immune system and predisposing animals also to mastitis onset [ 70 , 71 ]. Few other hypotheses have been made. For example, another possible explanation is that blood flows to the infected udder and result in a limited supply of blood sugar secreted by the cell. Another hypothesis is that glucose is transported from the milk to the extracellular pathway to maintain the osmotic balance between the extracellular environment and the milk with the increase in Na and chlorine (Cl) when the body suffers from mastitis. In addition, hypoglycemia may be due to the accumulation of cells at the site of infection, which depletes the glucose in the local area [ 36 ].
3.3. Electrical Conductivity
Electrical conductivity (EC) is a measure of the resistance of a particular material to an electric current. The electrical resistance of an electrolyte solution is defined as the resistance of a cube of the solution 1 cm 3 in volume. The conductivity is the reciprocal of the resistance. Resistance (impedance) is measured in ohms and is calculated by dividing voltage by amperes. Conductivity is measured in Siemens and is calculated by dividing ampere by voltage [ 72 ]. In milk, it is determined by the concentration of anions and cations, with Na, K, and Cl being the most important [ 73 ].
When a cow is exposed to IMI, tight junctions and the active Na, Cl, and K pumping system, characteristic of the mammary gland, are destroyed, leading to a leakage of these ions into the milk [ 74 ].
Several studies have demonstrated how EC can be influenced by several factors, such as milk fat content and other pathological processes. In particular, a 2009 study highlighted how milk fever was associated with the greatest increase in EC and the longest for cows diagnosed with pneumonia. Other metabolic and digestive problems such as ketosis, off feed, left displaced abomasum, as well as retained placenta, lameness, and other diseases were also associated with a significant increase in EC. Nonetheless, results also demonstrated a significant increase in EC as early as three days before detection of mild mastitis with other methodologies [ 75 ].
Different methods are applied to evaluate EC. In recent studies, an individual conductivity > 6.5 mS/cm and a difference between quarters with the highest and lowest conductivity > 1.0 mS/cm have been considered the cut-off for infection diagnosis [ 76 ].
A study highlighted that the accuracy of electrical conductivity compared favorably with other indirect methods, especially considering stripping milk rather than foremilk obtained after milk ejection [ 77 ]. Another study compared EC in case of mastitis caused by primary and secondary pathogens, observing in both significant differences with uninfected quarters. The authors also evaluated differential and absolute conductivity; the results from absolute conductivity were generally similar and sometimes superior to those from differential conductivity, especially when stripping were used [ 78 ]. Additionally, a research study from 1998 highlighted differences in infections detection relating to the bacteria. In fact, S. aureus and S. uberis were more readily detected than CNS infections. This may arise from less epithelial damage and inflammation generated by CNS and/or the possibility that such infections were localized in the teat canal, teat sinus or gland cistern and ductal regions [ 79 ].
The ability of EC to detect subclinical mastitis is still controversial, and further studies are needed, even if the association with other parameters significantly improve predictability [ 74 , 80 ].
3.4. California Mastitis Test
The California Mastitis Test (CMT) is an elementary, low-cost, quick evaluation that can be conducted on farm and is commonly used for identifying subclinical mastitis during milking [ 81 ]. Milk is appropriately collected in the CMT paddle, and an equivalent amount of the reagent (sodium alkyl aryl sulfonate) is added using a horizontal swirling motion for about 10–15 s. Sodium alkyl aryl sulfonate is an anionic surfactant that decreases surface tension, changes the structure and conductivity of cell membrane and nucleus, interferes with the osmotic balance, blocks oxidization, stimulates proteolytic enzymes, and increases milk viscosity [ 82 ]. The formation of gel reflects a leucocyte count of 200,000 to 5,000,000, indicative of IMI. Moreover, the purple color that results from the test is generally more intense in samples from infected quarters. This is because such samples have a basic or alkaline pH. The deep purple color indicates abnormal composition of milk due to mammary gland infection [ 83 ]. A study of 2014 also demonstrated a significant correlation between SCC and CMT, in which higher CMT scores corresponded to elevated trends of SCC [ 84 ]. These findings are supported by another article of 2012. Researchers divided CMT reaction into three groups, from the weakest to the strongest reaction (CMT+, CMT++, CMT+++) and compared them with SCC. In accordance with the previous article, they highlighted a direct correlation between these two diagnostic methods [ 85 ]. The use of the CMT to identify infected quarters has been extensively validated in cows that were not in early lactation [ 86 ]. Moreover, the possibility to identify not only clinical, but also subclinical mastitis at the end of lactation, before the dry off, would be very important in order to perform SDCT. A 1981 study reported that positive CMT scores in milk samples obtained several weeks before drying off predicted accurately IMI in over 80% of cases [ 87 ]. Another study demonstrated a high negative predictive value for IMI associated with the major pathogens performing CMT on the day of dry off [ 88 ]. Similar results have been highlighted in a 2012 article, in which CMT presented good negative predictive values, good sensitivity, even if positive predictive values and specificity were low [ 89 ]. Duplicate or multiple milk samples might positively affect the outcome of the test.
3.5. On-Farm Culture
On-farm culture (OFC) is a system which allows veterinarians to address the producers to the best strategic treatment decisions for clinical mastitis cases, without delay between the submission of milk samples and the reporting of results implied by laboratory culture. Initial on-farm culture systems were based on blood and MacConkey agar plates, which facilitated the categorization of microorganisms into Gram-positive, Gram-negative, or no growth within 24 to 32 h [ 90 ]. Several studies demonstrated its usefulness in guiding the decision of the most appropriate treatment decreasing intramammary antibiotic use, both in the short and long term. A study showed a decrease in intramammary antimicrobial use by half and a tendency to decrease withholding time by 1 day. This did not significantly affected days to clinical cure, bacteriological cure risk, and new infection risk or treatment failure risk within 21 days after the clinical mastitis event [ 91 ]. A companion paper, focused on long-term outcomes, demonstrated that selective treatment of clinical mastitis based on on-farm culture resulted in no difference, for example, concerning the recurrence of clinical mastitis in the same quarter, SCC, milk production, and cow survival for the rest of the lactation after clinical mastitis [ 92 ].
Chromogenic culture media are alternatives to rapid microbiological identification, as they make it possible to presumptively differentiate bacterial species and/or groups according to colony color [ 93 ]. The chromogenic substrate, when it contacts a specific microorganism after undergoing hydrolysis, releases a dye that sets in the microbial colonies, differentiating them by color [ 94 ]. According to a study, chromogenic media presented 100% Sensibility and 99.8% Specificity for the identification of S. aureus in clinical mastitis milk samples [ 95 ]. A study conducted on cases of subclinical mastitis caused by Gram-positive bacteria, showed satisfactory diagnostic performance results for the identification of the main pathogens, despite the limitation for the identification of S. aureus [ 96 ]. Another study performed comparing bi- and tri-plate both on clinical and subclinical mastitis suggested adequate accuracy for identification of S. agalactiae , S. dysgalactiae , S. uberis , E. coli , Klebsiella spp., and Enterobacter spp. [ 94 ]. However, the type of microorganism causing bovine mastitis and mastitis presentation influence diagnostic accuracy. Also, experience in milk microbiology substantially improved the interpretation of on-farm culture results, indicating that the observers’ experience is crucial to facilitate appropriate management decisions when adopting a selective treatment protocol [ 96 ]. Therefore, the use of chromogenic culture media on the dairy farm routine could be an alternative for the rapid identification of subclinical mastitis.
3.6. Infra-Red Thermography
The physical principal at the basis of the Infra-Red Thermography (IRT) is that a body that has a temperature higher than absolute zero emits electromagnetic radiation in the infrared spectrum. The relationship between the energy emitted by the body surface, the wavelength of this radiation and the temperature of the body is mathematically described. This radiation can be detected through a sensor array and used to build a thermographic image where the intensity, or the color, of each pixel is proportional to the corresponding temperature of the surface observed [ 97 , 98 ]. In a thermogram, the warmest region appears as white or red, whereas the coolest region appears as blue or black [ 99 ].
In bovine medicine, IRT is already extensively used for heat detection and prediction of ovulation in cows, detection and assessment of lameness, assessment of animal welfare, and feed utilization efficiency [ 100 ].
Mastitis is an inflammatory process of the mammary gland, leading to the production of prostaglandins and, consequently, to the increase in body and udder surface temperature, which can be detected via IRT ( Figure 3 ) [ 101 ].

Infra-Red Thermography of affected quarter. The presence of red color in correspondence with the teats and orifices indicates increased surface temperature. No relevant difference between udder and teat temperature is detected, which is present in healthy quarters.
Recently, several studies have also established a significant positive correlation between SCC and udder temperature. Performing thermal imaging, it is important to consider that IRT images can be influenced by several factors, such as mechanical brushing of the udder, direct solar radiation and wind speed, parity, stage of lactation, and pregnancy. It has also been shown that the temperature of the thermal images of the forequarters region of the udder had a greater correlation with SCC. This is because the rear region of the udder is more exposed to climatic variables and physical damage during milking, which may overestimate udder temperature during the IRT analysis. For this reason, it is fundamental to calibrate the thermal imager and to establish a standardized distance and acquisition method [ 101 , 102 ]. Commonly, an emissivity of 0.98 throughout is employed in accordance with previously published studies carried out on cow udders [ 97 ]. For the acquisition of thermogram, a study of the 2022 suggested photographing the udder from three angles to identify all quarters, and to photograph each one at least three times to exclude a low-quality image. Each angle had a range of distance and a range of tilt angle. Depending on the structure of the udder and the position of the cow in the stall, the specialist placed the thermal imager at 30–100 cm from the udder and teats of the animal at an angle of −15° to +15°. The choice of distance and angle of inclination of the thermal imager was based on the viewing angle of the thermal imager, the physiological dimensions of the udder, and the availability of all udder teats in the image. Thermographic images were obtained before the start of milking and after the end of milking, even if no significant difference between temperature of left and right udder quarters before and after milking was found [ 100 , 102 ].
4. Conclusions and Future Perspectives
Nowadays, mastitis still represents one of the most frequent diseases of dairy cows, having well-recognized detrimental effects on animal wellbeing and farming economy, and being one of the main causes of antimicrobial use in dairy farms. Due to the growing concern for antimicrobial resistance, regulations have been implemented nationally and internationally to reduce unnecessary antibiotic use. Not all IMIs require antibiotic treatment, and their administration must be based on the culture and sensitivity results rather than empirical therapy. Moreover, therapy success depends on several factors such as causal agent, parity, stage of lactation, other systemic diseases, and mammary parenchyma alterations. The management of mastitis must be under constant and continuous control of the veterinarian. This could certainly be facilitated by the possibility of establishing which animals are prone to have an improvement in treatment success; an improved treatment success would also result in a reduction in antimicrobial administration. Some on-farm diagnostic tools are already being routinely used to apply SDCT, such as CMT and OFC. IRT is a noninvasive and rapid method and can be of great value in early mastitis detection for optimum response from treatment. Even if it is an indirect measure, it has relatively good sensitivity, specificity, and positive and negative predictive values. Additionally, it can perceive changes in skin surface temperature in response to varying degrees of severity of the mammary gland infection. Ultrasound mammary evaluation has proved to be effective in identifying the presence of lesions or alterations of the mammary parenchyma and the teat. Such alterations might affect the prognosis of the animal. Also, blood gas analysis would provide interesting parameters to evaluate on farm and in real time the clinical conditions of the affected cows. These diagnostic techniques are still emerging in this field, and their application to detect mastitis and establish whether to treat a case of mastitis or not certainly has to be improved. Further studies are needed in order to establish objective parameters of detection and prognostic index.
Acknowledgments
This study was carried out within the Agritech National Research Center and received funding from the European Union Next-GenerationEU (PIANO NAZIONALE DI RIPRESA E RESILIENZA (PNRR)–MISSIONE 4 COMPONENTE 2, INVESTIMENTO 1.4–D.D. 1032 17/06/2022, CN00000022). This manuscript reflects only the authors’ views and opinions, and neither the European Union nor the European Commission can be considered responsible for them.
Funding Statement
This research was funded by European Union Next-GenerationEU (PIANO NAZIONALE DI RIPRESA E RESILIENZA (PNRR)–MISSIONE 4 COMPONENTE 2, INVESTIMENTO 1.4–D.D. 1032 17 June 2022, CN00000022).
Author Contributions
C.T., writing—original draft preparation; E.F., writing—review and editing; A.L., writing—review and editing; M.G., visualization and supervision. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Informed consent statement, data availability statement, conflicts of interest.
The authors declare no conflict of interest.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.

IMAGES
COMMENTS
The case follows her clinical presentation, assessments, diagnostics, and therapeutic interventions during the 45-day postpartum period. This case report highlights the situational and environmental context of the woman's ex-periences and emphasizes potential disconnections of care. Regarding her mastitis, the maternity care providers relied
Periductal mastitis is a benign inflammatory condition affecting the subareolar ducts and occurs most commonly in reproductive-aged women. Alternatively, IGM is a rare benign inflammatory condition that can clinically mimic breast cancer and occurs primarily in parous women ordinarily within 5 years of giving birth.
Idiopathic granulomatous mastitis (IGM) is a chronic inflammatory disease of the breast and mimics disorders such as breast cancer and breast abscess. Due to the uncommon of this disease, there is no definitive etiology, or treatment. A 38‐year‐old woman presented with a 3‐week history of painful right retro‐areolar mass.
Lactational mastitis is defined as inflammation of the breast tissue and is commonly experienced by breastfeeding women (Amir et al., 2007).It is a painful condition with high fever; flu-like symptoms, for example aches and chills; and red, tender, hot, and swollen areas of the breast (Lawrence, 1989; World Health Organization, 2000).It is diagnosed symptomatically and there is no broadly ...
Case: Mastitis. by Zach Winchester, MD, Margit Szabari MD, Hannah Milch, MD. Mastitis is defined as inflammation of the breast with or without infection. It is a relatively common condition typically affecting reproductive age women. Mastitis can be sub-categorized into lactational (puerperal) mastitis and non-lactational (non-puerperal) mastitis.
For most women, breastfeeding does not entail any major complications, but approximately 10-20% of women develop inflammation of the breast (mastitis) 5 - 7. Mastitis is an inflammatory reaction, either with or without a bacterial infection. The symptoms are red, painful, hot, swollen breasts, and sometimes fever, chills and flu-like symptoms.
Granulomatous mastitis (GM) is a chronic inflammatory mastitis disease that requires long-term treatment and has a high recurrence rate. Case management has been proven to be an effective mechanism in assisting patients with chronic illness to receive regular and targeted disease monitoring and health care service. The aim of this study was to investigate the application of a hospital-to ...
We report a case of granulomatous mastitis, an unusual presentation of breast disease. Granulomatous mastitis (GM) is an uncommon benign chronic inflammatory breast disease first described by Kessler and Wolloch in 1972 [] that can mimic inflammatory breast cancer and periductal mastitis [].It typically affects young women between 17 and 42 years of age within the reproductive and post ...
Amir L. H., Garland S. M., Lumley J. (2006). A case-control study of mastitis: Nasal carriage of Staphylococcus aureus. BioMed Central Family Practice, 7, 57.doi:10.1186 ... Marín M. (2014). Case-control study of risk factors for infectious mastitis in Spanish breastfeeding women. BioMed Central Pregnancy Childbirth, 14, 195.doi:10.1186/1471 ...
Mastitis can occur in lactating and nonlactating individuals, but lactational mastitis is the most common, with an incidence of 2% to 33% worldwide and approximately 10% in the United States. 1, 3 ...
Staphylococcus aureus is the most frequent pathogen isolated. Prompt and appropriate management of mastitis usually leads to a timely resolution and prevents complications, such as a breast abscess. Breast abscess requires both the removal of pus and antibiotic therapy. Interventions can include aspiration and incision and drainage procedures.
One study identified Candida species in the mouths of 34.55% of exclusively breastfed infants and in the mouths of 66.67% of strictly bottle-fed infants. Candida was found on the breasts of 34.55% of lactating women and on the breasts of 17.65% of nonlactating women. This information highlights the widespread colonization of Candida species ...
2.1 Incidence. Mastitis and breast abscess occur in all populations, whether or not breastfeeding is the norm. The reported incidence varies from a few to 33% of lactating women, but is usually under 10% (Table 1). Most studies have major methodological limitations, and there are no large prospective cohort studies.
Granulomatous mastitis: A case report. Koudouhonon Rita Oze, 1 Romeo Thierry Yehouenou Tessi, 1 Papys Mendes, 2 Nazik Allali, 1 Latifa Chat, 1 and Siham El Haddad 1 ... MRI in the study of this mastopathy is a technique that has shown suspicious lesions of malignancy in the form of irregular tissue masses. In 64% of cases, the lesions were ...
Streptococcus pneumoniae is an uncommon etiologic agent in soft-tissue infections. We report the case of a 35-year-old Caucasian woman who presented to our facility with puerperal pneumococcal mastitis, and review the only other three cases of pneumococcal mastitis described in the medical literature. The roles of the various pneumococcal vaccines in preventing this disease are discussed.
Mastitis is estimated to cost the US dairy industry $2 billion per year [].One of the most common mastitis causing pathogens remains Staphylococcus aureus (S. aureus), which can appear in both chronic and acute varieties, with markedly low cure rates [2, 3]. S. aureus is known to escape immune clearance by adhering and infiltrating epithelial cells of the mammary gland which contributes to the ...
Study design: A case. Place and duration of study: This study was carried out at Civil Hospital and Dow University of Health Sciences, Karachi and ... Clinical diagnosis was chronic mastitis in 40.9%, tuberculosis of breast in 36.3% and malignancy in 22.7% patients. Mammography showed an ill-defined mass in 45.5% and asymmetrical density in 31.8%.
Background Mastitis is a common problem for breastfeeding women. Researchers have called for an investigation into the possible role of maternal nasal carriage of S. aureus in the causation of mastitis in breastfeeding women. Methods The aim of the study was to investigate the role of maternal S. aureus nasal carriage in mastitis. Other factors such as infant nasal S. aureus carriage, nipple ...
Chapter - 6. Mastitis and Its Diagnosis: A Review. Abstract. Mastitis a persistent, inflammatory reactio n of the udder tissue in co ws. is a potentially fatal mammary gland infection and is the ...
This is the first study to report a case management model applied for GM patients. Although GM is a benign disease, its recurrence, one of the main challenges in the management of patients with the disease, has been reported to occur in 5%-50% of patients [ 10 - 12 ]. In our study, the recurrence rate of 11.2% is low in this range.
Mastitis is a very common maternal complication of lactation and contributes to early cessation of breastfeeding. Therapeutic ultrasound has been proposed as a non-invasive therapeutic alternative and has been added to the latest recommendations aimed at the treatment of mastitis spectrum disorders. In this article, we highlight the successful utilization of therapeutic ultrasound as part of ...
Intramammary bacterial infection is the primary reason of. bovine mastitis. Hence, Treatment of mastitis is solely based on. control of in fection by antibiotics. Case history and ob servations. A ...
Objective: To investigate a possible association between mastitis and breast cancer risk in a cohort of Greek women. Material and methods:A series of 343 women who visited two breast clinics in Greece and delivered live neonates were studied in our case-control retrospective study.The case group comprised women with breast cancer and the control group women without breast cancer.
Watch: Red Lobster's fall is a case study on how to kill a business. Advertisement. Two crossed lines that form an 'X'. It indicates a way to close an interaction, or dismiss a notification. ...
Another study compared EC in case of mastitis caused by primary and secondary pathogens, observing in both significant differences with uninfected quarters. The authors also evaluated differential and absolute conductivity; the results from absolute conductivity were generally similar and sometimes superior to those from differential ...