- Join our Mailing List

Working locally in primary care and collaborating globally to improve respiratory health
Clinical case study - asthma, clinical case study - asthma, resource information.
- Disease management
An official website of the United States government
The .gov means it's official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you're on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
- Browse Titles
NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-.

StatPearls [Internet].
Case study: 60-year-old female presenting with shortness of breath.
Deepa Rawat ; Sandeep Sharma .
Affiliations
Last Update: February 20, 2023 .
- Case Presentation
The patient is a 60-year-old white female presenting to the emergency department with acute onset shortness of breath. Symptoms began approximately 2 days before and had progressively worsened with no associated, aggravating, or relieving factors noted. She had similar symptoms approximately 1 year ago with an acute, chronic obstructive pulmonary disease (COPD) exacerbation requiring hospitalization. She uses BiPAP ventilatory support at night when sleeping and has requested to use this in the emergency department due to shortness of breath and wanting to sleep.
She denies fever, chills, cough, wheezing, sputum production, chest pain, palpitations, pressure, abdominal pain, abdominal distension, nausea, vomiting, and diarrhea.
She reports difficulty breathing at rest, forgetfulness, mild fatigue, feeling chilled, requiring blankets, increased urinary frequency, incontinence, and swelling in her bilateral lower extremities that are new-onset and worsening. Subsequently, she has not ambulated from bed for several days except to use the restroom due to feeling weak, fatigued, and short of breath.
There are no known ill contacts at home. Her family history includes significant heart disease and prostate malignancy in her father. Social history is positive for smoking tobacco use at 30 pack years. She quit smoking 2 years ago due to increasing shortness of breath. She denies all alcohol and illegal drug use. There are no known foods, drugs, or environmental allergies.
Past medical history is significant for coronary artery disease, myocardial infarction, COPD, hypertension, hyperlipidemia, hypothyroidism, diabetes mellitus, peripheral vascular disease, tobacco usage, and obesity. Past surgical history is significant for an appendectomy, cardiac catheterization with stent placement, hysterectomy, and nephrectomy.
Her current medications include fluticasone-vilanterol 100-25 mcg inhaled daily, hydralazine 50 mg by mouth, 3 times per day, hydrochlorothiazide 25 mg by mouth daily, albuterol-ipratropium inhaled every 4 hours PRN, levothyroxine 175 mcg by mouth daily, metformin 500 mg by mouth twice per day, nebivolol 5 mg by mouth daily, aspirin 81 mg by mouth daily, vitamin D3 1000 units by mouth daily, clopidogrel 75 mg by mouth daily, isosorbide mononitrate 60 mg by mouth daily, and rosuvastatin 40 mg by mouth daily.
Physical Exam
Initial physical exam reveals temperature 97.3 F, heart rate 74 bpm, respiratory rate 24, BP 104/54, HT 160 cm, WT 100 kg, BMI 39.1, and O2 saturation 90% on room air.
Constitutional: Extremely obese, acutely ill-appearing female. Well-developed and well-nourished with BiPAP in place. Lying on a hospital stretcher under 3 blankets.
HEENT:
- Head: Normocephalic and atraumatic
- Mouth: Moist mucous membranes
- Macroglossia
- Eyes: Conjunctiva and EOM are normal. Pupils are equal, round, and reactive to light. No scleral icterus. Bilateral periorbital edema present.
- Neck: Neck supple. No JVD present. No masses or surgical scarring.
- Throat: Patent and moist
Cardiovascular: Normal rate, regular rhythm, and normal heart sound with no murmur. 2+ pitting edema bilateral lower extremities and strong pulses in all four extremities.
Pulmonary/Chest: No respiratory status distress at this time, tachypnea present, (+) wheezing noted, bilateral rhonchi, decreased air movement bilaterally. The patient was barely able to finish a full sentence due to shortness of breath.
Abdominal: Soft. Obese. Bowel sounds are normal. No distension and no tenderness
Skin: Skin is very dry
Neurologic: Alert, awake, able to protect her airway. Moving all extremities. No sensation losses
- Initial Evaluation
Initial evaluation to elucidate the source of dyspnea was performed and included CBC to establish if an infectious or anemic source was present, CMP to review electrolyte balance and review renal function, and arterial blood gas to determine the PO2 for hypoxia and any major acid-base derangement, creatinine kinase and troponin I to evaluate the presence of myocardial infarct or rhabdomyolysis, brain natriuretic peptide, ECG, and chest x-ray. Considering that it is winter and influenza is endemic in the community, a rapid influenza assay was obtained as well.
Largely unremarkable and non-contributory to establish a diagnosis.
Showed creatinine elevation above baseline from 1.08 base to 1.81, indicating possible acute injury. EGFR at 28 is consistent with chronic renal disease. Calcium was elevated to 10.2. However, when corrected for albumin, this corrected to 9.8 mg/dL. Mild transaminitis is present as seen in alkaline phosphatase, AST, and ALT measurements which could be due to liver congestion from volume overload.
Initial arterial blood gas with pH 7.491, PCO2 27.6, PO2 53.6, HCO3 20.6, and oxygen saturation 90% on room air, indicating respiratory alkalosis with hypoxic respiratory features.
Creatinine kinase was elevated along with serial elevated troponin I studies. In the setting of her known chronic renal failure and acute injury indicated by the above creatinine value, a differential of rhabdomyolysis is determined.
Influenza A and B: Negative
Normal sinus rhythm with non-specific ST changes in inferior leads. Decreased voltage in leads I, III, aVR, aVL, aVF.
Chest X-ray
Findings: Bibasilar airspace disease that may represent alveolar edema. Cardiomegaly noted. Prominent interstitial markings were noted. Small bilateral pleural effusions
Radiologist Impression: Radiographic changes of congestive failure with bilateral pleural effusions greater on the left compared to the right
- Differential Diagnosis
- Acute on chronic COPD exacerbation
- Acute on chronic renal failure
- Bacterial pneumonia
- Congestive heart failure
- Pericardial effusion
- Hypothyroidism
- Influenza pneumonia
- Pulmonary edema
- Pulmonary embolism
- Confirmatory Evaluation
On the second day of the admission patient’s shortness of breath was not improved, and she was more confused with difficulty arousing on conversation and examination. To further elucidate the etiology of her shortness of breath and confusion, the patient's husband provided further history. He revealed that she is poorly compliant with taking her medications. He reports that she “doesn’t see the need to take so many pills.”
Testing was performed to include TSH, free T4, BNP, repeated arterial blood gas, CT scan of the chest, and echocardiogram. TSH and free T4 evaluate hypothyroidism. BNP evaluates fluid load status and possible congestive heart failure. CT scan of the chest will look for anatomical abnormalities. An echocardiogram is used to evaluate left ventricular ejection fraction, right ventricular function, pulmonary artery pressure, valvular function, pericardial effusion, and any hypokinetic area.
- TSH: 112.717 (H)
- Free T4: 0.56 (L)
- TSH and Free T4 values indicate severe primary hypothyroidism.
BNP can be falsely low in obese patients due to the increased surface area. Additionally, adipose tissue has BNP receptors which augment the true BNP value. Also, African American patients with more excretion may have falsely low values secondary to greater excretion of BNP. This test is not that helpful in renal failure due to the chronic nature of fluid overload. This allows for desensitization of the cardiac tissues with a subsequent decrease in BNP release.
Repeat arterial blood gas on BiPAP ventilation shows pH 7.397, PCO2 35.3, PO2 72.4, HCO3 21.2, and oxygen saturation 90% on 2 L supplemental oxygen.
CT chest without contrast was primarily obtained to evaluate the left hemithorax, especially the retrocardiac area.
Radiologist Impression: Tiny bilateral pleural effusions. Pericardial effusion. Coronary artery calcification. Some left lung base atelectasis with minimal airspace disease.
Echocardiogram
The left ventricular systolic function is normal. The left ventricular cavity is borderline dilated.
The pericardial fluid is collected primarily posteriorly, laterally but not apically. There appeared to be a subtle, early hemodynamic effect of the pericardial fluid on the right-sided chambers by way of an early diastolic collapse of the RA/RV and delayed RV expansion until late diastole. A dedicated tamponade study was not performed.
The estimated ejection fraction appears to be in the range of 66% to 70%. The left ventricular cavity is borderline dilated.
The aortic valve is abnormal in structure and exhibits sclerosis.
The mitral valve is abnormal in structure. Mild mitral annular calcification is present. There is bilateral thickening present. Trace mitral valve regurgitation is present.
- Myxedema coma or severe hypothyroidism
- Pericardial effusion secondary to myxedema coma
- COPD exacerbation
- Acute on chronic hypoxic respiratory failure
- Acute respiratory alkalosis
- Bilateral community-acquired pneumonia
- Small bilateral pleural effusions
- Acute mild rhabdomyolysis
- Acute chronic, stage IV, renal failure
- Elevated troponin I levels, likely secondary to Renal failure
- Diabetes mellitus type 2, non-insulin-dependent
- Extreme obesity
- Hepatic dysfunction
The patient was extremely ill and rapidly decompensating with multisystem organ failure, including respiratory failure, altered mental status, acute on chronic renal failure, and cardiac dysfunction. The primary concerns for the stability of the patient revolved around respiratory failure coupled with altered mental status. In the intensive care unit (ICU), she rapidly began to fail BiPAP therapy. Subsequently, the patient was emergently intubated in the ICU. A systemic review of therapies and hospital course is as follows:
Considering the primary diagnosis of myxedema coma, early supplementation with thyroid hormone is essential. Healthcare providers followed the American Thyroid Association recommendations, which recommend giving combined T3 and T4 supplementation; however, T4 alone may also be used. T3 therapy is given as a bolus of 5 to 20 micrograms intravenously and continued at 2.5 to 10 micrograms every 8 hours. An intravenous loading dose of 300 to 600 micrograms of T4 is followed by a daily intravenous dose of 50 to 100 micrograms. Repeated monitoring of TSH and T4 should be performed every 1 to 2 days to evaluate the effect and to titrate the dose of medication. The goal is to improve mental function. Until coexistent adrenal insufficiency is ruled out using a random serum cortisol measurement, 50 to 100 mg every 8 hours of hydrocortisone should be administered. In this case, clinicians used hydrocortisone 100 mg IV every 8 hours. Dexamethasone 2 to 4 mg every 12 hours is an alternative therapy.
The patient’s mental status rapidly worsened despite therapy. In the setting of her hypothyroidism history, this may be myxedema coma or due to the involvement of another organ system. The thyroid supplementation medications and hydrocortisone were continued. A CT head without contrast was normal.
Respiratory
For worsening metabolic acidosis and airway protection, the patient was emergently intubated. Her airway was deemed high risk due to having a large tongue, short neck, and extreme obesity. As the patient’s heart was preload dependent secondary to pericardial effusion, a 1-liter normal saline bolus was started. Norepinephrine was started at a low dose for vasopressor support, and ketamine with low dose Propofol was used for sedation. Ketamine is a sympathomimetic medication and usually does not cause hypotension as all other sedatives do. The patient was ventilated with AC mode of ventilation, tidal volume of 6 ml/kg ideal body weight, flow 70, initial fio2 100 %, rate 26 per minute (to compensate for metabolic acidosis), PEEP of 8.
Cardiovascular
She was determined to be hemodynamically stable with a pericardial effusion. This patient’s cardiac dysfunction was diastolic in nature, as suggested by an ejection fraction of 66% to 70%. The finding of posterior pericardial effusion further supported this conclusion. The posterior nature of this effusion was not amenable to pericardiocentesis. As such, this patient was preload dependent and showed signs of hypotension. The need for crystalloid fluid resuscitation was balanced against the impact increased intravascular volume would have on congestive heart failure and fluid overload status. Thyroid hormone replacement as above should improve hypotension. However, vasopressor agents may be used to maintain vital organ perfusion targeting a mean arterial pressure of greater than 65 mm Hg as needed. BP improved after fluid bolus, and eventually, the norepinephrine was stopped. Serial echocardiograms were obtained to ensure that the patient did not develop tamponade physiology. Total CK was elevated, which was likely due to Hypothyroidism compounded with chronic renal disease.
Infectious Disease
Blood cultures, urine analysis, and sputum cultures were obtained. The patient's white blood cell count was normal. This is likely secondary to her being immunocompromised due to hypothyroidism and diabetes. In part, the pulmonary findings of diffuse edema and bilateral pleural effusions can be explained by cardiac dysfunction. Thoracentesis of pleural fluid was attempted, and the fluid was analyzed for cytology and gram staining to rule out infectious or malignant causes as both a therapeutic and diagnostic measure. Until these results return, broad-spectrum antibiotics are indicated and may be discontinued once the infection is ruled out completely.
Gastrointestinal
Nasogastric tube feedings were started on the patient after intubation. She tolerated feedings well. AST and ALT were mildly elevated, which was thought to be due to hypothyroidism, and as the TSH and free T4 improved, her AST and ALT improved. Eventually, these values became normal once her TSH level was close to 50.
Her baseline creatinine was found to be close to 1.08 in prior medical records. She presented with a creatinine of 1.8 in the emergency department. Since hypothyroidism causes fluid retention in part because thyroid hormone encourages excretion of free water and partly due to decreased lymphatic function in returning fluid to vascular circulation. Aggressive diuresis was attempted. As a result, her creatinine increased initially but improved on repeated evaluation, and the patient had a new baseline creatinine of 1.6. Overall she had a net change in the fluid status of 10 liters negative by her ten days of admission in the ICU.
Mildly anemic otherwise, WBC and platelet counts were normal. Electrolyte balance should be monitored closely, paying attention to sodium, potassium, chloride, and calcium specifically as these are worsened in both renal failure and myxedema.
Daily sedation vacations were enacted, and the patient's mental status improved and was much better when TSH was around 20. The bilateral pleural effusions improved with aggressive diuresis. Breathing trials were initiated when the patient's fio2 requirements decreased to 60% and a PEEP of 8. She was eventually extubated onto BiPAP and then high-flow nasal cannula while off of BiPAP. Pericardial fluid remained stable, and no cardiac tamponade pathology developed. As a result, it was determined that a pericardial window was unnecessary. Furthermore, she was not a candidate for pericardiocentesis as the pericardial effusion was located posterior to the heart. Her renal failure improved with improved cardiac function, diuretics, and thyroid hormone replacement.
After extubation patient had speech and swallow evaluations and was able to resume an oral diet. The patient was eventually transferred out of the ICU to the general medical floor and eventually to a rehabilitation unit.
Despite the name myxedema coma, most patients will not present in a coma status. This illness is at its core a severe hypothyroidism crisis that leads to systemic multiorgan failure. Thyroid hormones T3, and to a lesser extent, T4 act directly on a cellular level to upregulate all metabolic processes in the body. Therefore, deficiency of this hormone is characterized by systemic decreased metabolism and decreased glucose utilization along with increased production and storage of osmotically active mucopolysaccharide protein complexes into peripheral tissues resulting in diffuse edema and swelling of tissue. [1]
Myxedema coma is an illness that occurs primarily in females at a rate of 4:1 compared to men. It typically impacts the elderly at the age of greater than 60 years old, and approximately 90% of cases occur during the winter months. Myxedema coma is the product of longstanding unidentified or undertreated hypothyroidism of any etiology. Thyroid hormone is necessary throughout the body and acts as a regulatory hormone that affects many organ systems. [2] In cardiac tissues, myxedema coma manifests as decreased contractility with subsequent reduction in stroke volume and overall cardiac output. Bradycardia and hypotension are typically present also. Pericardial effusions occur due to the accumulation of mucopolysaccharides in the pericardial sac, which leads to worsened cardiac function and congestive heart failure from diastolic dysfunction. Capillary permeability is also increased throughout the body leading to worsened edema. Electrocardiogram findings may include bradycardia and low-voltage, non-specific ST waveform changes with possible inverted T waves.
Neurologic tissues are impacted in myxedema coma leading to the pathognomonic altered mental status resulting from hypoxia and decreased cerebral blood flow secondary to cardiac dysfunction as above. Additionally, hypothyroidism leads to decreased glucose uptake and utilization in neurological tissue, thus worsening cognitive function.
The pulmonary system typically manifests this disease process through hypoventilation secondary to the central nervous system (CNS) depression of the respiratory drive with blunting of the response to hypoxia and hypercapnia. Additionally, metabolic dysfunction in the muscles of respiration leads to respiratory fatigue and failure, macroglossia from mucopolysaccharide driven edema of the tongue leads to mechanical obstruction of the airway, and obesity hypoventilation syndrome with the decreased respiratory drive as most hypothyroid patients suffer from obesity.
Renal manifestations include decreased glomerular filtration rate from the reduced cardiac output and increased systemic vascular resistance coupled with acute rhabdomyolysis lead to acute kidney injury. In the case of our patient above who has a pre-existing renal disease status post-nephrectomy, this is further worsened. The net effect is worsened fluid overload status compounding the cardiac dysfunction and edema. [3]
The gastrointestinal tract is marked by mucopolysaccharide-driven edema as well leading to malabsorption of nutrients, gastric ileus, and decreased peristalsis. Ascites is common because of increased capillary permeability in the intestines coupled with coexistent congestive heart failure and congestive hepatic failure. Coagulopathies are common to occur as a result of this hepatic dysfunction.
Evaluation: The diagnosis of myxedema coma, as with all other diseases, is heavily reliant on the history and physical exam. A past medical history including hypothyroidism is highly significant whenever decreased mental status or coma is identified. In the absence of identified hypothyroidism, myxedema coma is a diagnosis of exclusion when all other sources of coma have been ruled out. If myxedema coma is suspected, evaluation of thyroid-stimulating hormone (TSH), free thyroxine (T4), and serum cortisol is warranted. T4 will be extremely low. TSH is variable depending on the etiology of hypothyroidism, with a high TSH indicating primary hypothyroidism and a low or normal TSH indicating secondary etiologies. Cortisol may be low indicating adrenal insufficiency because of hypothyroidism. [4]
Prognosis: Myxedema coma is a medical emergency. With proper and rapid diagnosis and initiation of therapy, the mortality rate is still as high as 25% to 50%. The most common cause of death is due to respiratory failure. The factors which suggest a poorer prognosis include increased age, persistent hypothermia, bradycardia, low score Glasgow Coma Scale, or multi-organ impairment indicated by high APACHE (Acute Physiology and Chronic Health Evaluation) II score. For these reasons, placement in an intensive care unit with a low threshold for intubation and mechanical ventilation can improve mortality outcomes. [3] [5]
- Pearls of Wisdom
- Not every case of shortness of breath is COPD or congestive heart failure (CHF). While less likely, a history of hypothyroidism should raise suspicion of myxedema coma in a patient with any cognitive changes.
- Myxedema is the great imitator illness that impacts all organ systems. It can easily be mistaken for congestive heart failure, COPD exacerbation, pneumonia, renal injury or failure, or neurological insult.
- Initial steps in therapy include aggressive airway management, thyroid hormone replacement, glucocorticoid therapy, and supportive measures.
- These patients should be monitored in an intensive care environment with continuous telemetry. [6]
- Enhancing Healthcare Team Outcomes
This case demonstrates how all interprofessional healthcare team members need to be involved in arriving at a correct diagnosis, particularly in more challenging cases such as this one. Clinicians, specialists, nurses, pharmacists, laboratory technicians all bear responsibility for carrying out the duties pertaining to their particular discipline and sharing any findings with all team members. An incorrect diagnosis will almost inevitably lead to incorrect treatment, so coordinated activity, open communication, and empowerment to voice concerns are all part of the dynamic that needs to drive such cases so patients will attain the best possible outcomes.
- Review Questions
- Access free multiple choice questions on this topic.
- Comment on this article.
Case Study of 60 year old female presenting with Shortness of Breath Contributed by Sandeep Sharma, MD
Disclosure: Deepa Rawat declares no relevant financial relationships with ineligible companies.
Disclosure: Sandeep Sharma declares no relevant financial relationships with ineligible companies.
This book is distributed under the terms of the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0) ( http://creativecommons.org/licenses/by-nc-nd/4.0/ ), which permits others to distribute the work, provided that the article is not altered or used commercially. You are not required to obtain permission to distribute this article, provided that you credit the author and journal.
- Cite this Page Rawat D, Sharma S. Case Study: 60-Year-Old Female Presenting With Shortness of Breath. [Updated 2023 Feb 20]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-.
In this Page
Bulk download.
- Bulk download StatPearls data from FTP
Related information
- PubMed Links to PubMed
Similar articles in PubMed
- Case Study: 33-Year-Old Female Presents with Chronic SOB and Cough. [StatPearls. 2024] Case Study: 33-Year-Old Female Presents with Chronic SOB and Cough. Sharma S, Hashmi MF, Rawat D. StatPearls. 2024 Jan
- Acute Exacerbation of COPD. [J Educ Teach Emerg Med. 2023] Acute Exacerbation of COPD. Pappas D, Vempati A. J Educ Teach Emerg Med. 2023 Apr; 8(2):S35-S61. Epub 2023 Apr 30.
- Review Breathlessness with pulmonary metastases: a multimodal approach. [J Adv Pract Oncol. 2013] Review Breathlessness with pulmonary metastases: a multimodal approach. Brant JM. J Adv Pract Oncol. 2013 Nov; 4(6):415-22.
- Stress Cardiomyopathy in the Setting of COPD Exacerbation. [J Investig Med High Impact Cas...] Stress Cardiomyopathy in the Setting of COPD Exacerbation. Landefeld K, Saleh Q, Sander GE. J Investig Med High Impact Case Rep. 2015 Oct-Dec; 3(4):2324709615612847. Epub 2015 Oct 14.
- Review Palliative Chemotherapy: Does It Only Provide False Hope? The Role of Palliative Care in a Young Patient With Newly Diagnosed Metastatic Adenocarcinoma. [J Adv Pract Oncol. 2017] Review Palliative Chemotherapy: Does It Only Provide False Hope? The Role of Palliative Care in a Young Patient With Newly Diagnosed Metastatic Adenocarcinoma. Doverspike L, Kurtz S, Selvaggi K. J Adv Pract Oncol. 2017 May-Jun; 8(4):382-386. Epub 2017 May 1.
Recent Activity
- Case Study: 60-Year-Old Female Presenting With Shortness of Breath - StatPearls Case Study: 60-Year-Old Female Presenting With Shortness of Breath - StatPearls
Your browsing activity is empty.
Activity recording is turned off.
Turn recording back on
Connect with NLM
National Library of Medicine 8600 Rockville Pike Bethesda, MD 20894
Web Policies FOIA HHS Vulnerability Disclosure
Help Accessibility Careers
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- My Account Login
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
- Perspective
- Open access
- Published: 16 October 2014
A woman with asthma: a whole systems approach to supporting self-management
- Hilary Pinnock 1 ,
- Elisabeth Ehrlich 1 ,
- Gaylor Hoskins 2 &
- Ron Tomlins 3
npj Primary Care Respiratory Medicine volume 24 , Article number: 14063 ( 2014 ) Cite this article
16k Accesses
2 Citations
6 Altmetric
Metrics details
- Health care
A 35-year-old lady attends for review of her asthma following an acute exacerbation. There is an extensive evidence base for supported self-management for people living with asthma, and international and national guidelines emphasise the importance of providing a written asthma action plan. Effective implementation of this recommendation for the lady in this case study is considered from the perspective of a patient, healthcare professional, and the organisation. The patient emphasises the importance of developing a partnership based on honesty and trust, the need for adherence to monitoring and regular treatment, and involvement of family support. The professional considers the provision of asthma self-management in the context of a structured review, with a focus on a self-management discussion which elicits the patient’s goals and preferences. The organisation has a crucial role in promoting, enabling and providing resources to support professionals to provide self-management. The patient’s asthma control was assessed and management optimised in two structured reviews. Her goal was to avoid disruption to her work and her personalised action plan focused on achieving that goal.
Similar content being viewed by others

Psilocybin microdosers demonstrate greater observed improvements in mood and mental health at one month relative to non-microdosing controls
Joseph M. Rootman, Maggie Kiraga, … Zach Walsh

An overview of clinical decision support systems: benefits, risks, and strategies for success
Reed T. Sutton, David Pincock, … Karen I. Kroeker

Key recommendations for primary care from the 2022 Global Initiative for Asthma (GINA) update
Mark L. Levy, Leonard B. Bacharier, … Helen K. Reddel
A 35-year-old sales representative attends the practice for an asthma review. Her medical record notes that she has had asthma since childhood, and although for many months of the year her asthma is well controlled (when she often reduces or stops her inhaled steroids), she experiences one or two exacerbations a year requiring oral steroids. These are usually triggered by a viral upper respiratory infection, though last summer when the pollen count was particularly high she became tight chested and wheezy for a couple of weeks.
Her regular prescription is for fluticasone 100 mcg twice a day, and salbutamol as required. She has a young family and a busy lifestyle so does not often manage to find time to attend the asthma clinic. A few weeks previously, an asthma attack had interfered with some important work-related travel, and she has attended the clinic on this occasion to ask about how this can be managed better in the future. There is no record of her having been given an asthma action plan.
What do we know about asthma self-management? The academic perspective
Supported self-management reduces asthma morbidity.
The lady in this case study is struggling to maintain control of her asthma within the context of her busy professional and domestic life. The recent unfortunate experience which triggered this consultation offers a rare opportunity to engage with her and discuss how she can manage her asthma better. It behoves the clinician whom she is seeing (regardless of whether this is in a dedicated asthma clinic or an appointment in a routine general practice surgery) to grasp the opportunity and discuss self-management and provide her with a (written) personalised asthma action plan (PAAP).
The healthcare professional advising the lady is likely to be aware that international and national guidelines emphasise the importance of supporting self-management. 1 – 4 There is an extensive evidence base for asthma self-management: a recent synthesis identified 22 systematic reviews summarising data from 260 randomised controlled trials encompassing a broad range of demographic, clinical and healthcare contexts, which concluded that asthma self-management reduces emergency use of healthcare resources, including emergency department visits, hospital admissions and unscheduled consultations and improves markers of asthma control, including reduced symptoms and days off work, and improves quality of life. 1 , 2 , 5 – 12 Health economic analysis suggests that it is not only clinically effective, but also a cost-effective intervention. 13
Personalised asthma action plans
Key features of effective self-management approaches are:
Self-management education should be reinforced by provision of a (written) PAAP which reminds patients of their regular treatment, how to monitor and recognise that control is deteriorating and the action they should take. 14 – 16 As an adult, our patient can choose whether she wishes to monitor her control with symptoms or by recording peak flows (or a combination of both). 6 , 8 , 9 , 14 Symptom-based monitoring is generally better in children. 15 , 16
Plans should have between two and three action points including emergency doses of reliever medication; increasing low dose (or recommencing) inhaled steroids; or starting a course of oral steroids according to severity of the exacerbation. 14
Personalisation of the action plan is crucial. Focussing specifically on what actions she could take to prevent a repetition of the recent attack is likely to engage her interest. Not all patients will wish to start oral steroids without advice from a healthcare professional, though with her busy lifestyle and travel our patient is likely to be keen to have an emergency supply of prednisolone. Mobile technology has the potential to support self-management, 17 , 18 though a recent systematic review concluded that none of the currently available smart phone ‘apps’ were fit for purpose. 19
Identification and avoidance of her triggers is important. As pollen seems to be a trigger, management of allergic rhinitis needs to be discussed (and included in her action plan): she may benefit from regular use of a nasal steroid spray during the season. 20
Self-management as recommended by guidelines, 1 , 2 focuses narrowly on adherence to medication/monitoring and the early recognition/remediation of exacerbations, summarised in (written) PAAPs. Patients, however, may want to discuss how to reduce the impact of asthma on their life more generally, 21 including non-pharmacological approaches.
Supported self-management
The impact is greater if self-management education is delivered within a comprehensive programme of accessible, proactive asthma care, 22 and needs to be supported by ongoing regular review. 6 With her busy lifestyle, our patient may be reluctant to attend follow-up appointments, and once her asthma is controlled it may be possible to make convenient arrangements for professional review perhaps by telephone, 23 , 24 or e-mail. Flexible access to professional advice (e.g., utilising diverse modes of consultation) is an important component of supporting self-management. 25
The challenge of implementation
Implementation of self-management, however, remains poor in routine clinical practice. A recent Asthma UK web-survey estimated that only 24% of people with asthma in the UK currently have a PAAP, 26 with similar figures from Sweden 27 and Australia. 28 The general practitioner may feel that they do not have time to discuss self-management in a routine surgery appointment, or may not have a supply of paper-based PAAPs readily available. 29 However, as our patient rarely finds time to attend the practice, inviting her to make an appointment for a future clinic is likely to be unsuccessful and the opportunity to provide the help she needs will be missed.
The solution will need a whole systems approach
A systematic meta-review of implementing supported self-management in long-term conditions (including asthma) concluded that effective implementation was multifaceted and multidisciplinary; engaging patients, training and motivating professionals within the context of an organisation which actively supported self-management. 5 This whole systems approach considers that although patient education, professional training and organisational support are all essential components of successful support, they are rarely effective in isolation. 30 A systematic review of interventions that promote provision/use of PAAPs highlighted the importance of organisational systems (e.g., sending blank PAAPs with recall reminders). 31 A patient offers her perspective ( Box 1 ), a healthcare professional considers the clinical challenge, and the challenges are discussed from an organisational perspective.
Box 1: What self-management help should this lady expect from her general practitioner or asthma nurse? The patient’s perspective
The first priority is that the patient is reassured that her condition can be managed successfully both in the short and the long term. A good working relationship with the health professional is essential to achieve this outcome. Developing trust between patient and healthcare professional is more likely to lead to the patient following the PAAP on a long-term basis.
A review of all medication and possible alternative treatments should be discussed. The patient needs to understand why any changes are being made and when she can expect to see improvements in her condition. Be honest, as sometimes it will be necessary to adjust dosages before benefits are experienced. Be positive. ‘There are a number of things we can do to try to reduce the impact of asthma on your daily life’. ‘Preventer treatment can protect against the effect of pollen in the hay fever season’. If possible, the same healthcare professional should see the patient at all follow-up appointments as this builds trust and a feeling of working together to achieve the aim of better self-management.
Is the healthcare professional sure that the patient knows how to take her medication and that it is taken at the same time each day? The patient needs to understand the benefit of such a routine. Medication taken regularly at the same time each day is part of any self-management regime. If the patient is unused to taking medication at the same time each day then keeping a record on paper or with an electronic device could help. Possibly the patient could be encouraged to set up a system of reminders by text or smartphone.
Some people find having a peak flow meter useful. Knowing one's usual reading means that any fall can act as an early warning to put the PAAP into action. Patients need to be proactive here and take responsibility.
Ongoing support is essential for this patient to ensure that she takes her medication appropriately. Someone needs to be available to answer questions and provide encouragement. This could be a doctor or a nurse or a pharmacist. Again, this is an example of the partnership needed to achieve good asthma control.
It would also be useful at a future appointment to discuss the patient’s lifestyle and work with her to reduce her stress. Feeling better would allow her to take simple steps such as taking exercise. It would also be helpful if all members of her family understood how to help her. Even young children can do this.
From personal experience some people know how beneficial it is to feel they are in a partnership with their local practice and pharmacy. Being proactive produces dividends in asthma control.
What are the clinical challenges for the healthcare professional in providing self-management support?
Due to the variable nature of asthma, a long-standing history may mean that the frequency and severity of symptoms, as well as what triggers them, may have changed over time. 32 Exacerbations requiring oral steroids, interrupting periods of ‘stability’, indicate the need for re-assessment of the patient’s clinical as well as educational needs. The patient’s perception of stability may be at odds with the clinical definition 1 , 33 —a check on the number of short-acting bronchodilator inhalers the patient has used over a specific period of time is a good indication of control. 34 Assessment of asthma control should be carried out using objective tools such as the Asthma Control Test or the Royal College of Physicians three questions. 35 , 36 However, it is important to remember that these assessment tools are not an end in themselves but should be a springboard for further discussion on the nature and pattern of symptoms. Balancing work with family can often make it difficult to find the time to attend a review of asthma particularly when the patient feels well. The practice should consider utilising other means of communication to maintain contact with patients, encouraging them to come in when a problem is highlighted. 37 , 38 Asthma guidelines advocate a structured approach to ensure the patient is reviewed regularly and recommend a detailed assessment to enable development of an appropriate patient-centred (self)management strategy. 1 – 4
Although self-management plans have been shown to be successful for reducing the impact of asthma, 21 , 39 the complexity of managing such a fluctuating disease on a day-to-day basis is challenging. During an asthma review, there is an opportunity to work with the patient to try to identify what triggers their symptoms and any actions that may help improve or maintain control. 38 An integral part of personalised self-management education is the written PAAP, which gives the patient the knowledge to respond to the changes in symptoms and ensures they maintain control of their asthma within predetermined parameters. 9 , 40 The PAAP should include details on how to monitor asthma, recognise symptoms, how to alter medication and what to do if the symptoms do not improve. The plan should include details on the treatment to be taken when asthma is well controlled, and how to adjust it when the symptoms are mild, moderate or severe. These action plans need to be developed between the doctor, nurse or asthma educator and the patient during the review and should be frequently reviewed and updated in partnership (see Box 1). Patient preference as well as clinical features such as whether she under- or over-perceives her symptoms should be taken into account when deciding whether the action plan is peak flow or symptom-driven. Our patient has a lot to gain from having an action plan. She has poorly controlled asthma and her lifestyle means that she will probably see different doctors (depending who is available) when she needs help. Being empowered to self-manage could make a big difference to her asthma control and the impact it has on her life.
The practice should have protocols in place, underpinned by specific training to support asthma self-management. As well as ensuring that healthcare professionals have appropriate skills, this should include training for reception staff so that they know what action to take if a patient telephones to say they are having an asthma attack.
However, focusing solely on symptom management strategies (actions) to follow in the presence of deteriorating symptoms fails to incorporate the patients’ wider views of asthma, its management within the context of her/his life, and their personal asthma management strategies. 41 This may result in a failure to use plans to maximise their health potential. 21 , 42 A self-management strategy leading to improved outcomes requires a high level of patient self-efficacy, 43 a meaningful partnership between the patient and the supporting health professional, 42 , 44 and a focused self-management discussion. 14
Central to both the effectiveness and personalisation of action plans, 43 , 45 in particular the likelihood that the plan will lead to changes in patients’ day-to-day self-management behaviours, 45 is the identification of goals. Goals are more likely to be achieved when they are specific, important to patients, collaboratively set and there is a belief that these can be achieved. Success depends on motivation 44 , 46 to engage in a specific behaviour to achieve a valued outcome (goal) and the ability to translate the behavioural intention into action. 47 Action and coping planning increases the likelihood that patient behaviour will actually change. 44 , 46 , 47 Our patient has a goal: she wants to avoid having her work disrupted by her asthma. Her personalised action plan needs to explicitly focus on achieving that goal.
As providers of self-management support, health professionals must work with patients to identify goals (valued outcomes) that are important to patients, that may be achievable and with which they can engage. The identification of specific, personalised goals and associated feasible behaviours is a prerequisite for the creation of asthma self-management plans. Divergent perceptions of asthma and how to manage it, and a mismatch between what patients want/need from these plans and what is provided by professionals are barriers to success. 41 , 42
What are the challenges for the healthcare organisation in providing self-management support?
A number of studies have demonstrated the challenges for primary care physicians in providing ongoing support for people with asthma. 31 , 48 , 49 In some countries, nurses and other allied health professionals have been trained as asthma educators and monitor people with stable asthma. These resources are not always available. In addition, some primary care services are delivered in constrained systems where only a few minutes are available to the practitioner in a consultation, or where only a limited range of asthma medicines are available or affordable. 50
There is recognition that the delivery of quality care depends on the competence of the doctor (and supporting health professionals), the relationship between the care providers and care recipients, and the quality of the environment in which care is delivered. 51 This includes societal expectations, health literacy and financial drivers.
In 2001, the Australian Government adopted a programme developed by the General Practitioner Asthma Group of the National Asthma Council Australia that provided a structured approach to the implementation of asthma management guidelines in a primary care setting. 52 Patients with moderate-to-severe asthma were eligible to participate. The 3+ visit plan required confirmation of asthma diagnosis, spirometry if appropriate, assessment of trigger factors, consideration of medication and patient self-management education including provision of a written PAAP. These elements, including regular medical review, were delivered over three visits. Evaluation demonstrated that the programme was beneficial but that it was difficult to complete the third visit in the programme. 53 – 55 Accordingly, the programme, renamed the Asthma Cycle of Care, was modified to incorporate two visits. 56 Financial incentives are provided to practices for each patient who receives this service each year.
Concurrently, other programmes were implemented which support practice-based care. Since 2002, the National Asthma Council has provided best-practice asthma and respiratory management education to health professionals, 57 and this programme will be continuing to 2017. The general practitioner and allied health professional trainers travel the country to provide asthma and COPD updates to groups of doctors, nurses and community pharmacists. A number of online modules are also provided. The PACE (Physician Asthma Care Education) programme developed by Noreen Clark has also been adapted to the Australian healthcare system. 58 In addition, a pharmacy-based intervention has been trialled and implemented. 59
To support these programmes, the National Asthma Council ( www.nationalasthma.org.au ) has developed resources for use in practices. A strong emphasis has been on the availability of a range of PAAPs (including plans for using adjustable maintenance dosing with ICS/LABA combination inhalers), plans for indigenous Australians, paediatric plans and plans translated into nine languages. PAAPs embedded in practice computer systems are readily available in consultations, and there are easily accessible online paediatric PAAPs ( http://digitalmedia.sahealth.sa.gov.au/public/asthma/ ). A software package, developed in the UK, can be downloaded and used to generate a pictorial PAAP within the consultation. 60
One of the strongest drivers towards the provision of written asthma action plans in Australia has been the Asthma Friendly Schools programme. 61 , 62 Established with Australian Government funding and the co-operation of Education Departments of each state, the Asthma Friendly Schools programme engages schools to address and satisfy a set of criteria that establishes an asthma-friendly environment. As part of accreditation, the school requires that each child with asthma should have a written PAAP prepared by their doctor to assist (trained) staff in managing a child with asthma at school.
The case study continues...
The initial presentation some weeks ago was during an exacerbation of asthma, which may not be the best time to educate a patient. It is, however, a splendid time to build on their motivation to feel better. She agreed to return after her asthma had settled to look more closely at her asthma control, and an appointment was made for a routine review.
At this follow-up consultation, the patient’s diagnosis was reviewed and confirmed and her trigger factors discussed. For this lady, respiratory tract infections are the usual trigger but allergic factors during times of high pollen count may also be relevant. Assessment of her nasal airway suggested that she would benefit from better control of allergic rhinitis. Other factors were discussed, as many patients are unaware that changes in air temperature, exercise and pets can also trigger asthma exacerbations. In addition, use of the Asthma Control Test was useful as an objective assessment of control as well as helping her realise what her life could be like! Many people with long-term asthma live their life within the constraints of their illness, accepting that is all that they can do.
After assessing the level of asthma control, a discussion about management options—trigger avoidance, exercise and medicines—led to the development of a written PAAP. Asthma can affect the whole family, and ways were explored that could help her family understand why it is important that she finds time in the busy domestic schedules to take her regular medication. Family and friends can also help by understanding what triggers her asthma so that they can avoid exposing her to perfumes, pollens or pets that risk triggering her symptoms. Information from the national patient organisation was provided to reinforce the messages.
The patient agreed to return in a couple of weeks, and a recall reminder was set up. At the second consultation, the level of control since the last visit will be explored including repeat spirometry, if appropriate. Further education about the pathophysiology of asthma and how to recognise early warning signs of loss of control can be given. Device use will be reassessed and the PAAP reviewed. Our patient’s goal is to avoid disruption to her work and her PAAP will focus on achieving that goal. Finally, agreement will be reached with the patient about future routine reviews, which, now that she has a written PAAP, could be scheduled by telephone if all is well, or face-to-face if a change in her clinical condition necessitates a more comprehensive review.
Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention, 2012. Available from: http://www.ginasthma.org (accessed July 2013).
British Thoracic Society/Scottish Intercollegiate Guideline Network British Guideline on the Management of Asthma. Thorax 2008; 63 (Suppl 4 iv1–121, updated version available from: http://www.sign.ac.uk (accessed January 2014).
Article Google Scholar
National Asthma Council Australia. Australian Asthma Handbook. Available from: http://www.nationalasthma.org.au/handbook (accessed May 2014).
National Asthma Education and Prevention Program (NAEPP) Coordinating Committee. Expert Panel Report 3 (EPR3): Guidelines for the Diagnosis and Management of Asthma. Available from: https://www.nhlbi.nih.gov/guidelines/asthma/asthgdln.htm (accessed May 2014).
Taylor SJC, Pinnock H, Epiphaniou E, Pearce G, Parke H . A rapid synthesis of the evidence on interventions supporting self-management for people with long-term conditions. (PRISMS Practical Systematic Review of Self-Management Support for long-term conditions). Health Serv Deliv Res (in press).
Gibson PG, Powell H, Wilson A, Abramson MJ, Haywood P, Bauman A et al. Self-management education and regular practitioner review for adults with asthma. Cochrane Database Syst Rev 2002: (Issue 3) Art No. CD001117.
Tapp S, Lasserson TJ, Rowe BH . Education interventions for adults who attend the emergency room for acute asthma. Cochrane Database Syst Rev 2007: (Issue 3) Art No. CD003000.
Powell H, Gibson PG . Options for self-management education for adults with asthma. Cochrane Database Syst Rev 2002: (Issue 3) Art No: CD004107.
Toelle B, Ram FSF . Written individualised management plans for asthma in children and adults. Cochrane Database Syst Rev 2004: (Issue 1) Art No. CD002171.
Lefevre F, Piper M, Weiss K, Mark D, Clark N, Aronson N . Do written action plans improve patient outcomes in asthma? An evidence-based analysis. J Fam Pract 2002; 51 : 842–848.
PubMed Google Scholar
Boyd M, Lasserson TJ, McKean MC, Gibson PG, Ducharme FM, Haby M . Interventions for educating children who are at risk of asthma-related emergency department attendance. Cochrane Database Syst Rev 2009: (Issue 2) Art No.CD001290.
Bravata DM, Gienger AL, Holty JE, Sundaram V, Khazeni N, Wise PH et al. Quality improvement strategies for children with asthma: a systematic review. Arch Pediatr Adolesc Med 2009; 163 : 572–581.
Bower P, Murray E, Kennedy A, Newman S, Richardson G, Rogers A . Self-management support interventions to reduce health care utilisation without compromising outcomes: a rapid synthesis of the evidence. Available from: http://www.nets.nihr.ac.uk/projects/hsdr/11101406 (accessed April 2014).
Gibson PG, Powell H . Written action plans for asthma: an evidence-based review of the key components. Thorax 2004; 59 : 94–99.
Article CAS Google Scholar
Bhogal SK, Zemek RL, Ducharme F . Written action plans for asthma in children. Cochrane Database Syst Rev 2006: (Issue 3) Art No. CD005306.
Zemek RL, Bhogal SK, Ducharme FM . Systematic review of randomized controlled trials examining written action plans in children: what is the plan?. Arch Pediatr Adolesc Med 2008; 162 : 157–163.
Pinnock H, Slack R, Pagliari C, Price D, Sheikh A . Understanding the potential role of mobile phone based monitoring on asthma self-management: qualitative study. Clin Exp Allergy 2007; 37 : 794–802.
de Jongh T, Gurol-Urganci I, Vodopivec-Jamsek V, Car J, Atun R . Mobile phone messaging for facilitating self-management of long-term illnesses. Cochrane Database Syst Rev 2012: (Issue 12) Art No. CD007459.
Huckvale K, Car M, Morrison C, Car J . Apps for asthma self-management: a systematic assessment of content and tools. BMC Med 2012; 10 : 144.
Allergic Rhinitis and its Impact on Asthma. Management of Allergic Rhinitis and its Impact on Asthma: Pocket Guide. ARIA 2008. Available from: http://www.whiar.org (accessed May 2014).
Ring N, Jepson R, Hoskins G, Wilson C, Pinnock H, Sheikh A et al. Understanding what helps or hinders asthma action plan use: a systematic review and synthesis of the qualitative literature. Patient Educ Couns 2011; 85 : e131–e143.
Moullec G, Gour-Provencal G, Bacon SL, Campbell TS, Lavoie KL . Efficacy of interventions to improve adherence to inhaled corticosteroids in adult asthmatics: Impact of using components of the chronic care model. Respir Med 2012; 106 : 1211–1225.
Pinnock H, Bawden R, Proctor S, Wolfe S, Scullion J, Price D et al. Accessibility, acceptability and effectiveness of telephone reviews for asthma in primary care: randomised controlled trial. BMJ 2003; 326 : 477–479.
Pinnock H, Adlem L, Gaskin S, Harris J, Snellgrove C, Sheikh A . Accessibility, clinical effectiveness and practice costs of providing a telephone option for routine asthma reviews: phase IV controlled implementation study. Br J Gen Pract 2007; 57 : 714–722.
PubMed PubMed Central Google Scholar
Kielmann T, Huby G, Powell A, Sheikh A, Price D, Williams S et al. From support to boundary: a qualitative study of the border between self care and professional care. Patient Educ Couns 2010; 79 : 55–61.
Asthma UK . Compare your care report. Asthma UK, 2013. Available from: http://www.asthma.org.uk (accessed January 2014).
Stallberg B, Lisspers K, Hasselgren M, Janson C, Johansson G, Svardsudd K . Asthma control in primary care in Sweden: a comparison between 2001 and 2005. Prim Care Respir J 2009; 18 : 279–286.
Reddel H, Peters M, Everett P, Flood P, Sawyer S . Ownership of written asthma action plans in a large Australian survey. Eur Respir J 2013; 42 . Abstract 2011.
Wiener-Ogilvie S, Pinnock H, Huby G, Sheikh A, Partridge MR, Gillies J . Do practices comply with key recommendations of the British Asthma Guideline? If not, why not? Prim Care Respir J 2007; 16 : 369–377.
Kennedy A, Rogers A, Bower P . Support for self care for patients with chronic disease. BMJ 2007; 335 : 968–970.
Ring N, Malcolm C, Wyke S, Macgillivray S, Dixon D, Hoskins G et al. Promoting the Use of Personal Asthma Action Plans: A Systematic Review. Prim Care Respir J 2007; 16 : 271–283.
Taylor DR, Bateman ED, Boulet LP, Boushey HA, Busse WW, Casale TB et al. A new perspective on concepts of asthma severity and control. Eur Respir J 2008; 32 : 545–554.
Horne R . Compliance, adherence, and concordance: implications for asthma treatment. Chest 2006; 130 (suppl): 65S–72S.
Reddel HK, Taylor DR, Bateman ED, Boulet L-P, Boushey HA, Busse WW et al. An official American Thoracic Society/European Respiratory Society statement: asthma control and exacerbations standardizing endpoints for clinical asthma trials and clinical practice. Am J Respir Crit Care Med 2009; 180 : 59–99.
Thomas M, Kay S, Pike J, Rosenzweig JR, Hillyer EV, Price D . The Asthma Control Test (ACT) as a predictor of GINA guideline-defined asthma control: analysis of a multinational cross-sectional survey. Prim Care Respir J 2009; 18 : 41–49.
Hoskins G, Williams B, Jackson C, Norman P, Donnan P . Assessing asthma control in UK primary care: use of routinely collected prospective observational consultation data to determine appropriateness of a variety of control assessment models. BMC Fam Pract 2011; 12 : 105.
Pinnock H, Fletcher M, Holmes S, Keeley D, Leyshon J, Price D et al. Setting the standard for routine asthma consultations: a discussion of the aims, process and outcomes of reviewing people with asthma in primary care. Prim Care Respir J 2010; 19 : 75–83.
McKinstry B, Hammersley V, Burton C, Pinnock H, Elton RA, Dowell J et al. The quality, safety and content of telephone and face-to-face consultations: a comparative study. Qual Saf Health Care 2010; 19 : 298–303.
Gordon C, Galloway T . Review of Findings on Chronic Disease Self-Management Program (CDSMP) Outcomes: Physical, Emotional & Health-Related Quality of Life, Healthcare Utilization and Costs . Centers for Disease Control and Prevention and National Council on Aging: Atlanta, GA, USA, 2008.
Beasley R, Crane J . Reducing asthma mortality with the self-management plan system of care. Am J Respir Crit Care Med 2001; 163 : 3–4.
Ring N, Jepson R, Pinnock H, Wilson C, Hoskins G, Sheikh A et al. Encouraging the promotion and use of asthma action plans: a cross study synthesis of qualitative and quantitative evidence. Trials 2012; 13 : 21.
Jones A, Pill R, Adams S . Qualitative study of views of health professionals and patients on guided self-management plans for asthma. BMJ 2000; 321 : 1507–1510.
Bandura A . Self-efficacy: toward a unifying theory of behavioural change. Psychol Rev 1977; 84 : 191–215.
Gollwitzer PM, Sheeran P . Implementation intentions and goal achievement: a meta-analysis of effects and processes. Adv Exp Soc Psychol 2006; 38 : 69–119.
Google Scholar
Hardeman W, Johnston M, Johnston DW, Bonetti D, Wareham NJ, Kinmonth AL . Application of the theory of planned behaviour change interventions: a systematic review. Psychol Health 2002; 17 : 123–158.
Schwarzer R . Modeling health behavior change: how to predict and modify the adoption and maintenance of health behaviors. Appl Psychol 2008; 57 : 1–29.
Sniehotta F . Towards a theory of intentional behaviour change: plans, planning, and self-regulation. Br J Health Psychol 2009; 14 : 261–273.
Okelo SO, Butz AM, Sharma R, Diette GB, Pitts SI, King TM et al. Interventions to modify health care provider adherence to asthma guidelines: a systematic review. Pediatrics 2013; 132 : 517–534.
Grol R, Grimshaw RJ . From best evidence to best practice: effective implementation of change in patients care. Lancet 2003; 362 : 1225–1230.
Jusef L, Hsieh C-T, Abad L, Chaiyote W, Chin WS, Choi Y-J et al. Primary care challenges in treating paediatric asthma in the Asia-Pacific region. Prim Care Respir J 2013; 22 : 360–362.
Donabedian A . Evaluating the quality of medical care. Milbank Q 2005; 83 : 691–729.
Fardy HJ . Moving towards organized care of chronic disease. The 3+ visit plan. Aust Fam Physician 2001; 30 : 121–125.
CAS PubMed Google Scholar
Glasgow NJ, Ponsonby AL, Yates R, Beilby J, Dugdale P . Proactive asthma care in childhood: general practice based randomised controlled trial. BMJ 2003; 327 : 659.
Douglass JA, Goemann DP, Abramson MJ . Asthma 3+ visit plan: a qualitative evaluation. Intern Med J 2005; 35 : 457–462.
Beilby J, Holton C . Chronic disease management in Australia; evidence and policy mismatch, with asthma as an example. Chronic Illn 2005; 1 : 73–80.
The Department of Health. Asthma Cycle of Care. Accessed on 14 May 2014 at http://www.health.gov.au/internet/main/publishing.nsf/Content/asthma-cycle .
National Asthma Council Australia. Asthma and Respiratory Education Program. Accessed on 14 May 2014 at http://www.nationalasthma.org.au/health-professionals/education-training/asthma-respiratory-education-program .
Patel MR, Shah S, Cabana MD, Sawyer SM, Toelle B, Mellis C et al. Translation of an evidence-based asthma intervention: Physician Asthma Care Education (PACE) in the United States and Australia. Prim Care Respir J 2013; 22 : 29–34.
Armour C, Bosnic-Anticevich S, Brilliant M, Burton D, Emmerton L, Krass I et al. Pharmacy Asthma Care Program (PACP) improves outcomes for patients in the community. Thorax 2007; 62 : 496–502.
Roberts NJ, Mohamed Z, Wong PS, Johnson M, Loh LC, Partridge MR . The development and comprehensibility of a pictorial asthma action plan. Patient Educ Couns 2009; 74 : 12–18.
Henry RL, Gibson PG, Vimpani GV, Francis JL, Hazell J . Randomised controlled trial of a teacher-led asthma education program. Pediatr Pulmonol 2004; 38 : 434–442.
National Asthma Council Australia. Asthma Friendly Schools program. Accessed on 14 May 2014 at http://www.asthmaaustralia.org.au/Asthma-Friendly-Schools.aspx .
Download references
Author information
Authors and affiliations.
Asthma UK Centre for Applied Research, Centre for Population Health Sciences, The University of Edinburgh, Edinburgh, UK,
Hilary Pinnock & Elisabeth Ehrlich
NMAHP-RU, University of Stirling, Stirling, UK,
Gaylor Hoskins
Discipline of General Practice, University of Sydney, Sydney, NSW, Australia
Ron Tomlins
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Hilary Pinnock .
Ethics declarations
Competing interests.
The authors declare no conflict of interest.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
Reprints and permissions
About this article
Cite this article.
Pinnock, H., Ehrlich, E., Hoskins, G. et al. A woman with asthma: a whole systems approach to supporting self-management. npj Prim Care Resp Med 24 , 14063 (2014). https://doi.org/10.1038/npjpcrm.2014.63
Download citation
Received : 23 June 2014
Revised : 15 July 2014
Accepted : 15 July 2014
Published : 16 October 2014
DOI : https://doi.org/10.1038/npjpcrm.2014.63
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies
Sign up for the Nature Briefing newsletter — what matters in science, free to your inbox daily.
- Case report
- Open access
- Published: 21 February 2018
Pediatric severe asthma: a case series report and perspectives on anti-IgE treatment
- Virginia Mirra 1 ,
- Silvia Montella 1 &
- Francesca Santamaria 1
BMC Pediatrics volume 18 , Article number: 73 ( 2018 ) Cite this article
11k Accesses
11 Citations
12 Altmetric
Metrics details
The primary goal of asthma management is to achieve disease control for reducing the risk of future exacerbations and progressive loss of lung function. Asthma not responding to treatment may result in significant morbidity. In many children with uncontrolled symptoms, the diagnosis of asthma may be wrong or adherence to treatment may be poor. It is then crucial to distinguish these cases from the truly “severe therapy-resistant” asthmatics by a proper filtering process. Herein we report on four cases diagnosed as difficult asthma, detail the workup that resulted in the ultimate diagnosis, and provide the process that led to the prescription of omalizumab.
Case presentation
All children had been initially referred because of asthma not responding to long-term treatment with high-dose inhaled steroids, long-acting β 2 -agonists and leukotriene receptor antagonists. Definitive diagnosis was severe asthma. Three out four patients were treated with omalizumab, which improved asthma control and patients’ quality of life. We reviewed the current literature on the diagnostic approach to the disease and on the comorbidities associated with difficult asthma and presented the perspectives on omalizumab treatment in children and adolescents. Based on the evidence from the literature review, we also proposed an algorithm for the diagnosis of pediatric difficult-to-treat and severe asthma.

Conclusions
The management of asthma is becoming much more patient-specific, as more and more is learned about the biology behind the development and progression of asthma. The addition of omalizumab, the first targeted biological treatment approved for asthma, has led to renewed optimism in the management of children and adolescents with atopic severe asthma.
Peer Review reports
Children with poor asthma control have an increased risk of severe exacerbations and progressive loss of lung function, which results in the relevant use of health resources and impaired quality of life (QoL) [ 1 ]. Therefore, the primary goal of asthma management at all ages is to achieve disease control [ 2 , 3 , 4 ].
According to recent international guidelines, patients with uncontrolled asthma require a prolonged maintenance treatment with high-dose inhaled corticosteroids (ICS) in association with a long-acting β 2 -agonist (LABA) plus oral leukotriene receptor antagonist (LTRA) (Table 1 ) [ 5 ].
Nevertheless, in the presence of persistent lack of control, reversible factors such as adherence to treatment or inhalation technique should be first checked for, and diseases that can masquerade as asthma should be promptly excluded. Finally, additional strategies, in particular anti-immunoglobulin E (anti-IgE) treatment (omalizumab), are suggested for patients with moderate or severe allergic asthma that remains uncontrolled in Step 4 [ 5 ].
Herein, we reviewed the demographics, clinical presentation and treatment of four patients with uncontrolled severe asthma from our institution in order to explain why we decided to prescribe omalizumab. We also provided a review of the current literature that focuses on recent advances in the diagnosis of pediatric difficult asthma and the associated comorbidities, and summarizes the perspectives on anti-IgE treatment in children and adolescents.
Case presentations
Table 2 summarizes the clinical characteristics and the triggers/comorbidities of the cases at referral to our Institution. Unfortunately, data on psychological factors, sleep apnea, and hyperventilation syndrome were not available in any case. Clinical, lung function and airway inflammation findings at baseline and after 12 months of follow-up are reported in Table 3 . In the description of our cases, we used the terminology recommended by the ERS/ATS guidelines on severe asthma [ 6 ].
A full-term male had severe preschool wheezing and, since age 3, recurrent, severe asthma exacerbations with frequent hospital admissions. At age 11, severe asthma was diagnosed. Sensitization to multiple inhalant allergens (i.e., house dust mites, dog dander, Graminaceae pollen mix, and Parietaria judaica ) and high serum IgE levels (1548 KU/l) were found. Body mass index (BMI) was within normal range. Combined treatment with increasing doses of ICS (fluticasone, up to 1000 μg/day) in association with LABA (salmeterol, 100 μg/day) plus LTRA (montelukast, 5 mg/day) has been administered over 2 years. Nevertheless, persistent symptoms and monthly hospital admissions due to asthma exacerbations despite correct inhaler technique and good adherence were reported. Parents refused to perform any test to exclude gastroesophageal reflux (GER) as comorbidity [ 6 ]. However, an ex-juvantibus 2-month-course with omeprazole was added to asthma treatment [ 7 ], but poor control persisted. Anterior rhinoscopy revealed rhinosinusitis that was treated with nasal steroids for six months [ 8 ], but asthma symptoms were unmodified. Treatment with omalizumab was added at age 12. Reduced hospital admissions for asthma exacerbations, no further need for systemic steroids, and improved QoL score (from 2.0 up to 6.7 out of a maximum of 7 points) were documented over the following months. Unfortunately, after one year of treatment, adherence to omalizumab decreased because of family complaints, and eventually parents withdrew their informed consent and discontinued omalizumab. Currently, by age 17, treatment includes inhaled salmeterol/fluticasone (100 μg/500 μg∙day -1 , respectively) plus oral montelukast (10 mg/day). Satisfactory symptom control is reported, with no asthma exacerbations.
A full-term male, who had a recurrent severe preschool wheezing, at 6 years of age developed exercise-induced asthma. At age 10, severe asthma was diagnosed. High serum IgE levels (1300 KU/l) and skin prick tests positive to house dust mites were found. Despite a 3-year treatment with progressively increasing doses of inhaled fluticasone (up to 1000 μg/day) combined with salmeterol (100 μg/day) and oral montelukast (5 mg/day), monthly hospital admissions with systemic steroids use were reported. At age 13, a 24-h esophageal impedance/pH study demonstrated the presence of acid and non-acid GER [ 7 ]. Esomeprazole was added to asthma medications, but with an incomplete clinical benefit for respiratory symptoms. Esomeprazole was withdrawn after 3 months, and parents refused to re-test for GER. As respiratory symptoms persisted uncontrolled despite treatment, severe asthma was definitively diagnosed [ 6 ]. BMI was within the normal range and anterior rhinoscopy excluded rhinosinusitis. Inhaler technique and adherence were good; thus we considered the anti-IgE treatment option [ 9 ]. Subcutaneous omalizumab was started, with fast improvement of both symptoms and QoL score (from 3.9 up to 6.5). Seventeen months later, the dose of ICS had been gradually tapered and oral montelukast definitely discontinued. Currently, at age 14, treatment includes the combined administration of bimonthly subcutaneous omalizumab and of daily inhaled salmeterol/fluticasone (50 μg/100 μg∙day - 1 , respectively). Asthma control is satisfactory and no side effects are reported. Omalizumab has been continuously administered for 2.6 years and is still ongoing.
A full-term male had severe preschool wheezing and, since age 3, recurrent, severe asthma exacerbations with acute respiratory failure that frequently required intensive care unit (ICU) admission. At age 6, sensitization to multiple perennial inhalant (i.e., house dust mites, dog and cat danders, Alternaria alternata , Graminaceae pollen mix, Artemisia vulgaris , Parietaria judaica , and Olea europaea pollen) and food allergens (i.e., egg, milk, and peanut) was diagnosed. Serum IgE levels were 2219 KU/l. Weight and height were appropriate for age and sex. The patient has been treated over 3 years with a combined scheme of high-dose inhaled fluticasone (up to 1000 μg/day) plus salmeterol (100 μg/day) and oral montelukast (5 mg/day), with correct inhaler technique and good adherence. Despite this, monthly hospital admissions with systemic steroids use were recorded. Rhinosinusitis and GER were excluded on the basis of appropriate testing; thus treatment with omalizumab was started when the patient was 9 years old. At age 11, adherence to treatment is satisfactory, with no side effects. More importantly, reduced hospital admissions for asthma exacerbations, no further need for systemic steroids, and improved QoL score (from 6.4 to 6.8) were reported. Finally, progressive step-down of anti-asthma treatment was started, and at present (by 11.5 years) inhaled fluticasone (200 μg/day) plus bimonthly subcutaneous omalizumab provide good control of symptoms. Omalizumab has been continuously administered for 2.6 years and is still ongoing.
A full-term male had severe preschool wheezing and, since age 4, recurrent, severe asthma exacerbations with frequent hospital admissions. At age 8, multiple perennial inhalants and food sensitization (i.e., house dust mites, dog dander, Graminaceae pollen mix, Olea europaea pollen, tomatoes, beans, shrimps, and peas) and high serum IgE levels (1166 KU/l) were found. The patient has been treated over 5 years with inhaled fluticasone (up to 1000 μg/day) in association with salmeterol (100 μg/day) and oral montelukast (5 mg/day). Despite this, monthly hospital admissions with systemic steroids need were recorded. After checking the inhaler technique and adherence to treatment, comorbidities including obesity, rhinosinusitis and GER were excluded. Omalizumab was proposed, but parents refused it. By 13.6 years, despite a treatment including the association of inhaled salmeterol/fluticasone (100 μg/1000 μg∙day − 1 , respectively) plus oral montelukast (10 mg/day), monthly exacerbations requiring systemic steroids are reported.
Discussion and conclusions
Most children and adolescents with asthma respond well to inhaled short-acting beta 2 -agonists (SABA) on demand if symptoms are intermittent, or to low dose controller drugs plus as-needed SABA if the risk of exacerbations increases [ 1 ]. Nevertheless, a proportion of patients is referred to specialists because this strategy is not working and asthma is persistently uncontrolled [ 4 ]. For these children, assessment is primarily aimed at investigating the reasons for poor control. Indeed, when the child is initially referred, before the label of “severe, therapy-resistant asthma” (i.e., not responding to treatment even when factors as exposure to allergens and tobacco smoke have been considered) is assigned, three main categories need to be identified: 1) “not asthma at all”, in which response to treatment is suboptimal because the diagnosis is wrong; 2) “asthma plus ”, when asthma is mild but exacerbated by one or more comorbidities; and 3) “difficult-to-treat asthma”, when asthma is uncontrolled because of potentially reversible factors [ 10 ].
The reported cases highlight some aspects of the disease process that may expand the diagnosis and improve patients’ care. At our institution, the severe asthma program includes a multidisciplinary approach with consultations by gastroenterologists as well as ear, nose and throat experts. Recently, sleep medicine experts joined this multidisciplinary team; thus, unfortunately, sleep-disordered breathing (SDB) could not be excluded at the time of our patients’ assessment. Inhalation technique is periodically evaluated by nurses or doctors in each patient. Unfortunately, in Italy an individual prescription database is not available and thus we cannot assess patients’ use of medication. In two cases, the filtering process eventually identified GER and rhinosinusitis, but poor control of asthma persisted even after comorbidities were treated. In all subjects, inhaler skills, treatment adherence, and environmental exposure to indoor/outdoor allergens as well as to second- and third-hand smoke were excluded as cause of lack of control. Eventually, three out of four patients started anti-IgE treatment; asthma control was obtained and maintenance drugs were progressively reduced. In the case that refused omalizumab therapy, pulmonary function, clinical features and controller treatment including high-dose ICS were unchanged.
Previous studies have highlighted an association between increasing asthma severity in children and reduced QoL [ 11 , 12 , 13 ]. Uncontrolled asthma symptoms not only affect children physically, but can impair them socially, emotionally, and educationally [ 13 ]. In line with previous observations, 3 out 4 of our cases had poor QoL, assessed by a standardized questionnaire [ 14 ]. It is well known that improving QoL in difficult asthma is not an easy task, despite a variety of treatments aimed at achieving control [ 12 ], and much more remains to be done to address the problem. Nevertheless, 2 of our 3 cases showed a remarkable improvement of QoL after one year of treatment with omalizumab.
Reduction in forced expiratory volume in the first second (FEV 1 ) is often used to define childhood asthma severity in treatment guidelines and clinical studies [ 5 , 11 , 15 ]. Nevertheless, children with severe asthma often have a normal FEV 1 that does not improve after bronchodilators, indicating that spirometry may be a poor predictor of asthma severity in childhood [ 6 , 16 , 17 ]. Actually, children with a normal FEV 1 , both before and after β 2 -agonist, may show a bronchodilator response in terms of forced expiratory flow between 25% and 75% (FEF 25–75 ) [ 18 ]. However, the utility of FEF 25–75 in the assessment or treatment of severe asthma is currently unknown. Interestingly, all the reported cases showed normal or slightly reduced values of FEV 1 but severe impairment of FEF 25–75 . Two cases showed a bronchodilator response in terms of FEV 1 (subjects 3 and 4), while 3 patients had a significant increase of FEF 25–75 (cases 1, 3 and 4). Unfortunately, we could not provide the results of bronchodilator response during or after the treatment with omalizumab in any case.
Available literature on the diagnostic approach to difficult asthma in children offers a number of reviews which basically summarize the steps needed to fill the gap between a generic diagnosis of “difficult asthma” and more specific labels (i.e., “severe” asthma, “difficult-to-treat” asthma, or even different diagnoses) [ 3 , 5 , 6 , 8 , 10 , 19 , 20 , 21 ]. So far, few original articles and case reports have been published, probably due to the peculiarity of the issue, which makes retrospective discussion of cases easier than the design of a prospective clinical study [ 4 , 22 , 23 , 24 , 25 , 26 ]. Available knowledge mainly derives from the experience of specialized centers.
The evaluation of a child referred for uncontrolled asthma should start with a careful history focused on typical respiratory symptoms and on the definition of possible triggers. In the “severe asthma” process, it is crucial for clinicians to maintain a high degree of skepticism about the ultimate diagnosis, particularly in the presence of relevant discrepancies between history, physical features and lung function, as many conditions may be misdiagnosed as asthma. In order to simplify this process, herein we propose an algorithm for the diagnosis of difficult-to-treat and severe asthma (Fig. 1 ). Confirmation of the diagnosis through a detailed clinical and laboratory re-evaluation is important because in 12–50% of cases assumed to have severe asthma this might not be the correct diagnosis [ 10 ]. Several documents have indicated the main steps of the process that should be followed in children with uncontrolled asthma [ 3 , 8 , 10 ]. The translation of these procedures into real life practice may deeply change from one subject to another due to the variability of individual patients’ history and clinical features, which will often lead the diagnostic investigations towards the most likely reason for uncontrolled asthma. For children with apparently severe asthma, the first step is to confirm the diagnosis and, before proceeding to broader investigations, to verify that the poor control is not simply determined by poor adherence to treatment, inadequate inhaler skills and/or environmental exposure to triggers. A nurse-led assessment, including a home visit, despite not being applicable in all settings, may be useful for identifying potentially modifiable factors in uncontrolled pediatric asthma [ 27 ].
A practical algorithm for the diagnosis of difficult-to-treat and severe asthma. ICS, inhaled corticosteroids; OCS, oral corticosteroids
A number of comorbidities have been increasingly recognized as factors that may impact asthma clinical expression and control in childhood [ 10 , 28 ]. Children with uncontrolled disease should be investigated for GER, rhinosinusitis, dysfunctional breathing and/or vocal cord dysfunction, obstructive sleep apnea, obesity, psychological factors, smoke exposure, hormonal influences, and ongoing drugs [ 3 , 6 , 8 , 20 ]. Indeed, the exact role played by comorbidities in pediatric asthma control is still debated [ 28 ]. The most impressive example is GER. Several pediatric documents recommend assessing for GER because reflux may be a contributing factor to problematic or difficult asthma [ 7 , 29 ]. Nevertheless, GER treatment might not be effective for severe asthma [ 30 , 31 ], as confirmed by current cases 1 and 2. There is an established evidence that chronic rhinosinusitis is associated with more severe asthma in children [ 32 , 33 , 34 ]. Therefore, examination of upper airways and ad hoc treatment if rhinosinusitis is evident are recommended in children with severe asthma [ 3 , 8 , 35 ]. However, intranasal steroids for rhinitis resulted in a small reduction of asthma risk in school-aged children [ 36 ], and actual placebo-controlled studies on the effect of treatment of rhinosinusitis on asthma control in children are lacking [ 10 , 37 ].
Dysfunctional breathing, including hyperventilation and vocal cord dysfunction, is associated with poorer asthma control in children [ 8 , 10 , 38 , 39 ]. Unfortunately, there is scarce literature on the effect of its treatment on the control of severe asthma in children [ 40 ]. SDB ranging from primary snoring to obstructive sleep apnea syndrome is very common in children [ 41 ], and an increased prevalence of SDB together with increasing asthma severity has been reported [ 42 ]. Interestingly, GER may also be worsened by recurrent episodes of upper airway obstruction associated with SDB, and this may further trigger bronchial obstruction. Asthma guidelines recommend the assessment of SDB through nocturnal polysomnography in poorly controlled asthmatics, particularly if they are also obese [ 5 ]. There are no studies examining whether pediatric asthma improves after SDB has been treated, for example, with nasal steroids, adenotonsillectomy, continuous positive airway pressure or weight reduction if the child is also obese [ 43 ]. The parallel increase in obesity and asthma suggests that the two conditions are linked and that they can aggravate each other [ 44 , 45 ], even though the exact mechanisms that underlie this association remain unclear [ 46 ]. Indeed, other coexisting comorbidities such as SDB or GER may play a confounding role in the development of the interactions between obesity and the airways [ 47 , 48 ]. Obesity is associated with increased markers of inflammation in serum and adipose tissue and yet decreased airway inflammation in obese people with asthma [ 49 ]. Several interventions, including behavioral and weight reduction programs or bariatric surgery, may result in improved asthma control, quality of life and lung function in adult obese asthmatics [ 50 ]. Although reports of adolescent bariatric surgery demonstrate a significant body weight decrease, this approach is not widely available and there are no published reports on its effect on pediatric severe asthma control [ 51 ]. Finally, although it is still unclear whether food allergy is causative or shares a common pathway with difficult asthma, it might explain the loss of asthma control at least in some children and thus be considered as a comorbid condition [ 10 , 16 , 52 ].
In conclusion, establishing the impact of comorbidities on asthma control may be cumbersome, and an ex-juvantibus treatment is sometimes necessary to assess their role. Comorbid conditions can also worsen each other, and symptoms arising from some of them may mimic asthma [ 6 ]. Although the ability to improve pediatric severe asthma by treating comorbidities remains unconfirmed, they should be treated appropriately [ 9 ].
The vast majority of asthmatic children exhibit a mild or at most a moderate disease that can be fully controlled with low-to-medium dose ICS associated or not with other controllers [ 5 , 6 ]. However, a subset of asthmatics remains difficult-to-treat [ 5 , 6 ]. With the advent of biologics, these severe steroid-dependent asthmatics have alternative options for treatment, as steroid-related adverse events are common in severe asthma [ 53 ]. Omalizumab, an anti-IgE monoclonal antibody, is the only biologic therapy recommended in children with moderate-to-severe asthma by the recent guidelines [ 5 , 6 ]. In Italy, this treatment is fully covered by the National Health System. Therefore, there is no influence by any funding on treatment decisions. It was approved by the US (Food and Drug Administration) in 2003 and by the European Union (European Medicines Agency) in 2005 as an add-on treatment for patients aged > 12 years with severe persistent allergic asthma and who have a positive skin test or in-vitro reactivity to a perennial aeroallergen, FEV 1 < 80% predicted, frequent daytime symptoms or nighttime awakenings, and multiple documented severe asthma exacerbations despite daily ICS plus a LABA [ 54 , 55 ]. In 2009, it also received approval in Europe for treating patients aged 6–12 years. Figure 2 illustrates current indications for treatment with omalizumab in children and adolescents with severe asthma.
Indications for omalizumab in children and adolescents with severe asthma
IgE antibodies, Th 2 -derived cytokines and eosinophils play a major role in the development of chronic airway inflammation in asthmatic subjects [ 56 ]. Once released from plasma cells, IgE binds principally to the high-affinity IgE receptor (FcεRI) on mast cells, triggering different effector responses, including the release of mediators leading to allergic inflammatory reactions [ 56 ]. The activation of the allergic cascade by IgE, under constant allergen stimulation, leads to the establishment of chronic allergic inflammation in the airways of asthmatic patients, with IgE being a key element of the vicious circle that maintains it. Cytokines produced during the late phase and subsequent chronic inflammation stage have been directly associated with the induction of airway remodelling, indirectly implicating IgE in the process [ 56 ]. At present, omalizumab is the only commercially available recombinant humanized anti-IgE monoclonal antibody that specifically binds serum free IgE at its CH 3 domain, in the proximity of the binding site for FcεRI, thus preventing IgE from interacting with its receptor on mast cells, basophils, antigen-presenting cells and other inflammatory cells [ 57 ]. The rapid reduction of free IgE levels leads to a downregulation of the FcεRI expression on inflammatory cells and an interruption of the allergic cascade, which results in the reduction of peripheral and bronchial tissue eosinophilia and of levels of granulocyte macrophage colony stimulating factor, interleukin (IL)-2, IL-4, IL-5, and IL-13 [ 58 ]. Moreover, basophils have a relevant role in the initiation and progression of allergic inflammation, suggesting that they may represent a viable therapeutic target. Indeed, in children with severe asthma, it has been reported that omalizumab therapy is associated with a significant reduction in circulating basophil numbers, a finding that is concurrent with improved clinical outcomes [ 59 ]. This finding supports a mechanistic link between IgE levels and circulating basophil populations, and may provide new insights into one mechanism by which omalizumab improves asthma symptoms.
Several clinical controlled and real-life studies of adults with severe, inadequately controlled allergic asthma have demonstrated the efficacy and safety of omalizumab in reducing asthma-related symptoms, corticosteroid use, exacerbation rates, and healthcare resource utilization, and in improving QoL and lung function [ 60 , 61 , 62 , 63 ]. Fewer studies have been published in children. In two double-blind, randomized, placebo-controlled trials (RCTs) of children aged 6 to 12 years with moderate-to-severe allergic asthma, treatment with omalizumab reduced the requirement for ICS and protected against disease exacerbations, but there was little change in asthma symptom scores or spirometry [ 9 , 64 ]. These findings were confirmed and extended in older children [ 65 , 66 , 67 ].
The results of the ICATA study, a multicenter RCT of 419 inner-city children, adolescents and young adults with persistent allergic asthma, showed that, compared to placebo, omalizumab reduces the number of days with asthma symptoms and the proportion of participants with at least one exacerbation by approximately 25% and 19%, respectively ( p < 0.001), thus reducing the need for asthmatic symptom controllers [ 68 ]. Another multicenter RCT of inner-city children and adolescents showed that the addition of omalizumab to ongoing guidelines-based care before patients return to school reduces fall asthma exacerbations (odds ratio, 0.48), particularly in subjects with a recent exacerbation [ 69 ]. Moreover, in a real-life study of 104 children and adolescents with severe allergic refractory asthma followed over 1 year, treatment with omalizumab resulted in good asthma control in 67% of the cases ( p < 0.001), while FEV 1 improved by 4.9% ( p = 0.02) and exacerbation rates and healthcare utilisation decreased approximately by 30% ( p < 0.001) [ 70 ]. The same authors also showed that, after two years of treatment, exacerbation rate and healthcare utilisation were further decreased by 83% and 100%, respectively, while level of asthma control, steroid use and lung function remained unchanged [ 71 ].
A systematic review of pediatric RCTs pooled the data of 1381 children and adolescents with moderate-to-severe allergic asthma in order to establish the efficacy of omalizumab as an add-on therapy [ 72 ]. During the stable-steroid phase, omalizumab decreased the number of patients with at least one exacerbation (risk ratio, 0.69; p < 0.001), the mean number of asthma exacerbations per patient (risk ratio, 0.35; p < 0.001), and the asthma symptom score (mean difference, 0.12; p = 0.005) when compared to placebo. During the steroid reduction phase, omalizumab further reduced the number of patients with at least one exacerbation (risk ratio, 0.48; p < 0.001) and the mean number of asthma exacerbations per patient (mean difference, 0.12; p < 0.05).
Given the cost of omalizumab, many authors have argued for the importance of identifying specific asthma populations who will have significant benefit from it [ 68 , 73 , 74 ]. In the ICATA study, baseline predictors of good response to treatment were sensitization and exposure to cockroach allergen, sensitization to house dust mite allergens, a serum IgE level of more than 100 IU per milliliter, a BMI of 25 or more, and a history of at least one unscheduled medical visit in the previous year [ 68 ].
Several studies have assessed the long-term safety of omalizumab in children and adults. A pooled analysis of 67 RCTs conducted over 2 decades on 4254 children and adults treated with omalizumab showed no association between omalizumab treatment and risk of malignancy [ 75 ]. In an RCT evaluating 225 school-aged children, omalizumab was well tolerated, there were no serious adverse events, and the frequency and types of all adverse events were similar to the placebo group [ 9 ]. These results have been further confirmed by a recent systematic review of RCTs that concluded that treatment with omalizumab does not result in increased risk of malignancy or hypersensitivity reactions [ 72 ].
While the rationale for long-term treatment with omalizumab is supported by pharmacokinetic-pharmacodynamic models [ 76 ], the duration of treatment is still under discussion. Results from published studies suggest that omalizumab should be continued for > 1 year [ 77 , 78 ]. In a retrospective study of adults and children with uncontrolled severe asthma treated with omalizumab, the response to treatment was ‘excellent’ in 52.5% of patients, particularly in the subgroup of children aged 6 to 11 years [ 77 ]. After the discontinuation of treatment, loss of asthma control was documented in 69.2% of the patients who had received omalizumab for < 1 year, 59.1% of the subjects treated for 1–2 years, and 46.1% of the cases treated for > 2 years. Time to loss of control was shorter in younger children and longer in patients with an ‘excellent’ response compared with patients with a ‘good’ response. No early loss of control (within 6 months) was observed among patients with > 3.5 years of continuous treatment with omalizumab. Finally, 20% of patients in whom omalizumab was re-prescribed because of loss of control did not respond to the treatment anymore [ 77 ]. Despite these encouraging findings, the impact of omalizumab on the natural history of severe asthma in children deserves to be further investigated by long-term studies that will also define the criteria and timing for discontinuing the treatment.
It is well known that asthma pharmacotherapy is effective in controlling symptoms and bronchial inflammation, but cannot affect the underlying immune response, thus leading to the possibility of symptom reappearance after its discontinuation [ 79 ]. In this scenario, allergen-specific immunotherapy (AIT) has been proposed as the only therapeutic method that can modulate the underlying immune pathophysiology in allergic asthma [ 80 ].
AIT is currently indicated in children and adults with mild-moderate allergic asthma that is completely or partially controlled by pharmacotherapy and with the evidence of a clear relationship between symptoms and exposure to a specific allergen [ 81 , 82 , 83 , 84 ]. However, according to recent guidelines, the efficacy of AIT in asthmatic subjects is limited, and its potential benefits must be weighed against the risk of side effects and the inconvenience and costs of the prolonged therapy [ 5 ]. Moreover, severe or uncontrolled asthma (regardless of its severity) is a major independent risk factor for non-fatal or even fatal adverse reactions, thus representing a contraindication for AIT [ 85 , 86 , 87 ]. Finally, children with severe asthma are often sensitized to multiple allergens, thus making AIT prescription even more complicated [ 88 ].
In subjects with uncontrolled and/or severe allergic asthma, a combination of omalizumab and AIT has been proposed [ 88 ]. Surprisingly, only a few studies have addressed this issue [ 89 , 90 , 91 , 92 ]. However, pre-treatment with omalizumab seems to improve the efficacy and tolerability of subcutaneous AIT in children and adults with severe allergic asthma both during omalizumab treatment and after its discontinuation [ 89 , 91 , 92 ]. Omalizumab has also been successfully used as a supplementary treatment to AIT in order to improve asthma control in children ≥6 years with severe persistent allergic asthma [ 90 ]. Given the scarcity of studies on AIT plus omalizumab in children with severe allergic asthma, further research is warranted to assess risks and benefits of the combined treatment.
Children with severe asthma require a detailed and individualized approach including re-assessment for differential diagnoses, comorbidities and contributory factors, environmental triggers, lung function and inflammation, adherence and response to therapy, and QoL. Treatment of pediatric severe asthma still relies on the maximal optimal use of corticosteroids, bronchodilators and other controllers recommended for moderate-to-severe disease. However, the management of asthma is becoming much more patient-specific, as more and more is learned about the biology behind the development and progression of asthma.
In the current paper, we described the characteristics of four children with severe asthma in whom omalizumab was prescribed. A review of the relevant literature on the topic was also performed. Finally, we provided an algorithm for the diagnosis of difficult-to-treat and severe asthma in children and adolescents, based on the evidence from the literature review. As all algorithms, it is not meant to replace clinical judgment, but it should drive physicians to adopt a systematic approach towards difficult and severe asthma and provide a useful guide to the clinician.
The addition of omalizumab, the first targeted biological treatment approved for asthma, has led to renewed optimism of outcome improvements in patients with allergic severe asthma. As severe asthma is a heterogeneous condition consisting of different phenotypes, the future of asthma management will likely involve phenotypic and potentially even genotypic characterization in selected cases in order to determine appropriate therapy and thus to provide the highest possible benefit, especially if specific responder phenotypes can be identified and selected for this highly specific treatment.
Abbreviations
Anti-immunoglobulin E
Body mass index
IgE receptor
Forced expiratory flow between 25% and 75%
Forced expiratory volume in the first second
Gastroesophageal reflux
Inhaled corticosteroids
Intensive care unit
Interleukin
Long-acting β 2 -agonist
Oral leukotriene receptor antagonist
Quality of life
Randomized controlled trials
Short-acting β 2 -agonists
Sleep-disordered breathing
O'Byrne PM, Pedersen S, Schatz M, Thoren A, Ekholm E, Carlsson LG, et al. The poorly explored impact of uncontrolled asthma. Chest. 2013;143:511–3.
Article PubMed Google Scholar
National Asthma Education and Prevention Program. Expert panel report 3 (EPR-3): guidelines for the diagnosis and Management of Asthma-Summary Report 2007. J Allergy Clin Immunol. 2007;120:S94–8.
Article Google Scholar
Hedlin G. Management of severe asthma in childhood-state of the art and novel perspectives. Pediatr Allergy Immunol. 2014;25:111–21.
Konradsen JR, Nordlund B, Lidegran M, Pedroletti C, Grönlund H, van Hage M, et al. Problematic severe asthma: a proposed approach to identifying children who are severely resistant to therapy. Pediatr Allergy Immunol. 2011;22:9–18.
Global Initiative for Asthma Report. Global strategy for asthma management and prevention (updated 2016). https://www.ginasthma.org . Accessed 07 June 2017.
Chung KF, Wenzel SE, Brozek JL, Bush A, Castro M, Sterk PJ, et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur Respir J. 2014;43:343–53.
Article CAS PubMed Google Scholar
Vandenplas Y, Rudolph CD, Di Lorenzo C, Hassall E, Liptak G, Mazur L, et al. Pediatric gastroesophageal reflux clinical practice guidelines: joint recommendations of the north American Society for Pediatric Gastroenterology, Hepatology, and nutrition (NASPGHAN) and the European Society for Pediatric Gastroenterology, Hepatology, and nutrition (ESPGHAN). J Pediatr Gastroenterol Nutr. 2009;49:498–507.
Lødrup Carlsen KC, Hedlin G, Bush A, Wennergren G, de Benedictis FM, De Jongste JC, et al. Assessment of problematic severe asthma in children. Eur Respir J. 2011;37:432–40.
Milgrom H, Berger W, Nayak A, Gupta N, Pollard S, McAlary M, et al. Treatment of childhood asthma with anti-immunoglobulin E antibody (omalizumab). Pediatrics. 2001;108:E36.
Bush A, Saglani S. Management of severe asthma in children. Lancet. 2010;376:814–5.
Article CAS PubMed PubMed Central Google Scholar
Lang A, Mowinckel P, Sachs-Olsen C, Riiser A, Lunde J, Carlsen KH, et al. Asthma severity in childhood, untangling clinical phenotypes. Pediatr Allergy Immunol. 2010;21:945–53.
Nordlund B, Konradsen JR, Pedroletti C, Kull I, Hedlin G. The clinical benefit of evaluating health-related quality-of-life in children with problematic severe asthma. Acta Paediatr. 2011;100:1454–60.
Dean BB, Calimlim BC, Sacco P, Aguilar D, Maykut R, Tinkelman D. Uncontrolled asthma: assessing quality of life and productivity of children and their caregivers using a cross-sectional internet-based survey. Health Qual Life Outcomes. 2010;8:6.
Juniper EF, Guyatt GH, Feeny DH, Ferrie PJ, Griffith LE, Townsend M. Measuring quality of life in children with asthma. Qual Life Res. 1996;5:35–46.
British Thoracic Society. Scottish Intercollegiate Guidelines Network. British guideline on the management of asthma, 2014. https://www.brit-thoracic.org.uk/guidelines-and-quality-standards/asthma-guideline . Accessed 13 Apr 2016.
Montella S, Baraldi E, Cazzato S, Aralla R, Berardi M, Brunetti LM, et al. Severe asthma features in children: a case-control online survey. Ital J Pediatr. 2016;42:9.
Article PubMed PubMed Central Google Scholar
Fitzpatrick AM, Gaston BM, Erzurum SC, Teague WG, National Institutes of Health/National Heart, Lung, and Blood Institute Severe Asthma Research Program. Features of severe asthma in school-age children: Atopy and increased exhaled nitric oxide. J Allergy Clin Immunol. 2006;118:1218–25.
Simon MR, Chinchilli VM, Phillips BR, Sorkness CA, Lemanske RF Jr, Szefler SJ, et al. Forced expiratory flow between 25% and 75% of vital capacity and FEV1/forced vital capacity ratio in relation to clinical and physiological parameters in asthmatic children with normal FEV1 values. J Allergy Clin Immunol. 2010;126:527–34.
Hedlin G, Bush A, Lødrup Carlsen K, Wennergren G, De Benedictis FM, Melén E, et al. Problematic severe asthma in children, not one problem but many: a GA2LEN initiative. Eur Respir J. 2010;36:196–201.
Fitzpatrick AM, Teague WG. Severe asthma in children: insights from the National Heart, Lung, and Blood Institute's severe asthma research program. Pediatr Allergy Immunol Pulmonol. 2010;23:131–8.
Konradsen JR, Caffrey Osvald E, Hedlin G. Update on the current methods for the diagnosis and treatment of severe childhood asthma. Expert Rev Respir Med. 2015;9:769–77.
Lang AM, Konradsen J, Carlsen KH, Sachs-Olsen C, Mowinckel P, Hedlin G, et al. Identifying problematic severe asthma in the individual child—does lung function matter? Acta Paediatr. 2010;99:404–10.
Rao DR, Gaffin JM, Baxi SN, Sheehan WJ, Hoffman EB, Phipatanakul WJ. The utility of forced expiratory flow between 25% and 75% of vital capacity in predicting childhood asthma morbidity and severity. Asthma. 2012;49:586–92.
Eid N, Yandell B, Howell L, Eddy M, Sheikh S. Can peak expiratory flow predict airflow obstruction in children with asthma? Pediatrics. 2000;105:354–8.
Cicutto LC, Chapman KR, Chamberlain D, Downey GP. Difficult asthma: consider all of the possibilities. Can Respir J. 2000;7:415–8.
Wener RR, Bel EH. Severe refractory asthma: an update. Eur Respir Rev. 2013;22:227–35.
Bracken M, Fleming L, Hall P, et al. The importance of nurse-led home visits in the assessment of children with problematic asthma. Arch Dis Child. 2009;94:780–4.
De Groot EP, Kreggemeijer WJ, Brand PL. Getting the basics right resolves most cases of uncontrolled and problematic asthma. Acta Paediatr. 2015;104:916–21.
Grimaldi-Bensouda L, Zureik M, Aubier M, Humbert M, Levy J, Benichou J, et al. Does omalizumab make a difference to the real-life treatment of asthma exacerbations? Results from a large cohort of patients with severe uncontrolled asthma. Chest. 2013;143:398–405.
American Lung Association Asthma Clinical Research Centers, Mastronarde JG, Anthonisen NR, Castro M, Holbrook JT, Leone FT, et al. Efficacy of esomeprazole for treatment of poorly controlled asthma. N Engl J Med. 2009;360:1487–9.
Article PubMed Central Google Scholar
Writing Committee for the American Lung Association Asthma Clinical Research Centers, Holbrook JT, Wise RA, Gold BD, Blake K, Brown ED, et al. Lansoprazole for children with poorly controlled asthma: a randomized controlled trial. JAMA 2012;307:373-381.
Wright AL, Holberg CJ, Martinez FD, Halonen M, Morgan W, Taussig LM. Epidemiology of physician-diagnosed allergic rhinitis in childhood. Pediatrics. 1994;94:895–901.
CAS PubMed Google Scholar
De Groot EP, Nijkamp A, Duiverman EJ, Brand PL. Allergic rhinitis is associated with poor asthma control in children with asthma. Thorax. 2012;67:582–7.
Rotiroti G, Roberts G, Scadding GK. Rhinitis in children: common clinical presentations and differential diagnoses. Pediatr Allergy Immunol. 2015;26:103–10.
Bousquet J, Khaltaev N, Cruz AA, Denburg J, Fokkens WJ, Togias A, et al. Allergic rhinitis and its impact on asthma (ARIA). 2008 update (in collaboration with the World Health Organization, GA(2)LEN and AllerGen). Allergy. 2008;63:S8–160.
Deliu M, Belgrave D, Simpson A, Murray CS, Kerry G, Custovic A. Impact of rhinitis on asthma severity in school-age children. Allergy. 2014;69:1515–21.
Brozek JL, Bousquet J, Baena-Cagnani CE, Bonini S, Canonica GW, Casale TB, et al. Allergic rhinitis and its impact on asthma (ARIA) guidelines: 2010 revision. J Allergy Clin Immunol. 2010;126:466–76.
Weinberger M, Abu-Hasan M. Pseudo-asthma: when cough, wheezing, and dyspnea are not asthma. Pediatrics. 2007;120:855–64.
De Groot EP, Duiverman EJ, Brand PL. Dysfunctional breathing in children with asthma: a rare but relevant comorbidity. Eur Respir J. 2013;41:1068–73.
Barker NJ, Jones M, O'Connell NE, Everard ML. Breathing exercises for dysfunctional breathing/hyperventilation syndrome in children. Cochrane Database Syst Rev. 2013;12:CD010376.
Google Scholar
Section on Pediatric Pulmonology, Subcommittee on Obstructive Sleep Apnea Syndrome, American Academy of Pediatrics. Clinical practice guideline: diagnosis and management of childhood obstructive sleep apnea syndrome. Pediatrics. 2002;109:704–12.
Goldstein NA, Aronin C, Kantrowitz B, Hershcopf R, Fishkin S, Lee H, Weaver DE, et al. The prevalence of sleep-disordered breathing in children with asthma and its behavioral effects. Pediatr Pulmonol. 2015;50:1128–36.
Ross KR, Storfer-Isser A, Hart MA, Kibler AM, Rueschman M, Rosen CL, et al. Sleep-disordered breathing is associated with asthma severity in children. J Pediatr. 2012;160:736–42.
Santamaria F, Montella S, Greco L, Valerio G, Franzese A, Maniscalco M, et al. Obesity duration is associated to pulmonary function impairment in obese subjects. Obesity (Silver Spring). 2011;19:1623–8.
Sivapalan P, Diamant Z, Ulrik CS. Obesity and asthma: current knowledge and future needs. Curr Opin Pulm Med. 2015;21:80–5.
Rasmussen F, Hancox RJ. Mechanisms of obesity in asthma. Curr Opin Allergy Clin Immunol. 2014;14:35–43.
Santamaria F, Montella S, Pietrobelli A. Obesity and pulmonary disease: unanswered questions. Obes Rev. 2012;13:822–33.
Lang JE, Hossain J, Holbrook JT, Teague WG, Gold BD, Wise RA, et al. Gastro-oesophageal reflux and worse asthma control in obese children: a case of symptom misattribution? Thorax. 2016;71:238–46.
Santamaria F, Montella S, De Stefano S, Sperlì F, Barbarano F, Valerio G. Relationship between exhaled nitric oxide and body mass index in children and adolescents. J Allergy Clin Immunol. 2005;116:1163–4.
Van Huisstede A, Rudolphus A, Castro Cabezas M, Biter LU, van de Geijn GJ, Taube C, et al. Effect of bariatric surgery on asthma control, lung function and bronchial and systemic inflammation in morbidly obese subjects with asthma. Thorax. 2015;70:659–67.
Katzmarzyk PT, Bouchard C. Where is the beef? Waist circumference is more highly correlated with BMI and total body fat than with abdominal visceral fat in children. Int J Obes. 2014;38:753–4.
Article CAS Google Scholar
De Groot EP, Duiverman EJ, Brand PL. Comorbidities of asthma during childhood: possibly important, yet poorly studied. Eur Respir J. 2010;36:671–8.
Sweeney J, Patterson CC, Menzies-Gow A, Niven RM, Mansur AH, Bucknall C, et al. Comorbidity in severe asthma requiring systemic corticosteroid therapy: cross-sectional data from the optimum patient care research database and the British thoracic difficult asthma registry. Thorax. 2016; https://doi.org/10.1136/thoraxjnl-2015-207630 .
Federal Drug Administration Advisory for Omalizumab. Available at: https://wayback.archive-it.org/7993/20170111075347/ . http://www.fda.gov/Drugs/DevelopmentApprovalProcess/HowDrugsareDevelopedandApproved/ApprovalApplications/TherapeuticBiologicApplications/default.htm . Accessed 4 Feb 2018.
European Medicines Agency: assessment report for Xolair. Available at: http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/000606/human_med_001162.jsp&mid=WC0b01ac058001d124 . Accessed 7 June 2017.
Chung KF. Targeting the interleukin pathway in the treatment of asthma. Lancet. 2015;386:1086–96.
Jensen RK, Plum M, Tjerrild L, Jakob T, Spillner E, Andersen GR. Structure of the omalizumab Fab. Acta Crystallogr F Struct Biol Commun. 2015;71:419–26.
Holgate S, Smith N, Massanari M, Jimenez P. Effects of omalizumab on markers of inflammation in patients with allergic asthma. Allergy. 2009;64:1728–36.
Hill DA, Siracusa MC, Ruymann KR, Tait Wojno ED, Artis D, Spergel JM. Omalizumab therapy is associated with reduced circulating basophil populations in asthmatic children. Allergy. 2014;69:674–7.
Humbert M, Beasley R, Ayres J, Slavin R, Hébert J, Bousquet J, et al. Benefits of omalizumab as add-on therapy in patients with severe persistent asthma who are inadequately controlled despite best available therapy (GINA 2002 step 4 treatment): INNOVATE. Allergy. 2005;60:309–16.
Normansell R, Walker S, Milan SJ, Walters EH, Nair P. Omalizumab for asthma in adults and children. Cochrane Database Syst Rev. 2014;1:CD003559.
Lai T, Wang S, Xu Z, Zhang C, Zhao Y, Hu Y, Cao C, et al. Long-term efficacy and safety of omalizumab in patients with persistent uncontrolled allergic asthma: a systematic review and meta-analysis. Sci Rep. 2015;5:8191.
Abraham I, Alhossan A, Lee CS, Kutbi H, MacDonald K. “real-life” effectiveness studies of omalizumab in adult patients with severe allergic asthma: systematic review. Allergy. 2015; https://doi.org/10.1111/all.12815 .
Lanier B, Bridges T, Kulus M, Taylor AF, Berhane I, Vidaurre CF. Omalizumab for the treatment of exacerbations in children with inadequately controlled allergic (IgE-mediated) asthma. J Allergy Clin Immunol. 2009;124:1210–6.
Solèr M, Matz J, Townley R, Buhl R, O'Brien J, Fox H, et al. The anti-IgE antibody omalizumab reduces exacerbations and steroid requirement in allergic asthmatics. Eur Respir J. 2001;18:254–61.
Holgate ST. Cytokine and anti-cytokine therapy for the treatment of asthma and allergic disease. Cytokine. 2004;28:152–7.
Odajima H, Ebisawa M, Nagakura T, Fujisawa T, Akasawa A, Ito K, et al. Omalizumab in Japanese children with severe allergic asthma uncontrolled with standard therapy. Allergol Int. 2015;64:364–70.
Busse WW, Morgan WJ, Gergen PJ, Mitchell HE, Gern JE, Liu AH, et al. Randomized trial of omalizumab (anti-IgE) for asthma in inner-city children. N Engl J Med. 2011;364:1005–15.
Teach SJ, Gill MA, Togias A, Sorkness CA, Arbes SJ Jr, Calatroni A, et al. Preseasonal treatment with either omalizumab or an inhaled corticosteroid boost to prevent fall asthma exacerbations. J Allergy Clin Immunol. 2015;136:1476–85.
Deschildre A, Marguet C, Salleron J, Pin I, Rittié JL, Derelle J, et al. Add-on omalizumab in children with severe allergic asthma: a 1-year real life survey. Eur Respir J. 2013;42:1224–33.
Deschildre A, Marguet C, Langlois C, Pin I, Rittié JL, Derelle J, et al. Real-life long-term omalizumab therapy in children with severe allergic asthma. Eur Respir J. 2015;46:856–9.
Rodrigo GJ, Neffen H. Systematic review on the use of omalizumab for the treatment of asthmatic children and adolescents. Pediatr Allergy Immunol. 2015;26:551–6.
Oba Y, Salzman GA. Cost-effectiveness analysis of omalizumab in adults and adolescents with moderate-to-severe allergic asthma. J Allergy Clin Immunol. 2004;114:265–9.
Campbell JD, Spackman DE, Sullivan SD. The costs and consequences of omalizumab in uncontrolled asthma from a USA payer perspective. Allergy. 2010;65:1141–8.
Busse W, Buhl R, Fernandez Vidaurre C, Blogg M, Zhu J, Eisner MD, et al. Omalizumab and the risk of malignancy: results from a pooled analysis. J Allergy Clin Immunol. 2012;129:983–9.
Lowe PJ, Renard D. Omalizumab decreases IgE production in patients with allergic (IgE-mediated) asthma; PKPD analysis of a biomarker, total IgE. Br J Clin Pharmacol. 2011;72:306–10.
Molimard M, Mala L, Bourdeix I, Le Gros V. Observational study in severe asthmatic patients after discontinuation of omalizumab for good asthma control. Respir Med. 2014;108:571–6.
Busse WW, Trzaskoma B, Omachi TA, Canvin J, Rosen K, Chipps BE, et al. Evaluating Xolair persistency of response after long-term therapy (XPORT). Am J Respir Crit Care Med. 2014;189:A6576.
Guilbert TW, Morgan WJ, Zeiger RS, Mauger DT, Boehmer SJ, Szefler SJ, et al. Long-term inhaled corticosteroids in preschool children at high risk for asthma. N Engl J Med. 2006;354:1985–97.
Akdis CA. Therapies for allergic inflammation: refining strategies to induce tolerance. Nat Med. 2012;18:736–49.
National Heart, Lung, and Blood Institute. Expert panel report 3: Guidelines for the diagnosis and management of asthma—full report 2007. Available at: https://www.nhlbi.nih.gov/files/docs/guidelines/asthgdln.pdf . Accessed 4 Feb 2018.
Joint Task Force on Practice Parameters, American Academy of Allergy, Asthma and Immunology, American College of Allergy, Asthma and Immunology, Joint Council of Allergy, Asthma and Immunolgy. Allergen immunotherapy: a practice parameter second update. J Allergy Clin Immunol. 2007;120:S25–85.
Zuberbier T, Bachert C, Bousquet PJ, Passalacqua G, Walter Canonica G, Merk H, et al. GA(2) LEN/EAACI pocket guide for allergen-specific immunotherapy for allergic rhinitis and asthma. Allergy. 2010;65:1525–30.
Pajno GB, Bernardini R, Peroni D, Arasi S, Martelli A, Landi M, et al. Clinical practice recommendations for allergen-specific immunotherapy in children: the Italian consensus report. Ital J Pediatr. 2017;43:13.
Pitsios C, Demoly P, Bilo MB, Gerth van Wijk R, Pfaar O, Sturm GJ, et al. Clinical contraindications to allergen immunotherapy: an EAACI position paper. Allergy. 2015;70:897–909.
Tsabouri S, Mavroudi A, Feketea G, Guibas GV. Subcutaneous and sublingual immunotherapy in allergic asthma in children. Front Pediatr. 2017;5:82.
Jutel M, Agache I, Bonini S, Burks AW, Calderon M, Canonica W, et al. International consensus on allergy immunotherapy. J Allergy Clin Immunol. 2015;136:556–68.
Hedlin G, van Hage M. The role of immunotherapy in the management of childhood asthma. Ther Adv Respir Dis. 2012;6:137–46.
Lambert N, Guiddir T, Amat F, Just J. Pre-treatment by omalizumab allows allergen immunotherapy in children and young adults with severe allergic asthma. Pediatr Allergy Immunol. 2014;25:829–32.
Kopp MV, Hamelmann E, Zielen S, Kamin W, Bergmann K-C, Sieder C. Combination of omalizumab and specific immunotherapy is superior to immunotherapy in patients with seasonal allergic rhinoconjunctivitis and co-morbid seasonal allergic asthma. Clin Exp Allergy. 2009;39:271–9.
Massanari M, Nelson H, Casale T, Busse W, Kianifard F, Geba GP. Effect of pretreatment with omalizumab on the tolerability of specific immunotherapy in allergic asthma. J Allergy Clin Immunol. 2010;125:383–9.
Stelmach I, Kaczmarek-Woźniak J, Majak P, Olszowiec-Chlebna M, Jerzynska J. Efficacy and safety of high-doses sublingual immunotherapy in ultra-rush scheme in children allergic to grass pollen. Clin Exp Allergy. 2009;39:401–8.
Download references
Acknowledgements
The authors gratefully thank Dr. Marco Maglione for his contribution in the clinical assessment of the described cases. Medical writing assistance was provided by Stephen Walters on behalf of City Hills Proofreading.
No funding was secured for this study.
Availability of data and materials
All relevant data and materials are published in the manuscript.
Author information
Authors and affiliations.
Department of Translational Medical Sciences, Federico II University, Via Sergio Pansini 5, 80131, Naples, Italy
Virginia Mirra, Silvia Montella & Francesca Santamaria
You can also search for this author in PubMed Google Scholar
Contributions
VM, SM and FS, authors of the current manuscript, declare that they have participated sufficiently in the work to take public responsibility for appropriate portions of the content. VM and SM carried out the initial investigations, drafted the initial manuscript, revised the manuscript, and approved the final manuscript as submitted. FS conceptualized and designed the study, and critically reviewed and approved the final manuscript as submitted. All authors read and approved the final manuscript.
Corresponding author
Correspondence to Francesca Santamaria .
Ethics declarations
Ethics approval and consent to participate.
This study was approved by the ethics committee “Carlo Romano”, Federico II University, Naples, Italy. Children’s parents/legal guardians gave informed written consent to participate. The description of our cases adheres to the CARE standards of reporting checklist.
Consent for publication
Children’s parents/legal guardians provided informed written consent for the case report to be published.
Competing interests
The authors declare that they have no competing interests to disclose. Authors have no financial relationships relevant to this article to disclose.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License ( http://creativecommons.org/licenses/by/4.0/ ), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated.
Reprints and permissions
About this article
Cite this article.
Mirra, V., Montella, S. & Santamaria, F. Pediatric severe asthma: a case series report and perspectives on anti-IgE treatment. BMC Pediatr 18 , 73 (2018). https://doi.org/10.1186/s12887-018-1019-9
Download citation
Received : 24 May 2016
Accepted : 29 January 2018
Published : 21 February 2018
DOI : https://doi.org/10.1186/s12887-018-1019-9
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Severe asthma
- Adolescents
- Asthma exacerbations
BMC Pediatrics
ISSN: 1471-2431
- Submission enquiries: [email protected]
- General enquiries: [email protected]
Case Study: Managing Severe Asthma in an Adult
—he follows his treatment plan, but this 40-year-old male athlete has asthma that is not well-controlled. what’s the next step.
By Kirstin Bass, MD, PhD Reviewed by Michael E. Wechsler, MD, MMSc
This case presents a patient with poorly controlled asthma that remains refractory to treatment despite use of standard-of-care therapeutic options. For patients such as this, one needs to embark on an extensive work-up to confirm the diagnosis, assess for comorbidities, and finally, to consider different therapeutic options.

Case presentation and patient history
Mr. T is a 40-year-old recreational athlete with a medical history significant for asthma, for which he has been using an albuterol rescue inhaler approximately 3 times per week for the past year. During this time, he has also been waking up with asthma symptoms approximately twice a month, and has had three unscheduled asthma visits for mild flares. Based on the National Asthma Education and Prevention Program guidelines , Mr. T has asthma that is not well controlled. 1
As a result of these symptoms, spirometry was performed revealing a forced expiratory volume in the first second (FEV1) of 78% predicted. Mr. T then was prescribed treatment with a low-dose corticosteroid, fluticasone 44 mcg at two puffs twice per day. However, he remained symptomatic and continued to use his rescue inhaler 3 times per week. Therefore, he was switched to a combination inhaled steroid and long-acting beta-agonist (LABA) (fluticasone propionate 250 mcg and salmeterol 50 mcg, one puff twice a day) by his primary care doctor.
Initial pulmonary assessment Even with this step up in his medication, Mr. T continued to be symptomatic and require rescue inhaler use. Therefore, he was referred to a pulmonologist, who performed the initial work-up shown here:
- Spirometry, pre-albuterol: FEV1 79%, post-albuterol: 12% improvement
- Methacholine challenge: PC 20 : 1.0 mg/mL
- Chest X-ray: Within normal limits
Continued pulmonary assessment His dose of inhaled corticosteroid (ICS) and LABA was increased to fluticasone 500 mcg/salmeterol 50 mcg, one puff twice daily. However, he continued to have symptoms and returned to the pulmonologist for further work-up, shown here:
- Chest computed tomography (CT): Normal lung parenchyma with no scarring or bronchiectasis
- Sinus CT: Mild mucosal thickening
- Complete blood count (CBC): Within normal limits, white blood cells (WBC) 10.0 K/mcL, 3% eosinophils
- Immunoglobulin E (IgE): 25 IU/mL
- Allergy-skin test: Positive for dust, trees
- Exhaled NO: Fractional exhaled nitric oxide (FeNO) 53 parts per billion (pbb)
Assessment for comorbidities contributing to asthma symptoms After this work-up, tiotropium was added to his medication regimen. However, he remained symptomatic and had two more flares over the next 3 months. He was assessed for comorbid conditions that might be affecting his symptoms, and results showed:
- Esophagram/barium swallow: Negative
- Esophageal manometry: Negative
- Esophageal impedance: Within normal limits
- ECG: Within normal limits
- Genetic testing: Negative for cystic fibrosis, alpha1 anti-trypsin deficiency
The ear, nose, and throat specialist to whom he was referred recommended only nasal inhaled steroids for his mild sinus disease and noted that he had a normal vocal cord evaluation.
Following this extensive work-up that transpired over the course of a year, Mr. T continued to have symptoms. He returned to the pulmonologist to discuss further treatment options for his refractory asthma.
Diagnosis Mr. T has refractory asthma. Work-up for this condition should include consideration of other causes for the symptoms, including allergies, gastroesophageal reflux disease, cardiac disease, sinus disease, vocal cord dysfunction, or genetic diseases, such as cystic fibrosis or alpha1 antitrypsin deficiency, as was performed for Mr. T by his pulmonary team.
Treatment options When a patient has refractory asthma, treatment options to consider include anticholinergics (tiotropium, aclidinium), leukotriene modifiers (montelukast, zafirlukast), theophylline, anti-immunoglobulin E (IgE) antibody therapy with omalizumab, antibiotics, bronchial thermoplasty, or enrollment in a clinical trial evaluating the use of agents that modulate the cell signaling and immunologic responses seen in asthma.
Treatment outcome Mr. T underwent bronchial thermoplasty for his asthma. One year after the procedure, he reports feeling great. He has not taken systemic steroids for the past year, and his asthma remains controlled on a moderate dose of ICS and a LABA. He has also been able to resume exercising on a regular basis.
Approximately 10% to 15% of asthma patients have severe asthma refractory to the commonly available medications. 2 One key aspect of care for this patient population is a careful workup to exclude other comorbidities that could be contributing to their symptoms. Following this, there are several treatment options to consider, as in recent years there have been several advances in the development of asthma therapeutics. 2
Treatment options for refractory asthma There are a number of currently approved therapies for severe, refractory asthma. In addition to therapy with ICS or combination therapies with ICS and LABAs, leukotriene antagonists have good efficacy in asthma, especially in patients with prominent allergic or exercise symptoms. 2 The anticholinergics, such as tiotropium, which was approved for asthma in 2015, enhance bronchodilation and are useful adjuncts to ICS. 3-5 Omalizumab is a monoclonal antibody against IgE recommended for use in severe treatment-refractory allergic asthma in patients with atopy. 2 A nonmedication therapeutic option to consider is bronchial thermoplasty, a bronchoscopic procedure that uses thermal energy to disrupt bronchial smooth muscle. 6,7
Personalizing treatment for each patient It is important to personalize treatment based on individual characteristics or phenotypes that predict the patient's likely response to treatment, as well as the patient's preferences and practical issues, such as adherence and cost. 8
In this case, tiotropium had already been added to Mr. T's medications and his symptoms continued. Although addition of a leukotriene modifier was an option for him, he did not wish to add another medication to his care regimen. Omalizumab was not added partly for this reason, and also because of his low IgE level. As his bronchoscopy was negative, it was determined that a course of antibiotics would not be an effective treatment option for this patient. While vitamin D insufficiency has been associated with adverse outcomes in asthma, T's vitamin D level was tested and found to be sufficient.
We discussed the possibility of Mr. T's enrollment in a clinical trial. However, because this did not guarantee placement within a treatment arm and thus there was the possibility of receiving placebo, he opted to undergo bronchial thermoplasty.
Bronchial thermoplasty Bronchial thermoplasty is effective for many patients with severe persistent asthma, such as Mr. T. This procedure may provide additional benefits to, but does not replace, standard asthma medications. During the procedure, thermal energy is delivered to the airways via a bronchoscope to reduce excess airway smooth muscle and limit its ability to constrict the airways. It is an outpatient procedure performed over three sessions by a trained physician. 9
The effects of bronchial thermoplasty have been studied in several trials. The first large-scale multicenter randomized controlled study was the Asthma Intervention Research (AIR) Trial , which enrolled patients with moderate to severe asthma. 10 In this trial, patients who underwent the procedure had a significant improvement in asthma symptoms as measured by symptom-free days and scores on asthma control and quality of life questionnaires, as well as reductions in mild exacerbations and increases in morning peak expiratory flow. 10 Shortly after the AIR trial, the Research in Severe Asthma (RISA) trial was conducted to evaluate bronchial thermoplasty in patients with more severe, symptomatic asthma. 11 In this population, bronchial thermoplasty resulted in a transient worsening of asthma symptoms, with a higher rate of hospitalizations during the treatment period. 11 Hospitalization rate equalized between the treatment and control groups in the posttreatment period, however, and the treatment group showed significant improvements in rescue medication use, prebronchodilator forced expiratory volume in the first second (FEV1) % predicted, and asthma control questionnaire scores. 11
The AIR-2 trial followed, which was a multicenter, randomized, double-blind, sham-controlled study of 288 patients with severe asthma. 6 Similar to the RISA trial, patients in the treatment arm of this trial experienced an increase in adverse respiratory effects during the treatment period, the most common being airway irritation (including wheezing, chest discomfort, cough, and chest pain) and upper respiratory tract infections. 6
The majority of adverse effects occurred within 1 day of the procedure and resolved within 7 days. 6 In this study, bronchial thermoplasty was found to significantly improve quality of life, as well as reduce the rate of severe exacerbations by 32%. 6 Patients who underwent the procedure also reported fewer adverse respiratory effects, fewer days lost from work, school, or other activities due to asthma, and an 84% risk reduction in emergency department visits. 6
Long-term (5-year) follow-up studies have been conducted for patients in both the AIR and the AIR-2 trials. In patients who underwent bronchial thermoplasty in either study, the rate of adverse respiratory effects remained stable in years 2 to 5 following the procedure, with no increase in hospitalizations or emergency department visits. 7,12 Additionally, FEV1 remained stable throughout the 5-year follow-up period. 7,12 This finding was maintained in patients enrolled in the AIR-2 trial despite decreased use of daily ICS. 7
Bronchial thermoplasty is an important addition to the asthma treatment armamentarium. 7 This treatment is currently approved for individuals with severe persistent asthma who remain uncontrolled despite the use of an ICS and LABA. Several clinical trials with long-term follow-up have now demonstrated its safety and ability to improve quality of life in patients with severe asthma, such as Mr. T.
Severe asthma can be a challenge to manage. Patients with this condition require an extensive workup, but there are several treatments currently available to help manage these patients, and new treatments are continuing to emerge. Managing severe asthma thus requires knowledge of the options available as well as consideration of a patient's personal situation-both in terms of disease phenotype and individual preference. In this case, the patient expressed a strong desire to not add any additional medications to his asthma regimen, which explained the rationale for choosing to treat with bronchial thermoplasty. Personalized treatment necessitates exploring which of the available or emerging options is best for each individual patient.
Published: April 16, 2018
- 1. National Asthma Education and Prevention Program: Asthma Care Quick Reference.
- 2. Olin JT, Wechsler ME. Asthma: pathogenesis and novel drugs for treatment. BMJ . 2014;349:g5517.
- 3. Boehringer Ingelheim. Asthma: U.S. FDA approves new indication for SPIRIVA Respimat [press release]. September 16, 2015.
- 4. Peters SP, Kunselman SJ, Icitovic N, et al. Tiotropium bromide step-up therapy for adults with uncontrolled asthma. N Engl J Med . 2010;363:1715-1726.
- 5. Kerstjens HA, Engel M, Dahl R. Tiotropium in asthma poorly controlled with standard combination therapy. N Engl J Med . 2012;367:1198-1207.
- 6. Castro M, Rubin AS, Laviolette M, et al. Effectiveness and safety of bronchial thermoplasty in the treatment of severe asthma: a multicenter, randomized, double-blind, sham-controlled clinical trial. Am J Respir Crit Care Med . 2010;181:116-124.
- 7. Wechsler ME, Laviolette M, Rubin AS, et al. Bronchial thermoplasty: long-term safety and effectiveness in patients with severe persistent asthma. J Allergy Clin Immunol . 2013;132:1295-1302.
- 8. Global Initiative for Asthma: Pocket Guide for Asthma Management and Prevention (for Adults and Children Older than 5 Years).
- 10. Cox G, Thomson NC, Rubin AS, et al. Asthma control during the year after bronchial thermoplasty. N Engl J Med . 2007;356:1327-1337.
- 11. Pavord ID, Cox G, Thomson NC, et al. Safety and efficacy of bronchial thermoplasty in symptomatic, severe asthma. Am J Respir Crit Care Med . 2007;176:1185-1191.
- 12. Thomson NC, Rubin AS, Niven RM, et al. Long-term (5 year) safety of bronchial thermoplasty: Asthma Intervention Research (AIR) trial. BMC Pulm Med . 2011;11:8.
Treatable traits and future exacerbation risk in severe asthma, baker’s asthma, the long-term trajectory of mild asthma, age, gender, & systemic corticosteroid comorbidities, ask the expert: william busse, md, challenges the current definition of the atopic march, considering the curveballs in asthma treatment, do mucus plugs play a bigger role in chronic severe asthma than previously thought, an emerging subtype of copd is associated with early respiratory disease.

- Case Report
- Open access
- Published: 16 December 2008
Bronchial carcinoid in a 39-year-old man treated for bronchial asthma: a case report
- Justyna Emeryk 1 ,
- Elżbieta Czekajska-Chehab 2 ,
- Elżbieta Korobowicz 1 ,
- Marta Korbel 3 ,
- Irena Węgrzyn-Szkutnik 1 &
- Janusz Milanowski 1
Cases Journal volume 2 , Article number: 7414 ( 2008 ) Cite this article
14k Accesses
3 Citations
Metrics details
A case study of 39-year old man with persistent wheezing, episodes of haemoptysis and dry cough unsuccessfully treated with inhaled beta2-agonists and steroids for about 10 months. Chest radiograph revealed a disproportion in dimensions between both lungs, with the left one being smaller than the right one. Spirometry demonstrated a restrictive pattern. During bronchoscopy, a polypoid endobronchial tumor, localized in the left main bronchus, completely occluding its lumen, was found. The tumor was diagnosed as carcinoid. In this case, due to the lack of characteristic symptoms, diagnosis of carcinoid was delayed. Patients unsuccessfully treated for bronchial asthma or chronic obstructive pulmonary disease should undergo bronchoscopic examination.
Case presentation
A 39-year-old white man, working as a house builder, was admitted to our pulmonary ward in August 2007 with an eleven-month history of persistent wheezing, heard not only during auscultation but also by patient himself. This wheezing had no correlation with physical exertion. He also reported a few episodes of haemoptysis within last 7 months and dry cough for about a month. Patient's weight was 81.5 kg, his height was 180 cm. Except from nodular goitre (in euthyreosis), the patient reported no other complaints or diseases in his medical history. His family history was not clinically significant. He had a 10 pack-years smoking history, until the age of 31 when he stopped smoking. He drank alcohol occasionally. His symptoms were unsuccessfully treated with inhaled β 2 -agonists and steroids for about 10 months. Apart from these medications, he took no other drugs. On auscultation, there were wheezes in upper fields of both lungs. The chest radiograph (figure 1 ) showed a disproportion in dimensions and vascular markings between both lungs- the left one was significantly smaller and had decreased vascular markings in comparison to the right one- these findings might be the expression of left lung hypoplasia. The heart silhouette was discretely displaced to the left side.
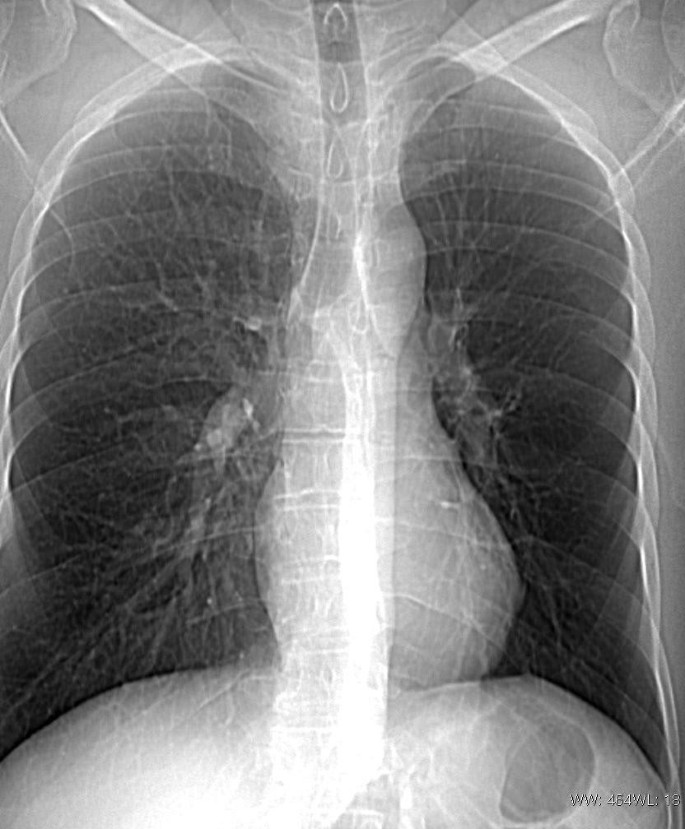
Chest radiograph performed at admission to the hospital .
Spirometry performed during hospitalization in our clinic showed a restrictive pattern (figure 2 ) with forced expiratory volume in one second (FEV 1 ) of 59% predicted and forced vital capacity (FVC) of 66% predicted. The FEV 1 /FVC ratio was 70% predicted.
Flow-volume curve showing a restrictive pattern performed at admission to the hospital .
Bronchoscopy was performed, revealing a polypoid endobronchial tumor, localized in the left main bronchus (2 cm from tracheal bifurcation), completely occluding its lumen. The tumor was biopsied and the specimen was sent to histological examination which gave the diagnosis of bronchial carcinoid.
Thoracic computed tomography showed a polypoid mass in the left main bronchus with a diameter of about 20 mm. The tumor showed very high enhancement after contrast medium administration: from 9 HU (Hounsfield units) (figure 3a ) to 119 HU in arterial phase (figure 3b ) and 47 HU in parenchymal phase (figure 3c ), which is characteristic for carcinoid.
High enhancement of the tumor (arrow) after contrast medium administration: on the left- before administration of contrast medium, in the middle-after administration; arterial phase, on the right-parenchymal phase .
Hilar and mediastinal nodes were not enlarged. In virtual bronchoscopy, as well as in transparent reconstruction of the airways, a total occlusion of the left main bronchus by the tumor could be observed (Figure 4 and 5 ).

Total occlusion of the left main bronchus by the tumor (arrow) .
Amputation of the left main bronchus by the tumor (arrow) .
The patient was transferred to thoracic surgery ward for surgical treatment. He underwent left pulmonectomy and histopathological analysis of post-operative material confirmed the diagnosis of bronchial carcinoid. There were no complications during post-operative period and the patient was discharged from hospital 10 days after the operation. Then, the patient went abroad and was lost for follow-up until May 2008. On control visit in May, he was in very good condition, presenting no signs of pulmonary disease. On physical examination, except from diminished breath sounds and dullness to percussion over left lung (due to pulmonectomy and consequent pleural fluid accumulation) no abnormalities were found. In control computed tomography there were no signs of recurrence of the disease.
Pulmonary carcinoids comprise 1-2% of all lung tumors. They may develop in many locations in the body but most often, they are found in small intestine (26%), respiratory system (25%) and appendix (19%) [ 1 ].
Carcinoid tumors are classified as typical or atypical according to histopathological criteria. This division has a strict extrapolation to survival rates: in case of typical pulmonary carcinoids 5-year survival rate is over 90%, in atypical ones- it is within the range of 40-60% [ 2 ].
The following symptoms are observed most often in patients with carcinoid localized in respiratory system: haemoptysis, cough, recurrent pulmonary infections, fever, chest discomfort, unilateral wheezing and shortness of breath [ 3 ].
Due to the lack of characteristic symptoms, diagnosis of pulmonary carcinoid is delayed and patients are often misdiagnosed with asthma or chronic obstructive pulmonary disease. According to three studies, there is an average delay of, respectively, 19, 13 and 10 months from the first symptoms to the final diagnosis of carcinoid [ 4 – 6 ]. Although general prognosis for patients with this neoplasm is quite favorable, it is obvious that the earlier diagnosis is made, the chances for radical treatment increase.
As the majority of pulmonary carcinoids (70%) are located in the main or lobar bronchi [ 7 ], they are within the reach of a bronchoscope. According to British Thoracic Society, flexible bronchoscopy is a safe procedure and there are no controlled studies concerning the factors disqualifying a patient from it [ 8 ]. Bronchoscopy cannot replace computed tomography, these both procedures are complementary, but to be able to orientate the treatment and prognosis, we have to know the histopathological diagnosis and the specimens for examination can be easily and safely provided by bronchoscopy.
This case is a good example of misdiagnosis of the disease. Extended clinical diagnosis, including computed tomography and bronchoscopy, should be considered in all cases of bronchial asthma or chronic obstructive pulmonary disease which do not respond to standard treatment.
"Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal."
Lips CJM, Lentjes EGWM, Höppener JWM: The spectrum of carcinoid tumours and carcinoid syndromes. Ann Clin Biochem. 2003, 40: 612-27. 10.1258/000456303770367207.
Article PubMed Google Scholar
Kulke MH, Mayer RJ: Carcinoid tumours. N Engl J Med. 1999, 340: 858-68. 10.1056/NEJM199903183401107.
Article CAS PubMed Google Scholar
Zuetenhorst JM, Taal BG: Metastatic carcinoid tumours: a clinical review. Oncologist. 2005, 10: 123-31. 10.1634/theoncologist.10-2-123.
Garcia-Rio FJ, Talavera IG, Rubio BG, Prados C, Rodriguez P: Clinical study of 20 cases of bronchial carcinoid. An Med Interna. 1992, 9 (1): 9-13.
CAS PubMed Google Scholar
Blondal T, Grimelius L, Nou E, Wilander E, Aberg T: Argyrophil carcinoid tumours of the lung. Incidence, clinical study and follow-up of 46 patients. Chest. 1980, 78 (6): 840-4. 10.1378/chest.78.6.840.
Van Damme H, Layachi N, Hermans G, Dekoster G: Carcinoid tumors of the bronchi. Our experience of these during the last 10 years, with a review of the literature. Acta Chir Belg. 1989, 89 (3): 166-74.
Hage R, de la Rivière AB, Seldenrijk CA, Bosch van den JMM: Update in pulmonary carcinoid tumours: a review article. Ann Surg Oncol. 2003, 10: 697-704. 10.1245/ASO.2003.09.019.
British Thoracic Society guidelines on diagnostic flexible bronchoscopy. Thorax. 2001, 56 (suppl 1): 1-21.
Download references
Author information
Authors and affiliations.
Chair and Department of Pulmonology, Oncology and Allergology, Medical University of Lublin, Jaczewski Street 8, 20-950, Lublin, Poland
Justyna Emeryk, Elżbieta Korobowicz, Irena Węgrzyn-Szkutnik & Janusz Milanowski
First Department of Radiology, Medical University of Lublin, Jaczewski Street 8, 20-950, Lublin, Poland
Elżbieta Czekajska-Chehab
Chair and Department of Clinical Pathomorphology, Medical University of Lublin, Jaczewski Street 8, 20-950, Lublin, Poland
Marta Korbel
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Justyna Emeryk .
Additional information
Competing interests.
The authors declare that they have no competing interests.
Authors' contributions
JE performed acquisition of data, review of literature and wrote the paper. ECC performed computed tomography imagings and prepared virtual bronchoscopy reconstructions. EK performed histopathological examination of the tumour. MK was responsible for patient care, follow-up and data collection. IWS was responsible for patient care and drafting of paper. JM performed acquisition of data, revised the manuscript and provided general support as the head of department. All authors read and approved the final manuscript.
Microscopic imaging of bronchial carcinoid (hematoxylin and eosin staining) .
Immunohistochemical staining positive for chromogranine A .
Immunohistochemical staining positive for synaptophysin .
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Authors’ original file for figure 1
Authors’ original file for figure 2, authors’ original file for figure 3, authors’ original file for figure 4, authors’ original file for figure 5, authors’ original file for figure 6, authors’ original file for figure 7, authors’ original file for figure 8, authors’ original file for figure 9, rights and permissions.
This article is published under license to BioMed Central Ltd. This is an open access article distributed under the terms of the Creative Commons Attribution License ( http://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Reprints and permissions
About this article
Cite this article.
Emeryk, J., Czekajska-Chehab, E., Korobowicz, E. et al. Bronchial carcinoid in a 39-year-old man treated for bronchial asthma: a case report. Cases Journal 2 , 7414 (2008). https://doi.org/10.1186/1757-1626-2-7414
Download citation
Received : 19 November 2008
Accepted : 16 December 2008
Published : 16 December 2008
DOI : https://doi.org/10.1186/1757-1626-2-7414
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Chronic Obstructive Pulmonary Disease
- Pleural Fluid
- Restrictive Pattern
- Left Main Bronchus
- British Thoracic Society
Cases Journal
ISSN: 1757-1626

COMMENTS
Case Presentation. History of Present Illness: A 33-year-old white female presents after admission to the general medical/surgical hospital ward with a chief complaint of shortness of breath on exertion. She reports that she was seen for similar symptoms previously at her primary care physician's office six months ago.
Clinical case study - asthma . 2019 . Clinical Case Study - Asthma. pdf. Clinical Case Study - Asthma. 6.34 MB. Resource information. Respiratory conditions. Asthma; Respiratory topics. Disease management; Diagnosis; Type of resource. Presentation . Author(s) Jaime Correia de Sousa Ioanna Tsiligianni Miguel Román Rodriguez
Case Presentation. The patient is a 60-year-old white female presenting to the emergency department with acute onset shortness of breath. Symptoms began approximately 2 days before and had progressively worsened with no associated, aggravating, or relieving factors noted. She had similar symptoms approximately 1 year ago with an acute, chronic ...
Quick-Relief Medication for All Patients: SABA as needed for symptoms. Intensity of treatment depends on severity of symptoms: up to 3 treatments at 20-minute intervals as needed. Short course of systemic oral corticosteroids may be needed. Caution: Increasing of b-agonist or use >2x/week for symptoms control indicates.
Supported self-management reduces asthma morbidity. The lady in this case study is struggling to maintain control of her asthma within the context of her busy professional and domestic life.
CASE PRESENTATION: A 58 years old Caucasian male, non smoker, with late onset allergic asthma was referred to our pulmonary rehabilitation clinic because of deconditioning, wheezing and recurrent asthma exacerbations despite treatment with budesonide-formoterol 200/6 mcg b.i.d., montelukast 10 mg q.i.d. He had daily complaints of dyspnea both at rest and on exertion using almost 200 ...
MANAGEMENT OF A CASE OF UNCONTROLLED BRONCHIAL ASTHMA. INTRODUCTION: poor asthma control.1 Our case reports an undiagnosed comorbid depression as cause of persistent uncontrolled asthma. A 58 years old Caucasian male, non smoker, with late onset allergic asthma was referred to our CASE PRESENTATION: pulmonary rehabilitation clinic because of ...
Moreover, in a real-life study of 104 children and adolescents with severe allergic refractory asthma followed over 1 year, treatment with omalizumab resulted in good asthma control in 67% of the cases (p < 0.001), while FEV 1 improved by 4.9% (p = 0.02) and exacerbation rates and healthcare utilisation decreased approximately by 30% (p < 0.001) .
Bronchial asthma is a disorder of the airways causing swelling and narrowing; which leads to wheezing, shortness of breath, chest tightness, and coughing. The impact of asthma in children depends on complex interaction between disease severity, reaction of children towards disease, treatment eficiency, social roles, and social environment.
A Case of Severe Acute Exacerbation of Bronchial Asthma Treated with Low Minute Ventilation - A Case Report - ... A Case Report -.pdf. ... In the first study, 88 ICU admissions for severe asthma ...
Abstract. Bronchial asthma is a common disease in this day and age, but despite our vast knowledge on it, it still represents a great challenge for contemporary medicine. The paper describes a ...
Case Summary. A 55-year-old Japanese female presented with severe bronchial asthma controlled with tacrolimus. She had been diagnosed with bronchial asthma during childhood. Her asthma worsened ...
Only approximately 25% of male subjects and 10% of female subjects with asthma developed low ratios. Subjects with a low ratio also had. more rapid decline in FEV1 and FEV1/FVC ratio from age 9 years to age 26 years, and they were less responsive to b-agonists at 26 years. More than half of subjects with.
The majority of adverse effects occurred within 1 day of the procedure and resolved within 7 days. 6. In this study, bronchial thermoplasty was found to significantly improve quality of life, as ...
A case study of 39-year old man with persistent wheezing, episodes of haemoptysis and dry cough unsuccessfully treated with inhaled beta2-agonists and steroids for about 10 months. Chest radiograph revealed a disproportion in dimensions between both lungs, with the left one being smaller than the right one. Spirometry demonstrated a restrictive pattern. During bronchoscopy, a polypoid ...
To The Editor, The Global Initiative for Asthma (GINA) 2021 update was published on the 28th of April, 2021. [1]. There are significant changes, including treatment of mild asthma, the role of ...
Summary. This chapter presents a case study of a 10-year-old girl with moderate intermittent asthma diagnosed at age 4 and was admitted to the pediatric intensive care unit with status asthmaticus. The case study includes details about history of present illness, past medical history, past surgical history, family history, and current status.
This prospective study was done at Dhaka Medical College Hospital from January 1997 to January 1998. Total 30 patients of bronchial asthma were included in this study. Diagnosis was established on ...